Dianeal Low Calcium Peritoneal Dialysis Solution Prescribing Information
Package insert / product label
Generic name: dextrose monohydrate, sodium chloride, sodium lactate, calcium chloride and magnesium chloride
Dosage form: injection, solution
Drug class: Intravenous nutritional products
Medically reviewed by Drugs.com. Last updated on Mar 25, 2024.
On This Page
Dianeal Low Calcium Peritoneal Dialysis Solution Description
Dianeal® Low Calcium peritoneal dialysis solutions are sterile, nonpyrogenic solutions in UltraBag™ containers for intraperitoneal administration only. They contain no bacteriostatic or antimicrobial agents. UltraBag™ containers are designed with an integrated “Y” set and drain container for infusion and drainage of Dianeal® Low Calcium when disconnection of the “Y” set from the transfer set during dwell is desired.
Composition, calculated osmolarity, pH and ionic concentrations are shown in the following table.
| Composition/100mL | OSMOLARITY (mOsmol/L) (calc) | pH | Ionic Concentration (mEq/L) | How Supplied | ||||||||||||
| Dextrose, Hydrous, USP | Sodium Chloride, USP (NaCl) | Sodium Lactate (C3H5NaO3) | Calcium Chloride, USP (CaCl2·2H2O) | Magnesium Chloride, USP (MgCl2·H2O) | Sodium | Calcium | Magnesium | Fill Chloride | Lactate | Fill Volume (mL) | Container (mL) | Code | NDC | |||
| Dianeal®Low Calcium Peritoneal Dialysis Solution with 1.5% Dextrose | 1.5 g | 538 mg | 448 mg | 18.3 mg | 5.08 mg | 344 | 5.2 (4.0 to 6.5) | 132 | 2.5 | 0.5 | 95 | 40 | 1500 2000 2500 3000 | 2000 2000 3000 5000 | 5B9765 5B9766 5B9768 5B9757 | 0941-0424-51 0941-0424-52 0941-0424-53 0941-0424-55 |
| Dianeal®Low Calcium Peritoneal Dialysis Solution with 2.5% Dextrose | 2.5 g | 538 mg | 448 mg | 18.3 mg | 5.08 mg | 395 | 5.2 (4.0 to 6.5) | 132 | 2.5 | 0.5 | 95 | 40 | 1500 2000 2500 3000 | 2000 2000 3000 5000 | 5B9775 5B9776 5B9778 5B9758 | 0941-0430-51 0941-0430-52 0941-0430-53 0941-0430-55 |
| Dianeal®Low Calcium Peritoneal Dialysis Solution with 4.25% Dextrose | 4.25 g | 538 mg | 448 mg | 18.3 mg | 5.08 mg | 483 | 5.2 (4.0 to 6.5) | 132 | 2.5 | 0.5 | 95 | 40 | 1500 2000 2500 3000 | 2000 2000 3000 5000 | 5B9795 5B9796 5B9798 5B9759 | 0941-0433-51 0941-0433-52 0941-0433-53 0941-0433-55 |
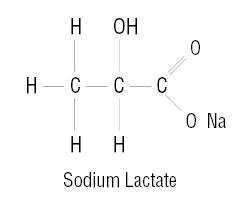
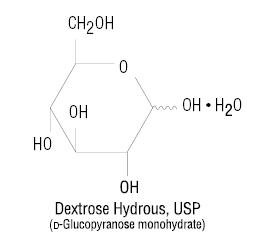
The plastic container tubing set is fabricated from polyvinyl chloride (PL 146® Plastic). Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period. The amount of water that can permeate from inside the solution container into the overpouch is insufficient to affect the solution significantly. Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the plastic container materials.
Related/similar drugs
Normosol-R
Dianeal Low Calcium Peritoneal Dialysis Solution - Clinical Pharmacology
Peritoneal dialysis is a procedure for removing toxic substances and metabolites normally excreted by the kidneys, and for aiding in the regulation of fluid and electrolyte balance.
