OsmoPrep Prescribing Information
Package insert / product label
Generic name: sodium phosphate, monobasic, monohydrate, sodium phosphate, dibasic anhydrous
Dosage form: tablet
Drug class: Laxatives
Medically reviewed by Drugs.com. Last updated on Apr 15, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
- Medication Guide
Highlights of Prescribing Information
OSMOPREP (sodium phosphate monobasic monohydrate and sodium phosphate dibasic anhydrous) tablets, for oral use
Initial U.S. Approval: 2006
WARNING: ACUTE PHOSPHATE NEPHROPATHY
See full prescribing information for complete boxed warning.
- •
- Rare, serious reports of acute phosphate nephropathy in patients who received oral sodium phosphate products, including OsmoPrep, for colon cleansing prior to colonoscopy. Some cases have resulted in permanent impairment of renal function and some patients required long-term dialysis. (5.1)
- •
- Patients at increased risk include those with increased age, hypovolemia, increased bowel transit time (such as bowel obstruction), active colitis, or baseline kidney disease, and those using medicines that affect renal perfusion or function (such as diuretics, angiotensin-converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs], and possibly nonsteroidal anti-inflammatory drugs [NSAIDs]). (5.1)
- •
- Advise patients of the importance of following the recommended split dosage regimen and the importance of adequate hydration before, during and after the use of OsmoPrep. Avoid additional sodium phosphate-based products. (2.1, 2.2)
Recent Major Changes
- Warnings and Precautions (5.7) 11/2018
Indications and Usage for OsmoPrep
OsmoPrep is an osmotic laxative indicated for cleansing of the colon as a preparation for colonoscopy in adults. (1)
OsmoPrep Dosage and Administration
Important Administration Instructions:
- •
- Two doses of OsmoPrep are required for a complete preparation for colonoscopy: the first dose the evening before the colonoscopy and the second dose on the morning of the colonoscopy. (2.1)
- •
- Do not take other laxatives, particularly additional sodium phosphate-based purgative or enema products. (2.1,7.3)
- •
- Do not take oral medications within 1 hour before or after starting each dose. (2.1, 7.2)
Dosage Regimen (2.2)
The recommended adult dosage is 32 tablets taken orally with a total of 2 quarts of clear liquids in the following manner:
- •
- Evening before colonoscopy: 4 tablets with 8 ounces of clear liquids every 15 minutes for a total of 20 tablets.
- •
- Next morning: 4 tablets with 8 ounces of clear liquids every 15 minutes for a total of 12 tablets.
Dosage Forms and Strengths
Tablets: 1.5 g of sodium phosphate (3)
Contraindications
Warnings and Precautions
- •
- Renal disease and electrolyte disorders: Use caution in severe renal impairment and those taking concurrent medications that increase risk, ensure adequate hydration, and consider laboratory assessments prior to and after use. (5.1, 7.1)
- •
- Cardiac arrhythmias: Consider pre-dose and post-colonoscopy ECGs in patients at increased risk. (5.2)
- •
- Seizures: Use caution in patients with a history of seizures and patients at increased risk of seizure, including medications that lower the seizure threshold. (5.3, 7.1)
- •
- Suspected GI obstruction or perforation: Rule out diagnosis before administration. (4, 5.4)
- •
- Colonic mucosal ulceration: Consider potential for mucosal ulcerations when interpreting colonoscopy findings in patients with known or suspected inflammatory bowel disease. Use with caution in patients with an acute exacerbation of chronic inflammatory bowel disease (5.5)
- •
- Patients at risk for aspiration: Observe during administration. (5.6)
- •
- Hypersensitivity reactions, including anaphylaxis: Inform patients to seek immediate medical care if symptoms occur. (5.7)
Adverse Reactions/Side Effects
Most common adverse reactions (≥3%) are: bloating, nausea, abdominal pain, and vomiting. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Salix Pharmaceuticals at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- •
- Drugs that increase risks due to fluid and electrolyte changes. (7.1)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 3/2019
Related/similar drugs
MiraLAX, docusate, erythromycin, lactulose, bisacodyl, Colace, senna
Full Prescribing Information
WARNING: ACUTE PHOSPHATE NEPHROPATHY
- •
- There have been rare, but serious reports of acute phosphate nephropathy in patients who received oral sodium phosphate products, including OsmoPrep, for colon cleansing prior to colonoscopy. Some cases have resulted in permanent impairment of renal function and some patients required long-term dialysis [see Warnings and Precautions (5.1)].
