Thrombin-JMI Prescribing Information
Package insert / product label
Generic name: thrombin, topical (bovine)
Dosage form: topical kit
Drug class: Miscellaneous coagulation modifiers
Medically reviewed by Drugs.com. Last updated on Apr 15, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- References
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
THROMBIN-JMI® (Thrombin, Topical (Bovine) U.S.P.), Solution for topical use
Initial U.S. Approval: 1986
WARNING: SEVERE BLEEDING AND THROMBOSIS COMPLICATIONS
See full prescribing information for complete boxed warning
- •
- THROMBIN-JMI can cause fatal severe bleeding or thrombosis. Thrombosis may result from the development of antibodies against bovine thrombin. Bleeding may result from the development of antibodies against factor V. These may cross-react with human factor V and lead to its deficiency. (5.2, 5.3)
- •
- Do not re-expose patients to THROMBIN-JMI if there are known or suspected antibodies to bovine thrombin and/or factor V. (4, 5.3)
- •
- Monitor patients for abnormal coagulation laboratory values, bleeding, or thrombosis. (5.3)
Recent Major Changes
Indications and Usage for Thrombin-JMI
- •
- THROMBIN-JMI is a topical thrombin indicated to aid hemostasis whenever oozing blood and minor bleeding from capillaries and small venules is accessible and control of bleeding by standard surgical techniques (such as suture, ligature, or cautery) is ineffective or impractical. (1)
- •
- THROMBIN-JMI may be used in conjunction with an absorbable gelatin sponge, USP. (1)
Thrombin-JMI Dosage and Administration
For topical use on the surface of bleeding tissue only. Do not inject. (2)
- •
- May be reconstituted with sterile isotonic saline at a recommended concentration of 1,000 to 2,000 International Units (IU)/mL or may be used in dry form on oozing surfaces. (2)
Dosage Forms and Strengths
- •
- THROMBIN-JMI is available as 5,000 IU vial with 5 mL diluent and 20,000 IU vial with 20 mL diluent. (3)
- •
- THROMBIN –JMI Pump Spray Kit is available as 20,000 IU vial with 20 mL diluent, spray pump and actuator. (3)
- •
- THROMBIN-JMI Syringe Spray Kit is available as 5,000 IU vial with 5 mL diluent, spray tip and syringe and 20,000 IU vial with 20 mL diluent, spray tip and syringe. (3)
- •
- THROMBIN-JMI Epistaxis Kit is available as 5,000 IU vial with 5 mL diluent, nasal drug delivery device and syringe. (3)
- •
- THROMBIN-JMI is available in the Gelfoam-JMI™ Powder Kit (Gelfoam® Absorbable Gelatin Powder and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU vial with 5 mL diluent. (3)
- •
- THROMBIN-JMI is available in the Gelfoam-JMI™ Sponge Kit (Gelfoam® Absorbable Gelatin Sponge, USP and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU vial with 5 mL diluent. (3)
- •
- THROMBIN-JMI® 5,000 IU Syringe Spray Kit is supplied in the GEL-FLOW™ Kit: GEL-FLOW™ Kit (GEL-FLOW™ NT Absorbable Gelatin Powder and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU Syringe Spray Kit). (3)
Contraindications
- •
- Do not inject directly into the circulatory system. (4, 5.2)
- •
- Do not re-expose patients to THROMBIN-JMI if there are known or suspected antibodies to bovine thrombin and/or factor V. (4, 5.3)
- •
- Do not administer to patients with a history of hypersensitivity to THROMBIN-JMI, its components and/or material of bovine origin. (4, 5.1)
- •
- Do not use for treatment of severe or brisk arterial bleeding. (4)
Warnings and Precautions
- •
- Allergic reactions, including anaphylactic/anaphylactoid reactions, have been reported following administration of THROMBIN-JMI. (5.1)
- •
- Institute intensive supportive measures and treat individual symptoms. Secure the airway and establish adequate respiratory exchange. (5.1)
- •
- THROMBIN-JMI causes thrombosis if it enters the circulatory system. Apply topically. DO NOT INJECT. (5.2)
- •
- Inhibitory antibodies may develop in patients and interfere with hemostasis. Monitor patients for abnormal coagulation laboratory values, bleeding, or thrombosis. (5.3)
Adverse Reactions/Side Effects
Most common adverse reactions (incidence ≥ 2%) are hypersensitivity, bleeding, anemia, post-operative wound infection, thromboembolic events, hypotension, pyrexia, tachycardia and thrombocytopenia. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Pfizer, Inc. at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2024
Related/similar drugs
thrombin topical, Evarrest, Recothrom
Full Prescribing Information
WARNING: SEVERE BLEEDING AND THROMBOSIS COMPLICATIONS
- •
- THROMBIN-JMI® can cause fatal severe bleeding or thrombosis. Thrombosis may result from the development of antibodies against bovine thrombin. Bleeding may result from the development of antibodies against factor V. These may cross-react with human factor V and lead to its deficiency. (5.2, 5.3)
- •
- Do not re-expose patients to THROMBIN-JMI if there are known or suspected antibodies to bovine thrombin and/or factor V. (4, 5.3)
- •
- Monitor patients for abnormal coagulation laboratory values, bleeding, or thrombosis. (5.3)
1. Indications and Usage for Thrombin-JMI
THROMBIN-JMI is topical bovine thrombin indicated to aid hemostasis whenever oozing blood and minor bleeding from capillaries and small venules is accessible and control of bleeding by standard surgical techniques (such as suture, ligature, or cautery) is ineffective or impractical.
In various types of surgeries, solutions of THROMBIN-JMI may be used in conjunction with an Absorbable Gelatin Sponge, USP for hemostasis.
2. Thrombin-JMI Dosage and Administration
For topical use on the surface of bleeding tissue only. Do not inject.
2.1 Reconstitution
- •
- For routine use, reconstitute THROMBIN-JMI with sterile isotonic saline at a recommended concentration of 1,000 to 2,000 IU per mL.
- •
- Where bleeding is profuse, as from abraded surfaces of liver or spleen, concentrations of 1,000 IU per mL may be required.
- •
- For general use in plastic surgery, dental extractions, skin grafting, etc. solutions containing approximately 100 IU per mL are frequently used. Prepare intermediate strengths to suit the needs of the case by diluting the contents of the THROMBIN-JMI container with an appropriate volume of sterile isotonic saline. THROMBIN-JMI can be used in a dry form on oozing surfaces.
- •
- In instances where a concentration of approximately 1,000 IU per mL is desired, the contents of the vial of sterile isotonic saline diluent may be transferred into the THROMBIN-JMI container with a sterile syringe or sterile transfer device.
- •
- For use with GEL-FLOW™ NT, reconstitute THROMBIN-JMI 5,000 IU powder with 5 mL sterile isotonic saline and use THROMBIN-JMI thrombin solution according to the directions for use in the GEL-FLOW™ NT package insert.
If the transfer device is used for reconstitution, transfer the diluent in the following manner:
- 1.
- Remove the plastic cap off of the diluent vial.
- 2.
- Remove the Tyvek cover from the transfer device container. Do not remove the device from the package.
