Abrysvo Dosage
Generic name: RECOMBINANT STABILIZED RSV A PREFUSION F ANTIGEN 0.06mg in 0.5mL, RECOMBINANT STABILIZED RSV B PREFUSION F ANTIGEN 0.06mg in 0.5mL;
Dosage form: injection
Drug class: Viral vaccines
Medically reviewed by Drugs.com. Last updated on Dec 22, 2023.
Dose and Schedule
Administer a single dose (approximately 0.5 mL) of ABRYSVO intramuscularly.
Preparation for Administration
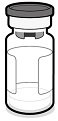
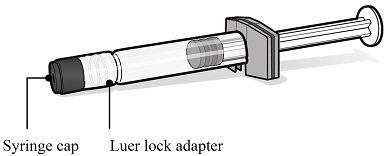
ABRYSVO is supplied in a kit that includes a vial of Lyophilized Antigen Component (a sterile white powder), a prefilled syringe containing Sterile Water Diluent Component and a vial adapter.
|
Vial of Lyophilized Antigen Component
|
Syringe of Sterile Water Diluent Component
|
Vial Adapter
|
 |
 |
 |
To form ABRYSVO, reconstitute the Lyophilized Antigen Component with the accompanying Sterile Water Diluent Component as described in the panels below.
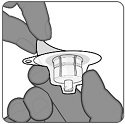 |
Step 1. Preparation of vial and vial adapter
- •
- Remove plastic flip off cap from vial and cleanse the rubber stopper.
- •
- Without removing the vial adapter from its packaging, peel off the top cover.
|
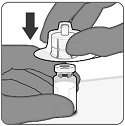 |
Step 2. Attachment of vial adapter
- •
- Hold the base of the vial on a flat surface.
- •
- Keep the vial adapter in the packaging and orient it vertically over the center of the vial so that the adapter spike aligns with the center of the vial’s rubber stopper.
- •
- Connect the vial adapter to the vial with a straight downward push. The vial adapter will lock into place.
- •
- Do not push vial adapter in at an angle as this may result in leaking during use.
- •
- Remove the vial adapter packaging.
|
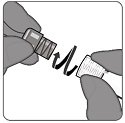 |
Step 3. Removal of syringe cap
- •
- For all syringe assembly steps, hold the syringe only by the Luer lock adapter located at the tip of the syringe. This will prevent the Luer lock adapter from detaching during use.
- •
- Remove the syringe cap by slowly turning the cap counter-clockwise while holding the Luer lock adapter.
|
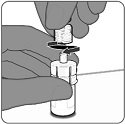 |
Step 4. Connection of syringe to vial adapter
- •
- Hold the syringe’s Luer lock adapter and connect it to the vial adapter by turning clockwise.
- •
- Stop turning when you feel resistance, overtightening the syringe may result in leaking during use.
- •
- Once the syringe is securely attached to the vial adapter, there will be a small space between the top of the vial adapter and the Luer lock adapter of the syringe.
|
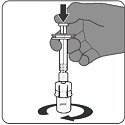 |
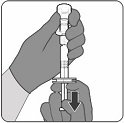
Step 5. Reconstitution of Lyophilized Antigen Component to form ABRYSVO
- •
- Inject the entire contents of the syringe containing the Sterile Water Diluent Component into the vial.
- •
- Do not remove the empty syringe.
- •
- While holding the plunger rod down, gently swirl the vial in a circular motion until the powder is completely dissolved (less than 1 minute).
- •
- Do not shake.
|
 |
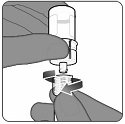
Step 6. Withdrawal of reconstituted vaccine
- •
- Invert the vial completely with the vial adapter and syringe still attached.
- •
- Slowly withdraw the entire contents into the syringe to ensure an approximately 0.5 mL dose of ABRYSVO for administration.
- •
- Do not pull the plunger rod out.
|
 |
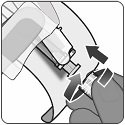
Step 7. Disconnection of syringe
- •
- Hold the Luer lock adapter of the syringe and disconnect the syringe from the vial adapter by turning counter-clockwise.
|
 |
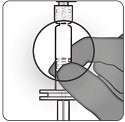
Step 8. Attachment of needle
- •
- Attach a sterile needle suitable for intramuscular injection to the syringe containing ABRYSVO.
|
 |
Step 9. Visual inspection
- •
- ABRYSVO is a clear and colorless solution.
- •
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard if either condition is present.
|
Administration
For intramuscular injection only
After reconstitution, administer ABRYSVO immediately or store at room temperature [15°C to 30°C (59°F to 86°F)] and use within 4 hours. Discard reconstituted vaccine if not used within 4 hours.
Frequently asked questions
More about Abrysvo (rsv vaccine, pref a-pref b, recombinant)
Patient resources
Professional resources
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
Medical Disclaimer