- •
- While some cases have occurred in patients without identifiable risk factors, patients at increased risk of acute phosphate nephropathy may include those with increased age, hypovolemia, increased bowel transit time (such as bowel obstruction), active colitis, or baseline kidney disease, and those using medicines that affect renal perfusion or function (such as diuretics, angiotensin-converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs], and possibly nonsteroidal anti-inflammatory drugs [NSAIDs]) [see Warnings and Precautions (5.1)].
- •
- Advise patients of the importance of following the recommended split dosage regimen and the importance of adequate hydration before, during and after the use of OsmoPrep. Avoid additional sodium phosphate-based purgative or enema products [see Dosage and Administration (2.1,2.2)].
1. Indications and Usage for OsmoPrep
OsmoPrep® is indicated for cleansing of the colon as a preparation for colonoscopy in adults.
2. OsmoPrep Dosage and Administration
2.1 Important Administration Instructions
- •
- Correct fluid and electrolyte abnormalities before treatment with OsmoPrep [see Warnings and Precautions (5.1)].
- •
- Clear liquids must be consumed before, during and after taking OsmoPrep [see Dosage and Administration (2.1), Warnings and Precautions (5.1)].
- •
- Do not administer OsmoPrep within 7 days of a previous use.
- •
- Two doses of OsmoPrep are required for a complete preparation for colonoscopy: the first dose the evening before the colonoscopy and the second dose on the morning of the colonoscopy [see Dosage and Administration (2.2)].
- •
- Consume only clear liquids (no solid food) from the start of OsmoPrep treatment until after the colonoscopy.
- •
- Do not eat or drink alcohol, milk, anything colored red or purple or any other foods containing pulp material.
- •
- Do not take other laxatives while taking OsmoPrep, particularly additional sodium phosphate-based purgative or enema products [see Drug Interactions (7.3)].
- •
- Do not take oral medications within 1 hour before or after starting each dose of OsmoPrep [see Drug Interactions (7.2)].
2.2 Dosage Regimen
Instruct adult patients that on the day before their colonoscopy, they can consume a light breakfast consisting of clear soup and/or plain yogurt (no solid foods) before noon, followed by only clear liquids until after the colonoscopy.
The recommended dose of OsmoPrep for colon cleansing for adult patients is 32 tablets (48 grams of sodium phosphate)
taken orally with a total of 2 quarts of clear liquids in the following manner:
The evening before the colonoscopy: Take 4 OsmoPrep tablets with 8 ounces of clear liquids every 15 minutes for a total
of 20 tablets.
On the day of the colonoscopy starting 3 to 5 hours before the procedure: Take 4 OsmoPrep tablets with 8 ounces of clear
liquids every 15 minutes for a total of 12 tablets.
3. Dosage Forms and Strengths
OsmoPrep is supplied as oval, white to off-white compressed tablets, debossed with “SLX” on one side of the bisect and
“102” on the other side of the bisect. Each 1.5 gram tablet contains 1.102 grams of sodium phosphate monobasic
monohydrate, USP and 0.398 grams of sodium phosphate dibasic anhydrous, USP.
4. Contraindications
OsmoPrep is contraindicated in the following conditions:
- •
- History of acute phosphate nephropathy [see Warnings and Precautions (5.1)]
- •
- Gastrointestinal (GI) obstruction [see Warnings and Precautions (5.7)]
- •
- Gastric bypass or stapling surgery
- •
- Bowel perforation
- •
- Toxic colitis
- •
- Toxic megacolon
- •
- Hypersensitivity to sodium phosphate salts or any component of OsmoPrep [see Warnings and Precautions (5.7)].
5. Warnings and Precautions
5.1 Renal Disease, Acute Phosphate Nephropathy, and Electrolyte Disorders
Renal Disease and Acute Phosphate Nephropathy
There have been rare, but serious, reports of renal failure, acute phosphate nephropathy, and nephrocalcinosis in patients
who received oral sodium phosphate products, including OsmoPrep, for colon cleansing prior to colonoscopy. These cases
often resulted in permanent impairment of renal function and several patients required long-term dialysis. The time to
onset is typically within days; however, in some cases, the diagnosis of these events has been delayed up to several
months after the ingestion of these products. Patients at increased risk of acute phosphate nephropathy may include
patients with the following: hypovolemia, baseline kidney disease, increased age, and patients using medicines that affect
renal perfusion or function [such as diuretics, angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor
blockers, and possibly nonsteroidal anti-inflammatory drugs (NSAIDs) [see Drug Interactions (7.1)].