- 3.
- Seat the blue end of the device on the diluent vial, pushing down until the spike penetrates the diaphragm and the device snaps in place.
- 4.
- Flip the plastic cover off on the THROMBIN-JMI container. DO NOT REMOVE THE ALUMINUM SEAL.
- 5.
- Remove the plastic package from the transfer device. Do not touch the exposed end of the device.
- 6.
- Invert the vial of diluent and insert the clear end of the transfer device into the diaphragm of the THROMBIN-JMI container.
CAUTION: Solutions should be used promptly upon removal from the container. However, the solution may be refrigerated at 2°C – 8°C for up to 24 hours, or may be stored at room temperature for up to 8 hours after reconstitution.
THROMBIN-JMI PUMP SPRAY KIT
Refer to THROMBIN-JMI Pump Spray Kit instructions for spray pump assembly and use.
Each spray kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray pump and actuator.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on inner tray to expose sterile contents.
- 3.
- Reconstitute the THROMBIN-JMI to desired potency by introducing sterile isotonic saline with a sterile syringe or a sterile transfer device. If the transfer device is used, follow the previously described procedure.
- 4.
- When the THROMBIN-JMI is completely dissolved, open vial by flipping up metal and tearing counterclockwise.
- 5.
- Remove the rubber diaphragm from vial. Remove pump with protective cap from tray and snap onto vial.
- 6.
- Remove protective cap and attach actuator.
- 7.
- To spray, hold vial upright or at a slight angle. Several strokes of the pump will be required to expel the solution.
- 8.
- Discard unused contents and pump: DO NOT TRANSFER SPRAY PUMP TO ANOTHER VIAL.
THROMBIN-JMI SYRINGE SPRAY KIT
Refer to THROMBIN-JMI Syringe Spray Kit instructions for spray syringe assembly and use. Each syringe kit contains one vial of THROMBIN-JMI, one vial of diluent and one spray tip and syringe.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix spray tip by pushing down and turning clockwise until the spray tip locks in place.
- 8.
- To spray, depress the syringe plunger in a normal fashion to dispense the THROMBIN-JMI thrombin solution through the tip in a fine spray.
- 9.
- Discard unused contents and syringe.
For use of THROMBIN-JMI 5,000 IU Syringe Spray Kit with GEL-FLOW™ NT:
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw all of the 5 mL of the saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- DISCARD the SPRAY TIP.
- 8.
- Use THROMBIN-JMI thrombin solution syringe according to the directions for use in the GEL-FLOW™ NT package insert.
THROMBIN-JMI EPISTAXIS KIT
Refer to THROMBIN-JMI Epistaxis Kit instructions for accessory assembly and use.
Each epistaxis kit contains one vial of THROMBIN-JMI, one vial of diluent and one nasal drug delivery device.
- 1.
- Remove the outer lid by pulling up at the indicated edge. The inner tray is sterile and suitable for introduction into any operating field.
- 2.
- Remove the cover on the inner tray to expose sterile contents.
- 3.
- Using the sterile syringe equipped with a transfer device, draw the desired amount of saline diluent from the vial into the syringe.
- 4.
- Inject the saline diluent into the THROMBIN-JMI thrombin vial from the syringe to reconstitute the THROMBIN-JMI thrombin powder.
- 5.
- When the THROMBIN-JMI powder is completely dissolved, draw the THROMBIN-JMI thrombin solution into the syringe.
- 6.
- Remove the syringe from the transfer device by turning syringe counterclockwise.
- 7.
- Affix the nasal drug delivery device on to the syringe by pushing the device down onto the THROMBIN-JMI thrombin solution filled syringe and turn clockwise until the nasal drug delivery device locks in place.
- 8.
- Insert the nasal drug delivery device into the naris and spray the THROMBIN-JMI thrombin solution onto the nasal mucosa through the nasal drug delivery device by depressing the syringe plunger using mild or moderate pressure on the syringe plunger.
- 9.
- After administration of THROMBIN-JMI, the device may be removed immediately or briefly held in the nasal passage.
- 10.
- Discard the unused contents, nasal drug delivery device, and the syringe.
2.2 Administration
Topical application of THROMBIN-JMI
- 1.
- The recipient surface should be sponged (not wiped) free of blood before THROMBIN-JMI is applied.
- 2.
- A spray may be used or the surface may be flooded using a sterile syringe and small gauge needle. The most effective hemostasis results occur when the THROMBIN-JMI mixes freely with the blood as soon as it reaches the surface.
- 3.
- Sponging of the treated surfaces should be avoided to assure that the clot remains securely in place.
Use in conjunction with Absorbable Gelatin Sponge
Consult the Absorbable Gelatin Sponge, USP labeling for complete information for use prior to utilizing the following thrombin saturated sponge procedure.
- 1.
- Prepare THROMBIN-JMI solution to desired strength.
- 2.
- Immerse sponge strips of the desired size in THROMBIN-JMI solution. Knead the sponge strips vigorously with moistened, gloved fingers to remove trapped air, thereby facilitating saturation of the sponge.
- 3.
- Apply saturated sponge to bleeding area. Hold in place with a pledget of cotton or a small gauze sponge until hemostasis occurs.
3. Dosage Forms and Strengths
THROMBIN-JMI (Thrombin, Topical (Bovine) U.S.P.), Solution for topical use is supplied in the following packages:
Vial: 5,000 IU vial with 5 mL diluent.
Vial: 20,000 IU vial with 20 mL diluent.
THROMBIN-JMI Pump Spray Kit is supplied in the following packages:
Pump Spray Kit: 20,000 IU vial with 20 mL diluent, spray pump and actuator.
THROMBIN-JMI Syringe Spray Kit is supplied in the following packages:
Syringe Spray Kit: 5,000 IU vial with 5 mL diluent, spray tip and syringe.
Syringe Spray Kit: 20,000 IU vial with 20 mL diluent, spray tip and syringe.
THROMBIN-JMI Epistaxis Kit is supplied in the following packages:
5,000 IU vial with 5 mL diluent, nasal drug delivery device and syringe.
THROMBIN-JMI® is supplied in the Gelfoam-JMI™ Kits:
Gelfoam-JMI™ Sponge Kit (Gelfoam® Absorbable Gelatin Sponge, USP and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU vial with 5 mL diluent).
Gelfoam-JMI™ Powder Kit (Gelfoam® Absorbable Gelatin Powder and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU with 5 mL diluent).
THROMBIN-JMI® 5,000 IU Syringe Spray Kit is supplied in the GEL- FLOW™ Kit:
GEL-FLOW™ Kit (GEL-FLOW™ NT Absorbable Gelatin Powder and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU Syringe Spray Kit).
4. Contraindications
- •
- Do not inject directly into the circulatory system. Because of its action in the clotting mechanism, THROMBIN-JMI can cause extensive intravascular clotting or death.
- •
- Do not re-expose patients to THROMBIN-JMI if there are known or suspected antibodies to bovine thrombin and/or factor V.
- •
- Do not administer to patients with a history of hypersensitivity to THROMBIN-JMI, its components and/or to material of bovine origin.
