Heparin Lock Flush Solution: Package Insert / Prescribing Info
Package insert / product label
Generic name: heparin sodium
Dosage form: injection, solution
Drug class: Heparins
Medically reviewed by Drugs.com. Last updated on Mar 24, 2025.
On This Page
Heparin Lock Flush Solution Description
SOLUTION IS INTENDED FOR MAINTENANCE OF PATENCY OF INTRAVENOUS INJECTION DEVICES ONLY AND IS NOT TO BE USED FOR ANTICOAGULANT THERAPY.
Heparin Lock Flush Solution, USP is a sterile, nonpyrogenic, hypertonic preparation of heparin sodium injection, USP with sodium chloride in water for injection.
Each milliliter (mL) contains: Heparin sodium, 10 or 100 USP units (derived from porcine intestinal mucosa); sodium chloride 9 mg; edetate disodium, anhydrous 0.1 mg added as a stabilizer; and benzyl alcohol 9 mg added as preservative in water for injection. May contain sodium hydroxide for pH adjustment. pH 6.5 (5.0 to 7.5). The osmolar concentrations of these solutions are 392 mOsmol/L (calc). Repeated withdrawals may be made from the multiple-dose vial.
Heparin Lock Flush Solution, USP is intended for maintenance of patency of intravenous injection devices only and is not to be used for anticoagulant therapy.
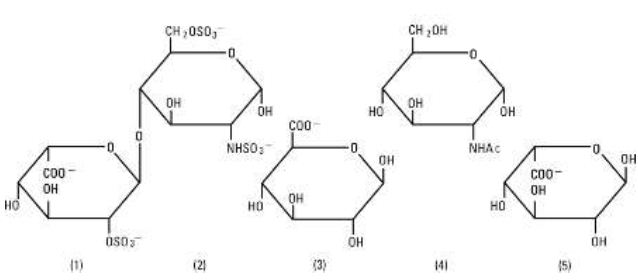
Heparin Sodium, USP is a heterogenous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans having anticoagulant properties. Although others may be present, the main sugars occurring in heparin are: (1) a-L-iduronic acid 2-sulfate, (2) 2-deoxy-2-sulfamino-a-D-glucose 6-sulfate, (3) b-D-glucuronic acid, (4) 2-acetamido-2-deoxy-a-D-glucose, and (5) a-L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2)greater than(1)greater than(4)greater than(3)greater than(5), and are joined by glycosidic linkages, forming polymers of varying sizes. Heparin is strongly acidic because of its content of covalently linked sulfate and carboxylic acid groups. In heparin sodium the acidic protons of the sulfate units are partially replaced by sodium ions. The potency is determined by a biological assay using a USP reference standard based on units of heparin activity per milligram.
Structure of Heparin Sodium (representative subunits):
Sodium Chloride, USP is chemically designated NaCl, a white crystalline compound freely soluble in water. The semi-rigid vials are fabricated from a specially formulated polyolefin. It is a copolymer of ethylene and propylene. The safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers. The container requires
no vapor barrier to maintain the proper drug concentration.