Electrolyte Disorders
Bowel preparations, including OsmoPrep can cause fluid and electrolyte disturbances, which can lead to serious adverse
reactions including cardiac arrhythmias, seizures and renal impairment [see Adverse Reactions (6.2)].
Patient Management
- •
- OsmoPrep is contraindicated in patients with a history of acute phosphate nephropathy [see Contraindications (4)]. Use OsmoPrep with caution in patients with severe renal impairment (creatinine clearance less than 30 mL/minute), with conditions, or who are taking medications, that increase the risk for fluid and electrolyte disturbances or may increase the risk of arrhythmias, seizures, or renal impairment. Consider performing baseline and post-colonoscopy labs (phosphate, calcium, potassium, sodium, creatinine, and BUN) in these patients [see Drug Interactions (7.1)].
- •
- Correct electrolyte abnormalities such as hypernatremia, hyperphosphatemia, hypokalemia, or hypocalcemia
before treatment with OsmoPrep [see Dosage and Administration (2.1)].
- •
- Avoid additional sodium phosphate-based purgative or enema products [see Drug Interactions (7.1)].
- •
- Advise all patients to hydrate adequately before, during, and after the use of OsmoPrep [see Dosage and Administration (2.1,2.2)].
- •
- If a patient develops significant vomiting or signs of dehydration while or after taking OsmoPrep, consider
performing post-colonoscopy lab tests [electrolytes, creatinine, and blood urea nitrogen (BUN)].
5.2 Cardiac Arrhythmias
There have been rare reports of serious arrhythmias associated with the use of ionic osmotic laxative products for bowel
preparation. QT prolongation with sodium phosphate tablets has been associated with electrolyte imbalances, such as
hypokalemia and hypocalcemia.
Use caution when prescribing OsmoPrep for patients at increased risk of arrhythmias (e.g., patients with a history of
prolonged QT, uncontrolled arrhythmias, recent myocardial infarction, unstable angina, congestive heart failure, or
cardiomyopathy) and those taking medications known to prolong the QT interval, since serious complications may occur.
Consider pre-dose and post-colonoscopy ECGs s in patients at increased risk of serious cardiac arrhythmias.
5.3 Seizures
There have been rare reports of generalized tonic-clonic seizures and/or loss of consciousness associated with use of sodium phosphate osmotic laxative products, such as OsmoPrep, in patients with no prior history of seizures. The seizure cases were associated with electrolyte abnormalities (e.g., hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia) and low serum osmolality. The neurologic abnormalities resolved with correction of fluid and electrolyte abnormalities.
Use OsmoPrep with caution in patients with a history of seizures and in patients at higher risk of seizure, such as patients taking medications that lower the seizure threshold (such as tricyclic antidepressants), patients withdrawing from alcohol or benzodiazepines, or patients with known or suspected hyponatremia [see Drug Interactions (7.1)].
5.4 Use in Patients with Significant Gastrointestinal Disease
If gastrointestinal obstruction or perforation is suspected, perform appropriate diagnostic studies to rule out these conditions before administering OsmoPrep [see Contraindications (4)].
Use with caution in patients with severe active ulcerative colitis.
5.5 Colonic Mucosal Ulceration and Inflammatory Bowel Disease
Osmotic laxatives, including OsmoPrep, may induce colonic mucosal aphthous ulcerations. In the OsmoPrep clinical program, aphthous ulcers were observed in 3% of patients who took the recommended OsmoPrep dosing regimen. Consider the potential for mucosal ulcerations resulting from bowel preparation when interpreting colonoscopic finding should in patients with known or suspected inflammatory bowel disease.
Use OsmoPrep with caution in patients experiencing an acute exacerbation of chronic inflammatory bowel disease as published data suggest that sodium phosphate absorption may be enhanced in such patients.
5.6 Aspiration
Patients with impaired gag reflex or other swallowing abnormalities are at risk for regurgitation or aspiration of OsmoPrep. Observed these patients during administration of OsmoPrep.
5.7 Hypersensitivity Reactions
OsmoPrep may cause serious hypersensitivity reactions including anaphylaxis, angioedema, rash, urticaria, and throat tightness [see Adverse Reactions (6.2)]. Inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care should signs and symptoms occur.