- •
- Do not use for treatment of severe or brisk arterial bleeding.
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
- •
- Allergic reactions, including anaphylactic/anaphylactoid reactions, have been reported following administration of THROMBIN-JMI.
- •
- Institute intensive supportive measures and treat individual symptoms. Secure the airway and establish adequate respiratory exchange.
5.2 Thrombosis
THROMBIN-JMI causes thrombosis if it enters the circulatory system due to its action in the clotting system.
Apply topically. DO NOT INJECT.
5.3 Immunogenicity
Inhibitory antibodies may develop in patients and interfere with hemostasis. Do not re-expose patients to THROMBIN-JMI if there are known or suspected antibodies to bovine thrombin and/or factor V, due to the potential for these antibodies to interfere with hemostasis. Monitor patients for abnormal coagulation laboratory values, bleeding, or thrombosis.
Bleeding
Bleeding may result from the development of antibodies against factor V. These antibodies may cross-react with human factor V and lead to human factor V deficiency.
Thrombosis
Thrombosis may result from the development of antibodies against bovine thrombin.
6. Adverse Reactions/Side Effects
The most common adverse reactions (incidence greater than or equal to 2%) following administration of THROMBIN-JMI were: hypersensitivity, bleeding, anemia, post-operative wound infection, thromboembolic events, hypotension, pyrexia, tachycardia and thrombocytopenia.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety assessment of THROMBIN-JMI is based primarily on the review of post marketing publications summarizing four (4) randomized controlled clinical trials in which THROMBIN-JMI was used as a comparator2–5, and one (1) observational study6 [see Clinical Studies (14)].
Study 1
In a randomized, double-blinded, controlled trial that compared recombinant human thrombin to THROMBIN-JMI, 206 patients received THROMBIN-JMI and 205 patients received recombinant human thrombin as adjuncts to hemostasis in liver resection, spine, peripheral arterial bypass, and dialysis access surgeries.2 Four hundred one (401) patients completed the trial. The reported adverse reactions in both treatment groups were: cardiac events (18%), hypersensitivity (17%), other infections (15%), bleeding (11%), postoperative wound infection (10%), and thromboembolic events (5%). Among 200 patients who were evaluated for the presence of antibodies to THROMBIN-JMI, 10 patients (5%) were positive at baseline and 43 (21.5%) after treatment. The seroconversion rate in THROMBIN-JMI group was 18.4%.
Study 2
In a multicenter, prospective, randomized, double-blinded, controlled trial that compared plasma-derived human thrombin to THROMBIN-JMI, 152 patients received THROMBIN-JMI and 153 patients received human thrombin applied topically to the target bleeding site with a gelatin sponge.3 Serious adverse reactions (pyrexia and post-procedural hematoma) were reported in two patients receiving THROMBIN-JMI. In this study, 16 out of 126 (12.7%) patients who received THROMBIN-JMI demonstrated seroconversion for at least one of the four antibodies assayed. The four separate ELISA assays used to detect development of antibodies and the corresponding antibody development rates included: 1) Anti-bovine thrombin 10/126 (7.94%), 2) Anti-bovine factor V/Va 12/126 (9.52%), 3) Anti-human thrombin 3/126 (2.38%) and 4) Anti-human factor V/Va 0/126 (0%).
Study 3
The effect of repeat exposure was evaluated in a multi-center, prospective, randomized, double-blinded, controlled trial on 72 patients with diabetic foot ulcers, using a gel prepared with THROMBIN-JMI and autologous platelet rich plasma that was applied weekly for 12 weeks.4 Forty (40) patients were treated with the gel at fourteen (14) sites. Safety parameters were evaluated during the 12 weeks of treatment and the three-month follow-up period. No serious adverse reactions related to the gel treatment were reported.
Study 4
In a prospective, randomized, phase 2, non-inferiority study, topical human thrombin was compared with THROMBIN-JMI during vascular, hepatic, soft tissue, and spinal open surgery procedures.5 A total of 205 patients were randomized in a 2:1 ratio to receive human thrombin (n=137) or THROMBIN-JMI (n=68). The most common treatment-emergent adverse reactions (experienced by >5% patients within a treatment group) were procedural pain, nausea, constipation, pruritus, muscle spasms, insomnia, pyrexia, and vomiting. Two patients in the THROMBIN-JMI group (3.2% of treated patients) showed low-level titers of antibodies to bovine factor V.
6.2 Postmarketing Experience
The following serious adverse reactions have been identified during post approval use of THROMBIN-JMI: anaphylactic reactions, prolonged prothrombin time, prolonged activated partial thromboplastin time, disseminated intravascular coagulation, factor V deficiency, post-procedural hematoma, swelling and Staphylococcal wound infection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Animal reproduction studies have not been conducted with THROMBIN-JMI. It is also not known whether THROMBIN-JMI can cause fetal harm when administered to a pregnant woman. THROMBIN-JMI should be given to a pregnant woman only if clearly needed.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
It is not known whether this drug is excreted in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for THROMBIN-JMI and any potential adverse effects on the breastfed infant from THROMBIN-JMI or from the underlying maternal condition.
8.5 Geriatric Use
Clinical studies of THROMBIN-JMI did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
11. Thrombin-JMI Description
THROMBIN-JMI, Thrombin, Topical (Bovine), is a protein substance produced through a conversion reaction in which prothrombin of bovine origin is activated by tissue thromboplastin of bovine origin in the presence of calcium chloride. It is supplied as a sterile powder that has been freeze-dried in the final container. Also contained in the preparation are mannitol and sodium chloride. Mannitol is included to make the dried product friable and more readily soluble. The product contains no preservative.
THROMBIN-JMI undergoes multistep chromatographic purification and ultrafiltration. The manufacturing process for THROMBIN-JMI has been further improved by the addition of viral filtration and impurity reduction processes. Analytical studies demonstrate the capability of the current manufacturing process to remove significant amounts of extraneous proteins, and result in a reduction of factor Va light chain content to levels below the limit of detection of semi-quantitative Western Blot assay (<92 ng/mL, when reconstituted as directed). The clinical relevance of these findings is unknown.
12. Thrombin-JMI - Clinical Pharmacology
12.1 Mechanism of Action
THROMBIN-JMI requires no intermediate physiological agent for its action. It activates platelets and catalyzes the conversion of fibrinogen to fibrin, which are essential steps for clot formation. Failure to clot blood occurs in the case where the primary clotting defect is the absence of fibrinogen itself. The speed with which thrombin clots blood is dependent upon the concentration of both thrombin and fibrinogen.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been undertaken examining carcinogenicity, genetic toxicity or fertility.