Heparin Lock Flush Solution - Clinical Pharmacology
Heparin inhibits reactions that lead to the clotting of blood and the formation of fibrin clots both in vitro and in vivo. Heparin acts at multiple sites in the normal coagulation system. Small amounts of heparin in combination with antithrombin III (heparin cofactor) can inhibit thrombosis by inactivating activated Factor X and inhibiting the conversion of prothrombin to thrombin. Once active thrombosis has developed, larger amounts of heparin can inhibit further coagulation by inactivating thrombin and preventing the conversion of fibrinogen to fibrin. Heparin also prevents the formation of a stable fibrin clot in inhibiting the activation of the fibrin stabilizing factor. Bleeding time is usually unaffected by heparin. Clotting time is prolonged by full therapeutic doses of heparin; in most cases, it is not measurably affected by low doses of heparin. Patients over 60 years of age, following similar doses of heparin, may have higher plasma levels of heparin and longer activated partial thromboplastin times (APTTs) compared with patients under 60 years of age. Peak plasma levels of heparin are achieved 2 to 4 hours following subcutaneous administration, although there are considerable individual variations. Loglinear plots of heparin plasma concentrations with time for a wide range of dose levels are linear which suggests the absence of zero order processes. Liver and the reticuloendothelial system are the site of biotransformation. The biphasic elimination curve, a rapidly declining alpha phase (t½ =10#) and after the age of 40 a slower beta phase, indicates uptake in organs. The absence of a relationship between anticoagulant half-life and concentration of half-life may reflect factors such as protein binding of heparin. Heparin does not have fibrinolytic activity; therefore, it will not lyse existing clots. Heparin Lock Flush Solution does not induce systemic anticoagulant action at single doses of 10 or 100 USP units per mL when used for maintenance of patency of intravenous injection devices. It may interfere with laboratory tests on blood samples withdrawn from such devices, unless the volume of in situ heparin-saline, equal to that of the priming volume of the catheter, is aspirated and discarded before such samples are taken. Isotonic concentrations of sodium chloride are suitable for parenteral replacement of chloride losses that exceed or equal the sodium loss. Hypotonic concentrations of sodium chloride are suited for parenteral maintenance of water requirements when only small quantities of salt are desired.
Sodium chloride in water dissociates to provide sodium (Na+) and chloride (Cl-) ions. Sodium (Na+) is the principal cation of the
extracellular fluid and plays a large part in the therapy of fluid and electrolyte disturbances. Chloride (Cl-) has an integral role in buffering action when oxygen and carbon dioxide exchange occurs in the red blood cells. The distribution and excretion of sodium (Na+) are largely under the control of the kidney which maintains a balance between intake and output. The small volume of fluid and amount of sodium chloride provided by Heparin Lock Flush Solution, USP, when used only as a vehicle for maintaining patency of an intravenous injection device, is unlikely to exert a significant effect on fluid and electrolyte balance except possibly in very small infants.
Indications and Usage for Heparin Lock Flush Solution
Heparin Lock Flush Solution, USP is indicated only to maintain patency of an intravenous injection device. It may be used following initial placement of the device in the vein, after each injection of a medication or after withdrawal of blood for laboratory analysis. The solution is not to be used for anticoagulation therapy.
Contraindications
Due to the potential toxicity of benzyl alcohol in neonates, solutions containing benzyl alcohol must not be used in this patient population. Parenteral preparations with benzyl alcohol should not be used for fluid or sodium chloride replacement. Heparin sodium should not be used in patients: With hypersensitivity to heparin; With an uncontrollable active bleeding state (see WARNINGS), except when this is due to disseminated intravascular coagulation; With inability to perform suitable blood-coagulation tests, e.g., whole-blood clotting time, partial thromboplastin time, etc. at required intervals. There is usually no need to monitor effect of low-dose heparin in patients with normal coagulation parameters.
Warnings
Benzyl alcohol, a preservative in the multiple-dose vial preparations of Heparin Lock Flush Solution, USP has been associated with toxicity in neonates. Benzyl alcohol has been reported to be associated with a fetal “Gasping Syndrome” in premature infants. Data are unavailable on the toxicity of other preservatives in this age group. Preservative-free Heparin Lock Flush Solution, USP should be used for maintaining patency of intravenous injection devices in neonates. Heparin Lock Flush Solution, USP is not intended for intramuscular use, systemic anticoagulation or injection by any parenteral route of administration.
Hypersensitivity: Patients with documented hypersensitivity to heparin should be given the drug only in clearly life-threatening situations.
Hemorrhage: Hemorrhage can occur at virtually any site in patients receiving heparin. An unexplained fall in hematocrit, fall in blood pressure, or any other unexplained symptom should lead to serious consideration of a hemorrhagic event. Heparin sodium should be used with extreme caution in disease states in which there is increased danger of hemorrhage. Some of the conditions in which increased danger of hemorrhage exists are:
Cardiovascular— Subacute bacterial endocarditis. Severe hypertension.
Surgical— During and immediately following (a) spinal tap or spinal anesthesia or (b) major surgery, especially involving the brain, spinal cord or eye.