6. Adverse Reactions/Side Effects
The following serious or otherwise important adverse reactions for bowel preparations are described elsewhere
in the labeling:
• Renal Disease, Acute Phosphate Nephropathy, and Electrolyte Disorders [see Warnings and Precautions (5.1)]
• Cardiac Arrhythmias [see Warnings and Precautions (5.2)]
• Seizures [see Warnings and Precautions (5.3)]
• Use in Patients with Significant Gastrointestinal Disease [see Warnings and Precautions (5.4)]
• Colonic Mucosal Ulceration and Inflammatory Bowel Disease [see Warnings and Precautions (5.5)]
• Aspiration [see Warnings and Precautions (5.6)]
• Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of OsmoPrep was evaluated in two randomized, investigator-blinded, active controlled trials in 931 adult patients undergoing elective colonoscopy. The mean age of the study population was 60 years (range 20 to 89 years), 88% of patients were Caucasian and 55% were female [see Clinical Studies (14)].
Table 1 shows the most common adverse reactions reported in greater than 3% of patients by treatment group in Study 1 [see Clinical Studies (14)]. Since diarrhea was considered as a part of the efficacy of OsmoPrep, diarrhea was not defined as an adverse event in this clinical trial.
|
OsmoPrep 32 tabs (48 g) N=272 |
OsmoPrep 40 tabs (60 g) N=265 |
Sodium Phosphate† 40 tabs (60 g) N=268 |
|
|
Bloating |
31% |
39% |
41% |
|
Nausea |
26% |
37% |
30% |
|
Abdominal Pain |
23% |
24% |
25% |
|
Vomiting |
4% |
10% |
9% |
Electrolyte Abnormalities in Study 1
Hyperphosphatemia
A total of, 96%, 96%, and 93% of patients who took 60 grams of oral sodium phosphate, 60 grams of OsmoPrep, and 48 grams of OsmoPrep, respectively, developed hyperphosphatemia (defined as phosphate level > 5.1 mg/dL) on the day of the colonoscopy. In this study, patients who took 60 grams of oral sodium phosphate, 60 grams of OsmoPrep, and 48 grams of OsmoPrep had baseline mean phosphate levels of 3.5, 3.5, and 3.6 mg/dL and subsequently developed mean phosphate levels of 7.6, 7.9, and 7.1 mg/dL, respectively, on the day of the colonoscopy.
Hyperkalemia
A total of 20%, 22%, and 18% of patients who took 60 grams of oral sodium phosphate, 60 grams of OsmoPrep, and 48 grams of OsmoPrep, respectively, developed hypokalemia (defined as a potassium level < 3.4 mEq/L) on the day of the colonoscopy. In this study, patients who took 60 grams of oral sodium phosphate, 60 grams of OsmoPrep, and 48 grams of OsmoPrep all had baseline potassium levels of about 4.3 mEq/L and then developed a mean potassium level of 3.7 mEq/L on the day of the colonoscopy.
Several patients on all three sodium phosphate regimens developed hypocalcemia and hypernatremia that did not require treatment.
The OsmoPrep 60-gram dosage regimen was associated with an increased risk of adverse reactions compared to the 48-gram dosage regimen and a similar overall response rate [see Clinical Studies (14)]. Therefore, the OsmoPrep 60-gram dosage is not a recommended regimen [see Dosage and Administration (2.2)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of OsmoPrep. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity reactions: anaphylaxis, angioedema (swelling of the lips, tongue and face), rash, pruritus, urticaria, throat tightness, bronchospasm, dyspnea, pharyngeal edema, dysphagia, and paresthesia.
Cardiovascular: Arrhythmias
Nervous system: Seizures
Renal: Renal impairment, increased blood urea nitrogen (BUN), increased creatinine, acute renal failure, acute phosphate nephropathy, nephrocalcinosis, and renal tubular necrosis.
7. Drug Interactions
7.1 Drugs That May Increase Risks due to Fluid and Electrolyte Abnormalities
Use caution when prescribing OsmoPrep for patients with conditions, or who are using medications, that increase the risk for fluid and electrolyte disturbances or may increase the risk of renal impairment, seizures, arrhythmias, or prolonged QT in the setting of fluid and electrolyte abnormalities. Consider additional patient evaluations as appropriate in patients taking these concomitant medications [see Warnings and Precautions (5.1,5.2,5.3)].
7.2 Potential for Reduced Drug Absorption
OsmoPrep can reduce the absorption of other coadministered oral medications. Administer oral medications at least 1 hour before or 1 hour after starting each OsmoPrep dose [see Dosage and Administration (2.1)].
7.3 Other Sodium Phosphate-Based Laxatives
Administration of additional sodium phosphate-based purgative or enema products with OsmoPrep may increase the risk of acute phosphate nephropathy. Avoid concomitant use [see Warnings and Precautions (5.1)].
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
There are no available data on sodium phosphate use in pregnant women to inform a drug-associated risk for adverse developmental outcomes.