13.2 Animal Toxicology and/or Pharmacology
Thrombin Concentration Study
In a blinded pre-clinical study conducted on a liver lesion model in swine, an inverse dose related response was observed between the visual bleeding scores and the thrombin1 concentration within the absorbable gelatin powder delivery system (GEL-FLOW™ NT) syringe, a device containing 550 mg of absorbable gelatin powder (GELFOAM® Powder).1 Bleeding was assigned scores according to a visual scale, with scores of 0 (no bleeding), 0.5 (ooze), 1 (very slight), 2 (slight), 3 (moderate), and 4 (severe). Scores of 1 and less were considered clinically acceptable. The 770 IU/mL thrombin concentration provided statistically significant lower bleeding scores than either 375 IU/mL or 250 IU/mL thrombin concentrations2 (Table 1). The results of this study showed that higher concentrations of thrombin within the GEL-FLOW™ NT syringe resulted in improved hemostasis as measured by lower bleeding scores.
| Parameter | 250 IU/mL
Mean (Standard Error) | 375 IU/mL
Mean (Standard Error) | 770 IU/mL
Mean (Standard Error) |
|---|---|---|---|
|
3 Minute Bleeding Score |
1.8 (0.2) |
1.6 (0.2) |
0.7 (0.2)* |
|
6 Minute Bleeding Score |
1.7 (0.2) |
1.5 (0.2) |
0.6 (0.2)* |
|
9 Minute Bleeding Score |
1.5 (0.2) |
1.2 (0.2) |
0.5 (0.2)* |
|
12 Minute Bleeding Score |
1.4 (0.2) |
1.0 (0.2)† |
0.4 (0.2)* |
14. Clinical Studies
Observational Study
A total of 554 subjects were enrolled, in a multicenter, open-label, observational study (MOSAIC) conducted to assess the effect of possible exposure to THROMBIN-JMI on activated partial thromboplastin time (aPTT) at 48 hours post-surgery in subjects with likelihood of prior exposure to THROMBIN-JMI within the past 4 years.6 Of the 554 subjects, 550 had undergone surgery and completed the study. A total of 384 subjects undergoing vascular surgeries, neurosurgeries and orthopedic surgeries were exposed to THROMBIN-JMI (5,000 to 20,000 IU).
In this study, the impact of exposure to THROMBIN-JMI in 78 subjects who were positive for anti-bovine thrombin (aBT) antibodies prior to surgery was compared with 140 subjects who did not have any aBT antibodies and were not exposed to THROMBIN-JMI. The study did not meet the pre-specified primary endpoint, a mean change from baseline in aPTT at 48 hours post-surgery. The study was not powered to detect coagulopathy related to an immune response after bovine thrombin use.
A post hoc analysis was performed in which subjects who underwent surgery were re-assigned to one of four exploratory cohorts based on the presence or absence of pre-surgery anti-bovine factor V/anti-bovine factor V active (aBV/Va) antibodies and whether or not they were administered THROMBIN-JMI during the study surgery. Non-inferiority (based on aPTT) was observed in these exploratory cohorts at all-time points of 48 hours, 4 weeks, and 8 weeks post-surgery.
For the primary study cohort (THROMBIN-JMI use in subjects with baseline positive aBT or positive aBV/Va), there was a higher incidence of seroconversion from anti human thrombin (aHT) negative at baseline to post-surgery positive compared to the primary reference cohort (no THROMBIN-JMI use in subjects with baseline negative aBT or negative aBV/Va). This difference was not present at 48 hours after surgery but was evident at 4 weeks and 8 weeks post-surgery. A similar immunological response with aBT and aBV/Va antibodies was observed following THROMBIN-JMI administration.
Secondary immune responses in patients treated with THROMBIN-JMI were evidenced by the generation of anti-bovine and anti-human thrombin and factor V/Va antibodies, consistent with known immunogenicity of topical bovine thrombin.
15. References
- 1.
- Morse DC, Silva E, Bartrom J, et al: Improved bleeding scores using Gelfoam Powder with incremental concentrations of bovine thrombin in a swine liver lesion model. J Throm Thrombolysis. 2016;42(3):352–359.
- 2.
- Chapman WC, Singla N, Genyk Y, et al: A Phase 3, Randomized, Double-Blind Comparative Study of the Efficacy and Safety of Topical Recombinant Human Thrombin and Bovine Thrombin in Surgical Hemostasis. J Am Coll Surg. 2007;205:256–265.
- 3.
- Doria C, Fischer CP, Wood CG, et al: Phase 3, randomized, double-blind study of plasma-derived human thrombin versus bovine thrombin in achieving hemostasis in patients undergoing surgery. Curr Med Res Opin. 2008;24(3):785–794.
- 4.
- Driver VR, Hanft J, Fylling CP, et al: A Prospective, Randomized, Controlled Trial of Autologous Platelet-Rich Plasma Gel for the Treatment of Diabetic Foot Ulcers. Ostomy/Wound Management. 2006;52(6):68–87.
- 5.
- Minkowitz H, Navarro-Puerto J, Lakshman S, et al: Prospective, Randomized, Phase II, Non-Inferiority Study to Evaluate the Safety and Efficacy of Topical Thrombin (Human) Grifols as Adjunct to Hemostasis During Vascular, Hepatic, Soft Tissue, and Spinal Open Surgery. J Am Coll Surg. 2019;229(5):497–507.
- 6.
- Paterson CA, Pixton GC, Proskin HM, et al: Immune responses associated with perioperative exposure and reexposure to topical bovine thrombin do not impair hemostasis. Clin Appl Thromb Hemost. 2011;17(6):620–632.
16. How is Thrombin-JMI supplied
Not made with natural rubber latex.
THROMBIN-JMI® is supplied in the following packages:
- NDC 60793-215-05
- Vial: 5,000 IU vial with 5 mL diluent.
- NDC 60793-217-20
- Vial: 20,000 IU vial with 20 mL diluent.
THROMBIN-JMI® Pump Spray Kit is supplied in the following package:
- NDC 60793-217-21
- Pump Spray Kit: 20,000 IU vial with 20 mL diluent, spray pump and actuator.
THROMBIN-JMI® Syringe Spray Kit is supplied in the following packages:
- NDC 60793-705-05
- Syringe Spray Kit: 5,000 IU vial with 5 mL diluent, spray tip and syringe.
NDC 60793-217-22 - Syringe Spray Kit: 20,000 IU vial with 20 mL diluent, spray tip and syringe.
THROMBIN-JMI® Epistaxis Kit is supplied in the following package:
- NDC 60793-205-05
- Epistaxis Kit: 5,000 IU vial with 5 mL diluent, nasal drug delivery device and syringe.
THROMBIN-JMI® is supplied in the Gelfoam-JMI™ Kits:
- GTIN 00360793310107 (60793-310-10)
- Gelfoam-JMI™ Sponge Kit (Gelfoam® Absorbable Gelatin Sponge, USP and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU vial with 5 mL diluent).
- GTIN 00360793410104 (60793-410-10)
- Gelfoam-JMI™ Powder Kit (Gelfoam® Absorbable Gelatin Powder and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU with 5 mL diluent).
THROMBIN-JMI® 5,000 IU Syringe Spray Kit is supplied in the GEL- FLOW™ Kit:
- GTIN 00300092250018
- GEL-FLOW™ Kit (GEL-FLOW™ NT Absorbable Gelatin Powder and Thrombin, Topical (Bovine) U.S.P., THROMBIN-JMI®, 5,000 IU Syringe Spray Kit).