Hematologic — Conditions associated with increased bleeding tendencies, such as hemophilia, thrombocytopenia, and some vascular purpuras.
Gastrointestinal — Ulcerative lesions and continuous tube drainage of the stomach or small intestine.
Other— Menstruation, liver disease with impaired hemostasis.
Coagulation Testing: If the coagulation test is unduly prolonged or if hemorrhage occurs, heparin sodium should be discontinued promptly. Heparin solutions having a concentration of 10 USP Heparin Units/mL may alter the results of blood coagulation tests. Heparin concentrations greater than 10 USP Heparin Units/mL will alter the results of blood coagulation tests.
Thrombocytopenia: Thrombocytopenia has been reported to occur in patients receiving heparin with a reported incidence of 0 to 30%. Mild thrombocytopenia (count greater than 100,000/mm3) may remain stable or reverse even if heparin is continued. However, thrombocytopenia of any degree should be monitored closely. If the count falls below 100,000/mm3 or if recurrent thrombosis
develops (see White Clot Syndrome, PRECAUTIONS), the heparin product should be discontinued. If continued heparin therapy is essential, administration of heparin from a different organ source can be reinstituted with caution. Solutions containing sodium ions should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency and in clinical states in which there exists edema with sodium retention.
Precautions
Do not use unless solution is clear and container is undamaged. Use aseptic technique for single or multiple entry and withdrawal from all containers. Since heparin sodium in this product is derived from animal tissue, it should be used with caution in patients with a history of allergy.
Interference with Laboratory Tests
Heparin interferes with competitive protein binding methods for serum thyroxine determinations resulting in falsely elevated levels. Heparin Lock Flush Solution may interfere with laboratory analyses or alter the results of blood chemistry tests such as glucose, serum sodium and serum chloride, blood coagulation studies, etc. (See CLINICAL PHARMACOLOGY.) The following information which pertains to the use of heparin sodium as a systemic anticoagulant is included as a matter of interest only since it is not known to apply to the use of the drug for heparin lock.
General:
a. White Clot Syndrome:
It has been reported that patients on heparin may develop new thrombus formation in association with thrombocytopenia resulting from irreversible aggregation of platelets induced by heparin, the so-called “white clot syndrome”. The process may lead to severe thromboembolic complications like skin necrosis, gangrene of the extremities that may lead to amputation, myocardial infarction, pulmonary embolism, stroke, and possibly death. Therefore, heparin administration should be promptly discontinued if a patient develops new thrombosis in association with thrombocytopenia.
b. Heparin Resistance:
Increased resistance to heparin is frequently encountered in fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in postsurgical patients.
c. Increased Risk in Older Patients, Especially Women:
A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age.
Drug Interactions:
As Heparin Lock Flush Solution, USP is intended only for use in maintaining patency of intravenous injection devices, no additives should be made to multiple-dose vials.
Oral anticoagulants: Heparin sodium may prolong the one-stage prothrombin time. Therefore, when heparin sodium is given with dicumarol or warfarin sodium, a period of at least 5 hours after the last intravenous dose should elapse before blood is drawn if a valid PROTHROMBIN time is to be obtained.
Platelet inhibitors: Drugs such as acetylsalicylic acid, dextran, phenylbutazone, ibuprofen, indomethacin, dipyridamole, hydroxychloroquine and others that interfere with platelet-aggregation reactions (the main hemostatic defense of heparinized patients) may induce bleeding and should be used with caution in patients receiving heparin sodium.
Other interactions: Digitalis, tetracyclines, nicotine, anti-histamines or I.V. nitroglycerin may partially counteract the anticoagulant action of heparin sodium.
Drug/Laboratory Interactions:
Hyperaminotransferasemia: Significant elevations of aminotransferase (SGOT [S-AST] and SGPT [S-ALT]) levels have occurred in a high percentage of patients (and healthy subjects) who have received heparin. Since aminotransferase determinations are important in the differential diagnosis of myocardial infarction, liver disease, and pulmonary emboli, rises that might be caused by drugs (like heparin) should be interpreted with caution.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
No long-term studies in animals have been performed to evaluate carcinogenic potential of heparin. Also, no reproduction studies in animals have been performed concerning mutagenesis or impairment of fertility.