Animal reproduction studies have not been conducted with sodium phosphate.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data available to assess the presence of sodium phosphate in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production.
The lack of clinical data during lactation precludes a clear determination of the risk of OsmoPrep to a child during lactation; therefore, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for OsmoPrep and any potential adverse effects on the breastfed child from OsmoPrep or from the underlying maternal condition.
8.5 Geriatric Use
Of the 599 patients in clinical trials receiving at least 48 grams of OsmoPrep, 134 (22%) were 65 years of age or older, while 27 (5%) were 75 years of age or older.
No overall differences in safety or effectiveness were observed between geriatric patients and younger patients. However, the mean phosphate levels in geriatric patients were greater than the phosphate levels in younger patients after OsmoPrep administration. The mean colonoscopy-day phosphate levels in patients 18-64, 65-74, and ≥ 75 years old who received the recommended OsmoPrep dosage regimen in Study 1were 7.0, 7.3, and 8.0 mg/dL, respectively. After OsmoPrep administration, the mean phosphate levels in patients 18-64, 65-74, and ≥ 75 years old were 7.4, 7.9, and 8.0 mg/dL, respectively. Greater sensitivity of some older individuals cannot be ruled out; therefore, use OsmoPrep with caution in geriatric patients. Advise geriatric patients to adequately hydrate before, during, and after the use of OsmoPrep.
Sodium phosphate is known to be substantially excreted by the kidney, and the risk of adverse reactions with sodium phosphate may be greater in patients with impaired renal function. Since geriatric patients are more likely to have impaired renal function, consider performing baseline and post-colonoscopy labs (phosphate, calcium, potassium, sodium, creatinine, and BUN) in these patients [see Warnings and Precautions (5.1)].
8.6 Renal Impairment
Sodium phosphate is substantially excreted by the kidney. Use OsmoPrep with caution in patients with severe renal impairment (creatinine clearance less than 30 mL/min) or patients taking concomitant medications that may affect renal function. These patients may be at risk for renal injury. Advise these patients of the importance of adequate hydration before, during and after the use of OsmoPrep, and consider performing baseline and post-colonoscopy labs (phosphate, calcium, potassium, sodium, creatinine, and BUN) in these patients [see Warnings and Precautions (5.1), Drug Interactions (7.1)].
10. Overdosage
Overdosage of OsmoPrep may lead to severe electrolyte disturbances, including hyperphosphatemia, hypocalcemia, hypernatremia, or hypokalemia, as well as dehydration and hypovolemia, with attendant signs and symptoms of these disturbances. Certain severe electrolyte disturbances resulting from overdose may lead to cardiac arrhythmias, seizure, renal failure, and death [see Warnings and Precautions (5.1, 5.2, 5.3)]. Monitor for fluid and electrolyte disturbances and treated symptomatically.
11. OsmoPrep Description
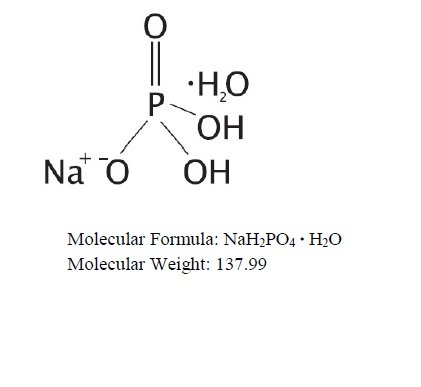
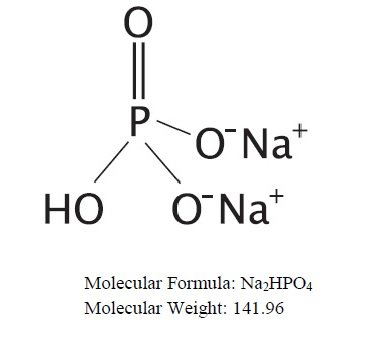
OsmoPrep (sodium phosphate monobasic monohydrate and sodium phosphate dibasic anhydrous) is an osmotic laxative used to clean the colon prior to colonoscopy. OsmoPrep is manufactured with a highly soluble tablet binder and does not contain microcrystalline cellulose (MCC). OsmoPrep tablets are oval, white to off-white compressed tablets, debossed with “SLX” on one side of the bisect and “102” on the other side of the bisect. Each OsmoPrep tablet, for oral administration, contains 1.102 grams of sodium phosphate monobasic monohydrate, USP and 0.398 grams of sodium phosphate dibasic anhydrous, USP for a total of 1.5 grams of sodium phosphate per tablet. Inert ingredients include polyethylene glycol 8000, NF; and magnesium stearate, NF. OsmoPrep is gluten-free.