17. Patient Counseling Information
Because THROMBIN-JMI may cause the formation of clots in blood vessels if it enters the bloodstream, advise patients to consult their physician if they experience leg tenderness or swelling, chest pain, shortness of breath, or difficulty speaking or swallowing.
Manufactured by: GenTrac, Incorporated, Middleton, Wisconsin 53562
Distributed by Pfizer Inc, New York, New York 10001
US License No. 977
PRINCIPAL DISPLAY PANEL - 5,000 IU Vial Label
NDC 60793-315-01
THROMBIN,
TOPICAL
(BOVINE), U.S.P.
THROMBIN-JMI®
5,000 International Units
Pfizer
Rx Only
PRINCIPAL DISPLAY PANEL - 5 mL Vial Label
NDC 60793-218-09
SODIUM
CHLORIDE
U.S.P. 0.9% for
THROMBIN, TOPICAL
(BOVINE), U.S.P.
THROMBIN-JMI®
5 mL
Pfizer
Rx Only
PRINCIPAL DISPLAY PANEL - 5000 IU Vial Kit Carton
FOR TOPICAL USE ONLY – DO NOT INJECT
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
5,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
CONTENTS:
ONE VIAL 5,000
UNITS THROMBIN
ONE VIAL 5 mL
DILUENT
ONE TRANSFER
DEVICE
Pfizer
PRINCIPAL DISPLAY PANEL - 20,000 IU Vial Label
NDC 60793-317-01
THROMBIN,
TOPICAL
(BOVINE), U.S.P.
THROMBIN-JMI®
20,000
International Units
Pfizer
Rx Only
PRINCIPAL DISPLAY PANEL - 20 mL Vial Label
NDC 60793-221-09
SODIUM
CHLORIDE
U.S.P. 0.9% for
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
20 mL
Pfizer
Rx Only
PRINCIPAL DISPLAY PANEL- 20,000 IU Vial Kit Carton
FOR TOPICAL USE ONLY – DO NOT INJECT
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
20,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
CONTENTS:
ONE VIAL 20,000
UNITS THROMBIN
ONE VIAL 20 mL
DILUENT
ONE TRANSFER
DEVICE
Pfizer