Pregnancy:
Teratogenic Effects: Pregnancy Category C. Animal reproduction studies have not been conducted with heparin sodium or sodium chloride. It is also not known whether heparin sodium or sodium chloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Heparin sodium or sodium chloride should be given to a pregnant woman only if clearly needed.
Nonteratogenic Effects: Heparin does not cross the placental barrier.
Nursing Mothers:
Heparin is not excreted in human milk.
Pediatric Use:
Safety and effectiveness in pediatric patients have not been established. Not for use in neonates (see WARNINGS).
Geriatric Use:
A higher incidence of bleeding has been reported in patients over 60 years of age, especially women (see PRECAUTIONS, General). Clinical studies indicate that lower doses of heparin may be indicated in these patients (see CLINICAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION).
Adverse Reactions/Side Effects
Although adverse reactions to intravenous intramuscular or subcutaneous injection of 0.9% benzyl alcohol are not known to occur in man, experimental studies of small volume parenteral preparations containing 0.9% benzyl alcohol in several species of animals have indicated that an estimated intravenous dose up to 30 mL may be safely given to an adult without toxic effects. Administration of an estimated 9 mL to 6 kg infant is potentially capable of producing blood pressure changes. Heparin Lock Flush Solution is not known to cause adverse local or systemic effects of any kind. Although a remote possibility of hypersensitivity reaction exists with entry of extremely small subtherapeutic amounts of the solution into the circulation, such an occurrence has not been reported.
Heparin Lock Flush Solution Dosage and Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Slight discoloration does not alter potency. (See PRECAUTIONS.) Heparin Lock Flush Solution, USP 10 or 100 USP Units/mL, is injected as a single dose into an intravenous injection device using a volume of solution equivalent to that of the indwelling venipuncture device. A single dose should be injected following venipuncture when the indwelling device is not to be used immediately. After each use
of the indwelling venipuncture device for injection or infusion of medication, or withdrawal of blood samples, another dose should be injected to restore the effectiveness of the heparin lock. The amount of heparin solution is sufficient to prevent clotting within the lumen of indwelling venipuncture devices (usually not holding more than 0.2 to 0.3 mL) for up to twenty-four hours. When the indwelling device is used to administer a drug which is incompatible with heparin, the entire heparin lock set should be flushed with 0.9% Sodium Chloride Injection, USP before and after the medication is administered. Following the second flush, another dose of heparin solution should be injected to restore the effectiveness of the heparin lock. When the indwelling device is used for repeated withdrawal of blood samples for laboratory analyses and the presence of heparin or saline is likely to interfere with or alter results of the desired blood tests, the in situ heparin flush solution should be cleared from the device by aspirating and discarding 1 mL before the desired blood sample is drawn. (See PRECAUTIONS.)
How is Heparin Lock Flush Solution supplied
Heparin Lock Flush Solution, USP is supplied in the following dosage forms:
| List No. | Dosage Form | Heparin Sodium (USP Units/mL) | Solution Volume (mL) |
| 0409-1151-12 | LifeShield Plastic Multple-dose Fliptop vial | 10 | 10 |
| 0409-1151-70 | Plastic Mutliple- dose Fliptop vial | 10 | 10 |
| 0409-1151-78 | Plastic Mutliple- dose Fliptop vial | 10 | 30 |
| 0409-1152-14 | LifeShield Plastic Multple-dose Fliptop vial | 100 | 30 |
| 0409-1152-70 | Plastic Mutliple- dose Fliptop vial | 100 | 10 |
| 0409-1152-78 | Plastic Mutliple- dose Fliptop vial | 100 | 30 |
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Rev: June 2007
EN-1543
Printed in USA
HOSPIRA, INC., LAKE FOREST, IL 60045 USA
| HEPARIN LOCK FLUSH
heparin sodium solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - General Injectables & Vaccines, Inc (108250663) |
Frequently asked questions
More about heparin
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (2)
- Latest FDA alerts (19)
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Drug class: heparins
- Breastfeeding
- En español