The structural and molecular formulae and molecular weights of the active ingredients are shown below:
• Sodium phosphate monobasic monohydrate, USP
• Sodium phosphate dibasic anhydrous, USP
12. OsmoPrep - Clinical Pharmacology
12.1 Mechanism of Action
The primary mode of action is the osmotic effect of sodium, which induces a laxative effect. The physiological consequence is increased water retention in the lumen of the colon, resulting in loose stools.
12.2 Pharmacodynamics
Administration of the recommended OsmoPrep dosage regimen has a purgative effect for approximately 1 to 3 hours.
12.3 Pharmacokinetics
Pharmacokinetic studies with OsmoPrep have not been conducted. However, the following pharmacokinetic study was conducted with another oral tablet formulation of sodium phosphate which contain the same active ingredients as OsmoPrep at a dose that is 25% greater than the OsmoPrep dose.
Absorption
An open-label pharmacokinetic study of oral sodium phosphate in healthy subjects was performed to determine the concentration-time profile of serum inorganic phosphorus levels after oral sodium phosphate administration. All subjects received the approved dosing regimen for colon cleansing of 60 grams of sodium phosphate with a total liquid volume of 3.6 quarts. A 30-gram dose (20 tablets given as 3 tablets every 15 minutes with 8 ounces of clear liquids) was given beginning at 6 PM in the evening. The 30-gram dose (20 tablets given as 3 tablets every 15 minutes with 8 ounces of clear liquids) was repeated the following morning beginning at 6 AM.
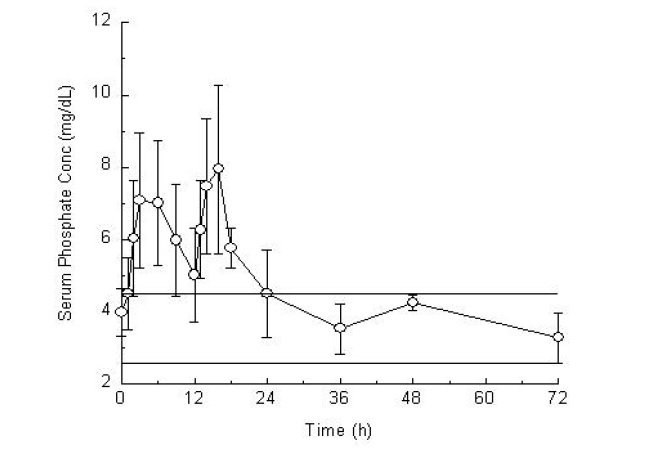
Twenty-three healthy subjects (mean age 57 years old; 57% male and 43% female; and 65% Hispanic, 30% Caucasian, and 4% African-American) participated in this pharmacokinetic study. The serum phosphorus level rose from a mean (± standard deviation) baseline of 4.0 (± 0.7) mg/dL to 7.7 (± 1.6 mg/dL), at a median of 3 hours after the administration of the first 30-gram dose of sodium phosphate tablets (see Figure 1). The serum phosphorus level rose to a mean of 8.4 (± 1.9) mg/dL, at a median of 4 hours after the administration of the second 30-gram dose of sodium phosphate tablets.
The serum phosphorus level remained above baseline for a median of 24 hours after the administration of the initial dose of sodium phosphate tablets (range 16 to 48 hours).
Figure 1. Mean (±Standard Deviation) Serum Phosphorus Concentrations
The upper (4.5 mg/dL) and lower (2.6 mg/dL) reference limits for serum phosphate are represented by solid bars.
Specific Populations
Male and Female Patients: No difference in serum phosphate AUC values were observed in the single pharmacokinetic study conducted with sodium phosphate tablets in 13 male and 10 female healthy subjects.
Elderly Patients: In a single pharmacokinetic study of sodium phosphate tablets, which included 6 elderly subjects, plasma half-life increased two-fold in subjects > 70 years of age compared to subjects < 50 years of age (3 subjects and 5 subjects, respectively) [see Use in Specific Populations (8.5)].
Patients with Renal Impairment: The effect of renal impairment on the pharmacokinetics of OsmoPrep has not been studied. The inorganic form of phosphate in the circulating plasma is excreted almost entirely by the kidneys [see Warnings and Precautions (5.1), Use in Specific Populations (8.6)].
14. Clinical Studies
The colon cleansing efficacy and safety of OsmoPrep was evaluated in a randomized, investigator-blinded, actively controlled, multicenter, U.S. trial in patients scheduled to have an elective colonoscopy (Study 1).