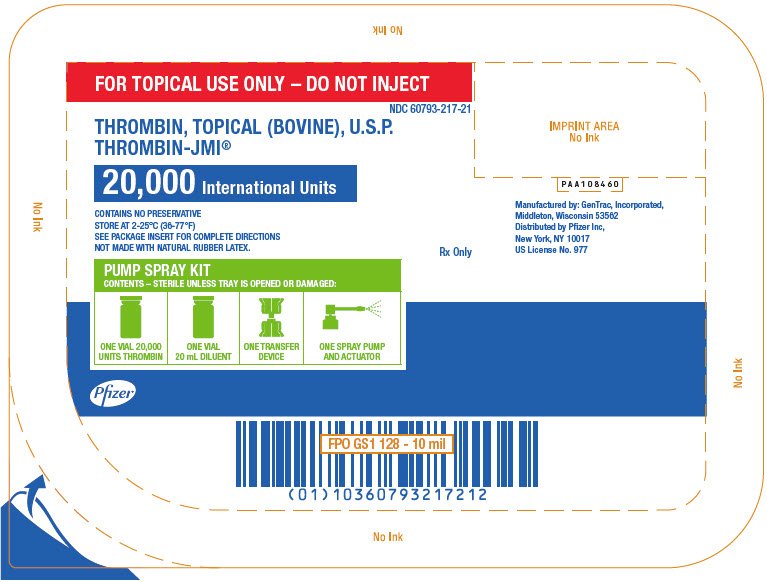
PRINCIPAL DISPLAY PANEL - 20,000 IU Pump Spray Kit Tray
FOR TOPICAL USE ONLY – DO NOT INJECT
NDC 60793-217-21
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
20,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
PUMP SPRAY KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 20,000
UNITS THROMBIN
ONE VIAL
20 mL DILUENT
ONE TRANSFER
DEVICE
ONE SPRAY PUMP
AND ACTUATOR
Pfizer
PAA108460
Manufactured by: GenTrac, Incorporated,
Middleton, Wisconsin 53562
Distributed by Pfizer Inc,
New York, NY 10017
US License No. 977
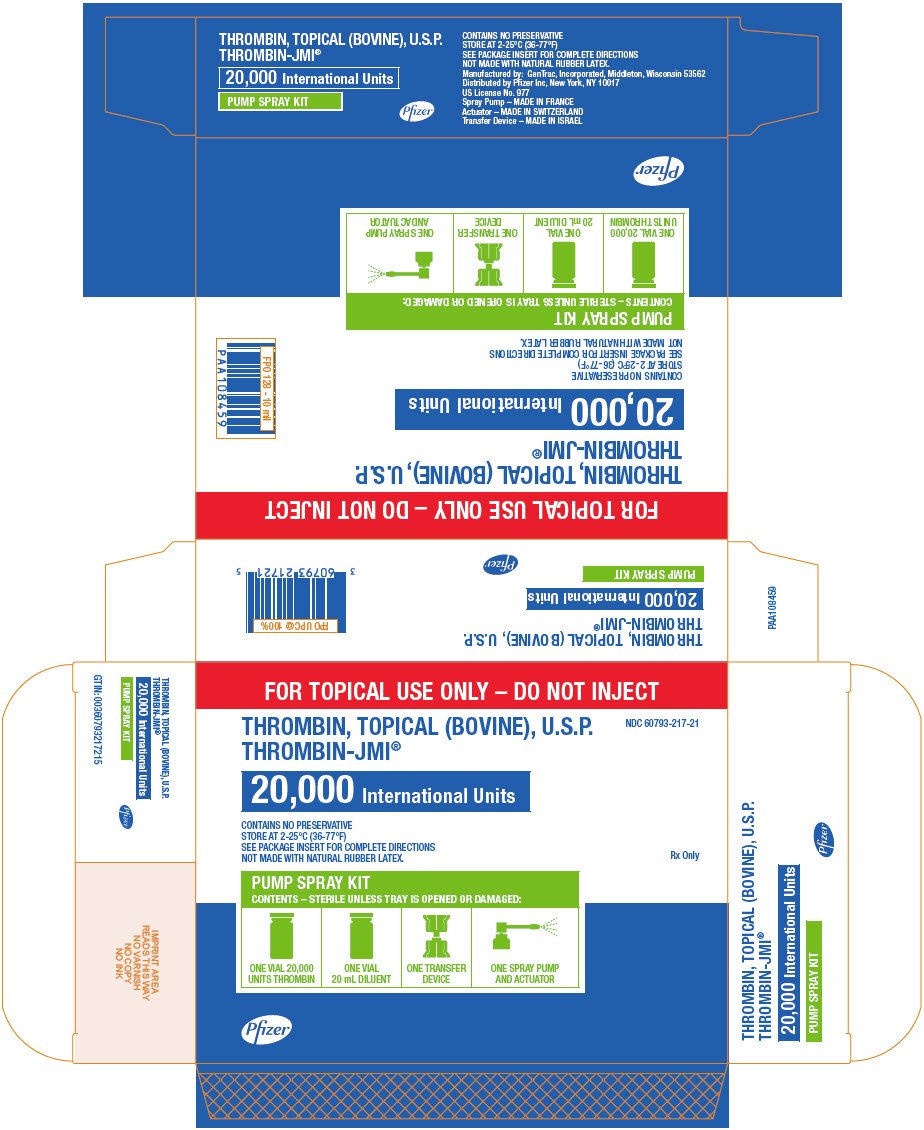
PRINCIPAL DISPLAY PANEL - 20,000 IU Pump Spray Kit Carton
FOR TOPICAL USE ONLY – DO NOT INJECT
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
NDC 60793-217-21
20,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
PUMP SPRAY KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 20,000
UNITS THROMBIN
ONE VIAL
20 mL DILUENT
ONE TRANSFER
DEVICE
ONE SPRAY PUMP
AND ACTUATOR
Pfizer
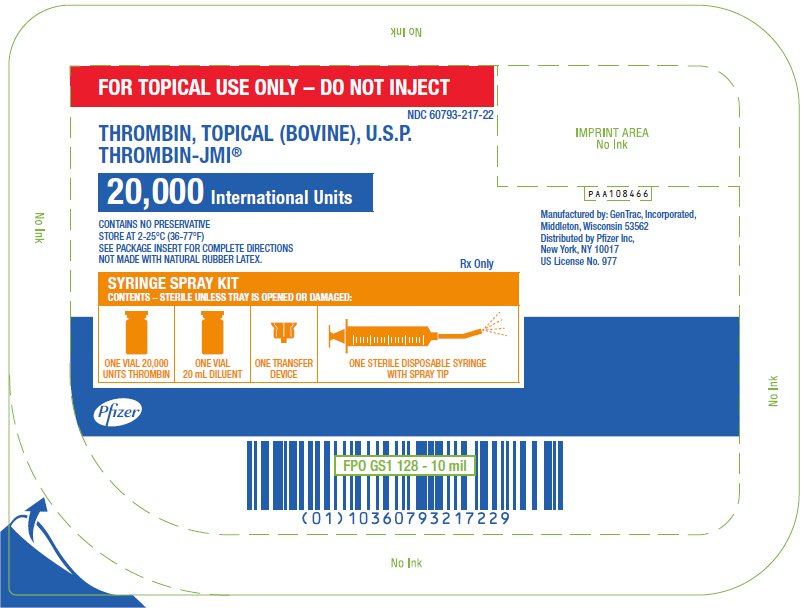
PRINCIPAL DISPLAY PANEL - 20,000 IU Syringe Spray Kit Tray
FOR TOPICAL USE ONLY – DO NOT INJECT
NDC 60793-217-22
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
20,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
SYRINGE SPRAY KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 20,000
UNITS THROMBIN
ONE VIAL
20 mL DILUENT
ONE TRANSFER
DEVICE
ONE STERILE DISPOSABLE SYRINGE
WITH SPRAY TIP
Pfizer
PAA108466
Manufactured by: GenTrac, Incorporated,
Middleton, Wisconsin 53562
Distributed by Pfizer Inc,
New York, NY 10017
US License No. 977
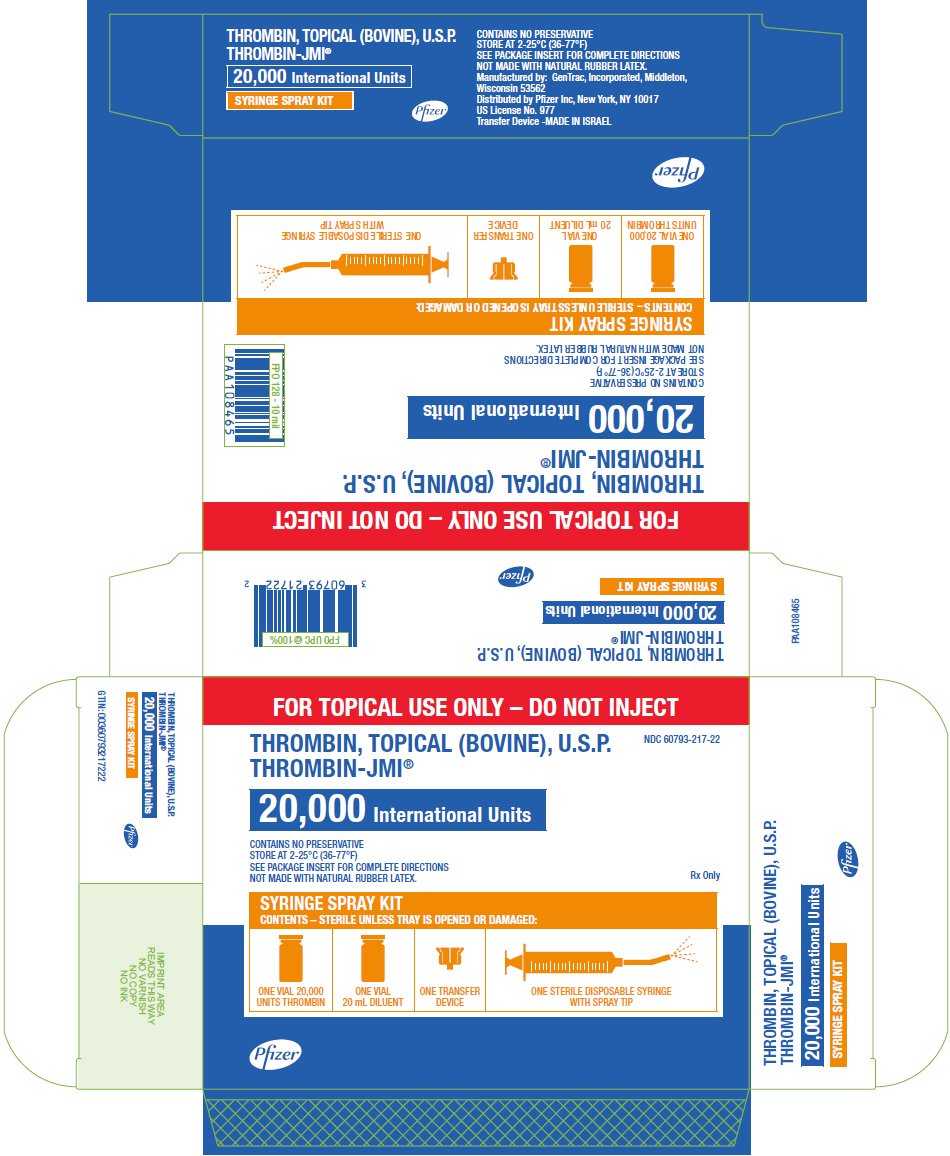