In Study 1, patients were randomized into one of the following three sodium phosphate treatment groups:
- •
- An oral tablet formulation of sodium phosphate containing 60 grams of sodium phosphate given in split doses (30 grams in the evening before the colonoscopy and 30 grams on the next day) with at least 3.6 quarts of clear liquids;
- •
- OsmoPrep containing 60 grams of sodium phosphate given in split doses (30 grams in the evening before the colonoscopy and 30 grams on the next day) with 2.5 quarts of clear liquids; and
- •
- OsmoPrep containing 48 grams of sodium phosphate (30 grams in the evening before the colonoscopy and 18 grams on the next day) with 2 quarts of clear liquids.
Patients were instructed to eat a light breakfast before noon on the day prior to the colonoscopy and then were told to drink only clear liquids after noon on the day prior to the colonoscopy.
The primary efficacy endpoint was the overall colon cleansing response rate in the 4-point Colonic Contents Scale. Response was defined as a rating of “excellent” or “good” on the 4-point scale as determined by the blinded colonoscopist. This trial was designed to assess the non-inferiority of the two OsmoPrep groups compared to the active control group.
The efficacy analysis included 704 adult patients who had an elective colonoscopy. Patients ranged in age from 21 to 89 years old (mean age 56 years old) with 55% female and 45% male patients. Race was distributed as follows: 87% Caucasian, 10% African American, and 3% other race. The OsmoPrep 60-gram and 48-gram treatment groups demonstrated non-inferiority compared to the active control. See Table 2 for the results.
|
|||||||
|
Treatment Arm (grams of sodium phosphate) |
No. of tablets taken at 6 PM on the day prior to colonoscopy |
No. of tablets taken the next day† |
Excellent |
Good |
Fair |
Inadequate |
Overall Response Rate (Excellent or Good) |
|
OsmoPrep 32 tabs (48 g) n=236 |
20 |
12 |
76% |
19% |
3% |
2% |
95% |
|
OsmoPrep 40 tabs (60 g) n=233 |
20 |
20 |
73% |
24% |
2% |
1% |
97% |
|
Sodium Phosphate Tablets 40 tabs (60 g) n=235 |
20 |
20 |
51% |
43% |
6% |
0% |
94% |
The OsmoPrep 60-gram dosage regimen had a similar overall response rate as the 48-gram dosage regimen and was associated with an increased risk of adverse reactions [see Adverse Reactions (6.1)]. Therefore, the OsmoPrep 60-gram dosage is not a recommended regimen [see Dosage and Administration (2.2)].
16. How is OsmoPrep supplied
OsmoPrep (sodium phosphate monobasic monohydrate and sodium phosphate dibasic anhydrous) tablets are supplied as oval, white to off-white compressed tablets, debossed with “SLX” on one side of the bisect and “102” on the other side of the bisect. Each 1.5 gram tablet contains 1.102 grams of sodium phosphate monobasic monohydrate, USP and 0.398 grams of sodium phosphate dibasic anhydrous, USP.
OsmoPrep is packaged in a multi-dose, child-resistant bottle containing 100 tablets: NDC 65649-701-41.
Each bottle contains two silica desiccant packets, which should not be ingested.
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Discard any unused portion.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Instruct patients:
- •
- On the importance of taking the recommended fluid regimen. Advise them to hydrate adequately with clear liquids before, during, and after the use of OsmoPrep [see Dosage and Administration (2.1), Warnings and Precautions (5.1)]. Examples of clear liquids can be found in the Medication Guide.
- •
- Two doses of OsmoPrep are required for a complete preparation for colonoscopy.
- •
- Do not eat or drink alcohol, milk, anything colored red or purple or any other foods containing pulp material.
- •
- Not to take other laxatives or enemas made with sodium phosphate while they are taking OsmoPrep [see Drug Interactions (7.3)].
- •
- Not to take oral medications within one hour before or after starting each dose of OsmoPrep [see Drug Interactions (7.2)].
- •
- To contact their healthcare provider if they develop significant vomiting or signs of dehydration after taking OsmoPrep or if they experience cardiac arrhythmias or seizures [see Warnings and Precautions (5.1,5.2,5.3)].
- •
- To seek immediate medical care should signs and symptoms of a hypersensitivity reaction occur [see Warnings and Precautions (5.7)].
Distributed by:
Salix Pharmaceuticals, a division of Bausch Health US, LLC,
Bridgewater, NJ 08807 USA
U.S. Patent Number: 7,687,075 and other pending applications.
®/™ are trademarks of Salix Pharmaceuticals, Inc. or its affiliates.
All other product/brand names are trademarks of their respective owners.
© 2019 Salix Pharmaceuticals, Inc. or its affiliates
9516403
70014026
|
Medication Guide
(sodium phosphate monobasic monohydrate and sodium phosphate dibasic anhydrous) tablets |
||
|
Read this Medication Guide before you start taking OsmoPrep. This information does not take the place of talking with your doctor about your medical condition or your treatment. |
||
|
What is the most important information I should know about OsmoPrep? OsmoPrep can cause serious side effects, including: Serious kidney problems. Rare, but serious kidney problems can happen in people who take medicines made with sodium phosphate, including OsmoPrep, to clean the colon before colonoscopy. These kidney problems can sometimes lead to kidney failure or the need for dialysis for a long time. These problems often happen within a few days, but sometimes may happen several months after taking OsmoPrep. Conditions that can make you more at risk for having serious kidney problems with OsmoPrep include if you:
Increased age may increase your risk for having serious kidney problems with OsmoPrep. Severe fluid loss (dehydration) and severe changes in body salts in the blood (electrolytes). People who take medicines to clean their colon before a colonoscopy, including OsmoPrep, can have severe loss of body fluid, with severe changes in body salts in the blood. These changes can be serious and can cause:
Tell your doctor if you have any of these symptoms of loss of too much body fluid (dehydration) while taking OsmoPrep:
See “What are the possible side effects of OsmoPrep?” for more information about side effects. Important information about taking OsmoPrep:
See “How should I take OsmoPrep?” for more information about how to take OsmoPrep. |
||
|
What is OsmoPrep? OsmoPrep is a prescription medicine used in adults to clean your colon before a colonoscopy. OsmoPrep cleans your colon by causing you to have diarrhea. Cleaning your colon helps your doctor see the inside of your colon more clearly during the colonoscopy. It is not known if OsmoPrep is safe and effective in children. |
||
|
Who should not take OsmoPrep? Do not take OsmoPrep if you:
|
||
|
What should I tell my doctor before taking OsmoPrep? Before you take OsmoPrep, tell your doctor about all of your medical conditions, including if you:
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. OsmoPrep may affect how other medicines work. Do not take medicines by mouth within 1 hour of starting each dose of OsmoPrep or 1 hour after you start taking each dose of OsmoPrep. Especially tell your doctor if you take:
Ask your doctor or pharmacist if you are not sure if you take any of the medicines listed above. Know the medicines you take. Keep a list of your medicines to show your doctor or pharmacist when you get a new prescription. |
||
|
How should I take OsmoPrep?
On the day of your colonoscopy, take a total of 12 OsmoPrep tablets, starting about 3 to 5 hours before your colonoscopy, as follows:
If you take too much OsmoPrep, call your doctor or get medical help right away. |
||
|
What should I avoid while taking OsmoPrep?
|
||
|
What are the possible side effects of OsmoPrep? OsmoPrep can cause serious side effects, including:
The most common side effects of OsmoPrep are:
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of OsmoPrep. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
How should I store OsmoPrep?
Keep OsmoPrep and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of OsmoPrep. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use OsmoPrep for a condition for which it was not prescribed. Do not give OsmoPrep to other people, even if they are going to have the same procedure that you are. It may harm them. You can ask your doctor or pharmacist for information that is written for health professionals. |
||
|
What are the ingredients in OsmoPrep? Active ingredients: sodium phosphate monobasic monohydrate and sodium phosphate dibasic anhydrous Inactive ingredients: polyethylene glycol 8000 and magnesium stearate. OsmoPrep is gluten-free. Distributed by: Salix Pharmaceuticals, a division of Bausch Health US, LLC, Bridgewater, NJ 08807 USA U.S. Patent Number: 7,687,075 and other pending applications. ®/™ are trademarks of Salix Pharmaceuticals, Inc. or its affiliates. © 2019 Salix Pharmaceuticals, Inc. or its affiliates For more information, call 1-800-321-4576 (toll-free) or go to www.Salix.com. |
||
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 03/2019
9516403
70014026
| OSMOPREP
sodium phosphate, monobasic, monohydrate, sodium phosphate, dibasic anhydrous tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Salix Pharmaceuticals, Inc. (793108036) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Novel Laboratories, Inc. | 793518643 | MANUFACTURE(65649-701) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Carton Service | 928861723 | PACK(65649-701) | |
More about OsmoPrep (sodium biphosphate / sodium phosphate)
- Check interactions
- Compare alternatives
- Reviews (6)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: laxatives