PRINCIPAL DISPLAY PANEL - 20,000 IU Syringe Spray Kit Carton
FOR TOPICAL USE ONLY – DO NOT INJECT
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
NDC 60793-217-22
20,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
SYRINGE SPRAY KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 20,000
UNITS THROMBIN
ONE VIAL
20 mL DILUENT
ONE TRANSFER
DEVICE
ONE STERILE DISPOSABLE SYRINGE
WITH SPRAY TIP
Pfizer
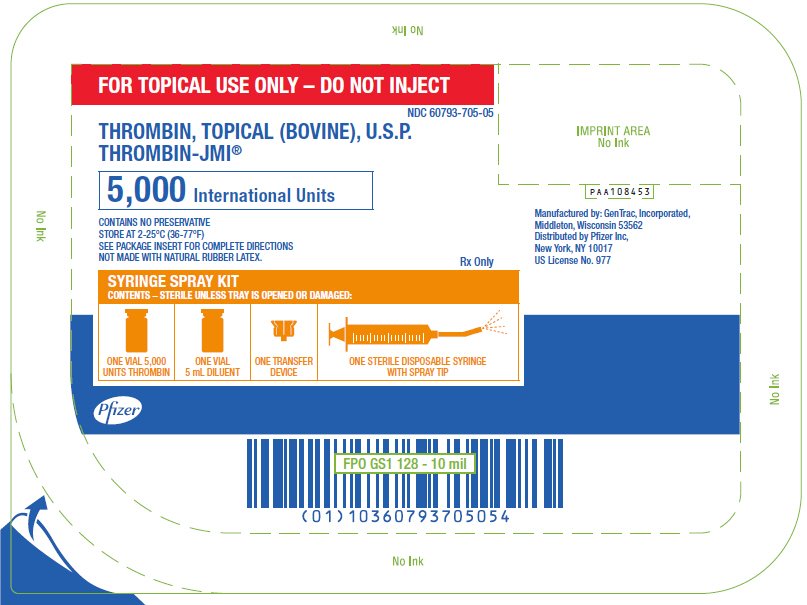
PRINCIPAL DISPLAY PANEL - 5,000 IU Syringe Spray Kit Tray
FOR TOPICAL USE ONLY – DO NOT INJECT
NDC 60793-705-05
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
5,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
SYRINGE SPRAY KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 5,000
UNITS THROMBIN
ONE VIAL
5 mL DILUENT
ONE TRANSFER
DEVICE
ONE STERILE DISPOSABLE SYRINGE
WITH SPRAY TIP
Pfizer
PAA108453
Manufactured by: GenTrac, Incorporated,
Middleton, Wisconsin 53562
Distributed by Pfizer Inc,
New York, NY 10017
US License No. 977
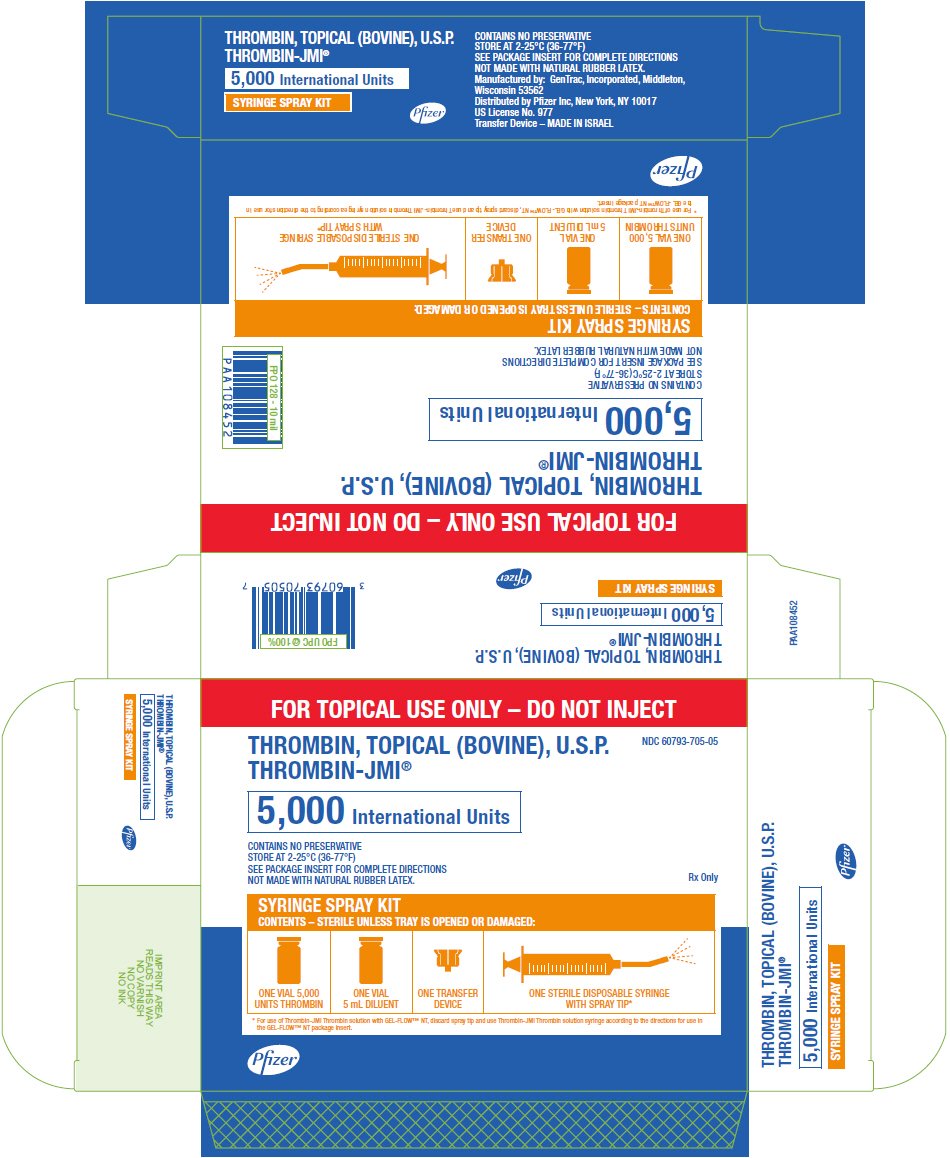
PRINCIPAL DISPLAY PANEL - 5,000 IU Syringe Spray Kit Carton
FOR TOPICAL USE ONLY – DO NOT INJECT
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
NDC 60793-705-05
5,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
SYRINGE SPRAY KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 5,000
UNITS THROMBIN
ONE VIAL
5 mL DILUENT
ONE TRANSFER
DEVICE
ONE STERILE DISPOSABLE SYRINGE
WITH SPRAY TIP*
* For use of Thrombin-JMI Thrombin solution with GEL-FLOW™ NT, discard spray tip and use Thrombin-JMI Thrombin solution syringe according to the directions for use in
the GEL-FLOW™ NT package insert.
Pfizer
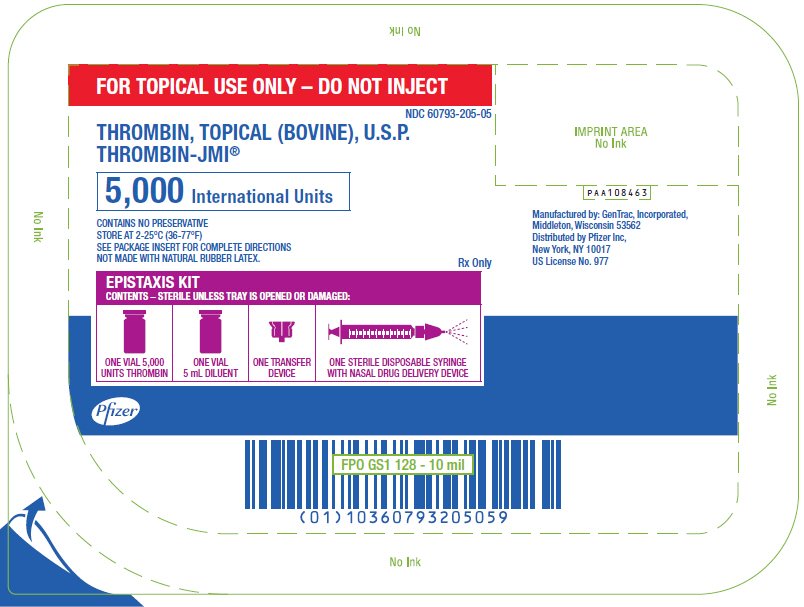
PRINCIPAL DISPLAY PANEL - 5,000 IU Epistaxis Kit Tray
FOR TOPICAL USE ONLY – DO NOT INJECT
NDC 60793-205-05
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
5,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
EPISTAXIS KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 5,000
UNITS THROMBIN
ONE VIAL
5 mL DILUENT
ONE TRANSFER
DEVICE
ONE STERILE DISPOSABLE SYRINGE
WITH NASAL DRUG DELIVERY DEVICE
Pfizer
PAA108463
Manufactured by: GenTrac, Incorporated,
Middleton, Wisconsin 53562
Distributed by Pfizer Inc,
New York, NY 10017
US License No. 977
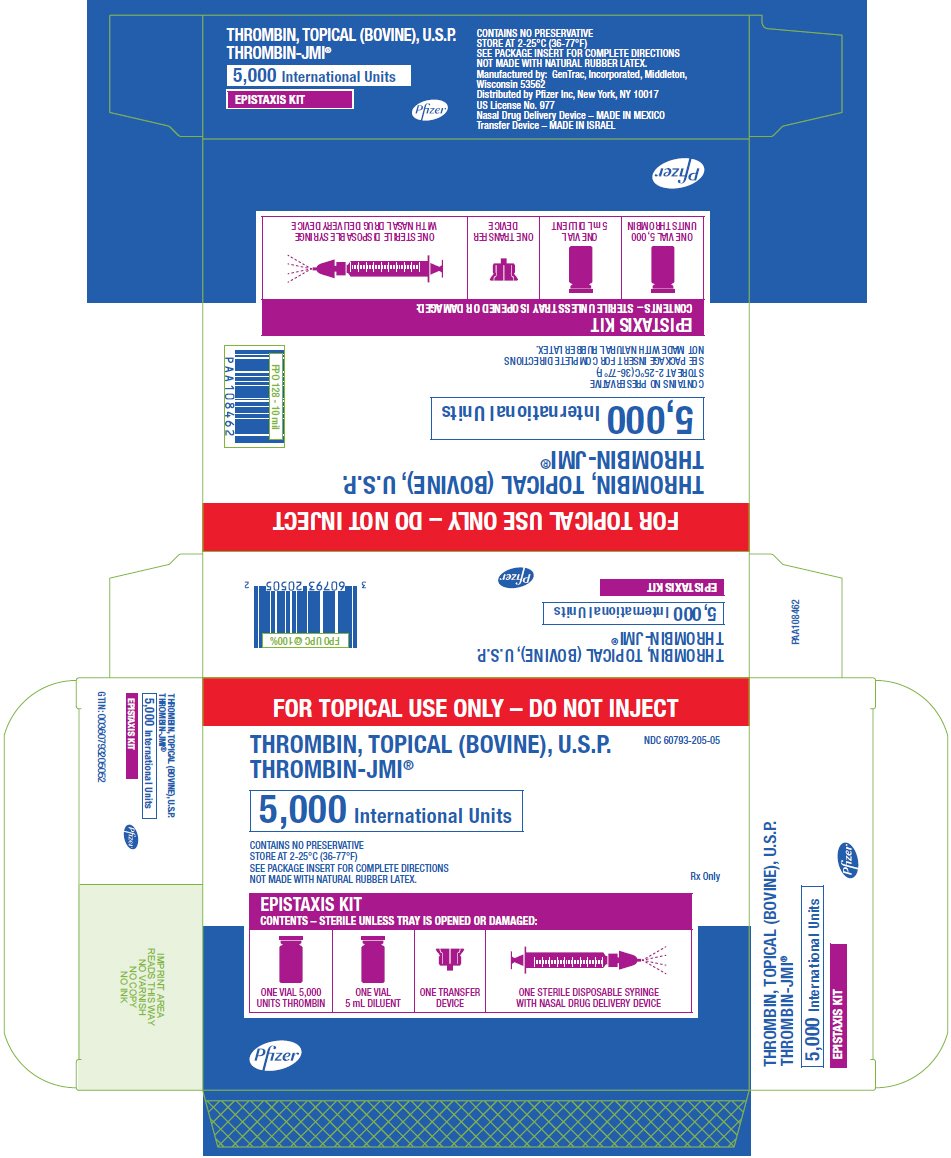
PRINCIPAL DISPLAY PANEL - 5,000 IU Epistaxis Kit Carton
FOR TOPICAL USE ONLY – DO NOT INJECT
THROMBIN, TOPICAL (BOVINE), U.S.P.
THROMBIN-JMI®
NDC 60793-205-05
5,000 International Units
CONTAINS NO PRESERVATIVE
STORE AT 2-25°C (36-77°F)
SEE PACKAGE INSERT FOR COMPLETE DIRECTIONS
NOT MADE WITH NATURAL RUBBER LATEX.
Rx Only
EPISTAXIS KIT
CONTENTS – STERILE UNLESS TRAY IS OPENED OR DAMAGED:
ONE VIAL 5,000
UNITS THROMBIN
ONE VIAL
5 mL DILUENT
ONE TRANSFER
DEVICE
ONE STERILE DISPOSABLE SYRINGE
WITH NASAL DRUG DELIVERY DEVICE
Pfizer
| THROMBIN-JMI
thrombin, topical (bovine) kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| THROMBIN-JMI
thrombin, topical (bovine) kit |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| THROMBIN-JMI
thrombin, topical (bovine) kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| THROMBIN-JMI
thrombin, topical (bovine) kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Pfizer Laboratories Div Pfizer Inc (134489525) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Pharmacia & Upjohn Company LLC | 618054084 | ANALYSIS(60793-205, 60793-215, 60793-217, 60793-705) , MANUFACTURE(60793-205, 60793-215, 60793-217, 60793-705) , PACK(60793-205, 60793-215, 60793-217, 60793-705) , LABEL(60793-205, 60793-215, 60793-217, 60793-705) | |
More about Thrombin-JMI (thrombin topical)
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- Drug class: miscellaneous coagulation modifiers