The procedure is accomplished by instilling peritoneal dialysis fluid through a conduit into the peritoneal cavity. Toxic substances and metabolites, present in high concentration in the blood, cross the peritoneal membrane into the dialyzing fluid. Dextrose in the dialyzing fluid is used to produce a solution hyperosmolar to the plasma, creating an osmotic gradient which facilitates fluid removal from the patient’s plasma into the peritoneal cavity. After a period of time, (dwell time), the fluid is drained by gravity from the cavity.
The solution does not contain potassium. In situations in which there is a normal serum potassium level or hypokalemia, the addition of potassium chloride (up to a concentration of 4 mEq/L) may be indicated to prevent severe hypokalemia. Addition of potassium chloride should be made after careful evaluation of serum and total body potassium and only under the direction of a physician.
Clinical studies have demonstrated the use of this solution resulted in significant increases in serum CO2 and decreases in serum magnesium levels. The decrease in magnesium levels did not cause clinically significant hypomagnesemia.
Indications and Usage for Dianeal Low Calcium Peritoneal Dialysis Solution
Dianeal® Low Calcium peritoneal dialysis solutions in UltraBag™ containers are indicated for use in chronic renal failure patients being maintained on continuous ambulatory peritoneal dialysis when nondialytic medical therapy is judged to be inadequate.
Warnings
Not for Intravenous Injection.
Use aseptic technique. Contamination of Luer lock connector may result in peritonitis.
An improper clamping sequence may result in infusion of air into the peritoneum.
Peritoneal dialysis should be done with great care, if at all, in patients with a number of conditions, including disruption of the peritoneal membrane or diaphragm by surgery or trauma, extensive adhesions, bowel distention, undiagnosed abdominal disease, abdominal wall infection, hernias or burns, fecal fistula or colostomy, tense ascites, obesity, large polycystic kidneys, recent aortic graft replacement, lactic acidosis, and severe pulmonary disease. When assessing peritoneal dialysis as the mode of therapy in such extreme situations, the benefits to the patient must be weighed against the possible complications.
An accurate fluid balance record must be kept and the weight of the patient carefully monitored to avoid over or under hydration with severe consequences, including congestive heart failure, volume depletion, and shock.
Excessive use of Dianeal® Low Calcium peritoneal dialysis solution with 4.25% dextrose during a peritoneal dialysis treatment can result in significant removal of water from the patient.
Stable patients undergoing maintenance peritoneal dialysis should have routine periodic evaluation of blood chemistries and hematologic factors, as well as other indicators of patient status.
If the resealable rubber plug on the medication port is missing or partially removed, do not use product.
After removing overpouch, check for minute leaks by squeezing container firmly. If leaks are found, discard the solution because the sterility may be impaired.
After the pull ring has been removed from the outlet, check for broken connector frangible seal as evidenced by continuous fluid flow from port. A few drops of solution within the connector or protector cap may be present. If a continuous stream or droplets of fluid are noted, discard solution because sterility may be impaired.
During solution drainage, fibrin strands may be observed in the solution and may become attached to the connector frangible closure. In occasional instances, partial or complete obstruction of draining may occur. Manipulation of the connector frangible closure in the tubing may free the fibrin obstruction.
Precautions
General:
Do not administer unless solution is clear.
Aseptic technique must be used throughout the procedure and at its termination in order to reduce the possibility of infection.
Significant losses of protein, amino acids and water soluble vitamins may occur during peritoneal dialysis. Replacement therapy should be provided as necessary.
When prescribing the solution to be used for an individual patient, consideration should be given to the potential interaction between the dialysis treatment and therapy directed at other existing illnesses. For example, rapid potassium removal may create arrhythmias in cardiac patients using digitalis or similar drugs; digitalis toxicity may be masked by elevated potassium or magnesium, or by hypocalcemia. Correction of electrolytes by dialysis may precipitate signs and symptoms of digitalis excess. Conversely, toxicity may occur at suboptimal dosages of digitalis if potassium is low or calcium high. Azotemic diabetics require careful monitoring of insulin requirements during and following dialysis with dextrose containing solutions.
Laboratory tests:
Serum electrolytes, magnesium, bicarbonate levels and fluid balance should be periodically monitored.
Carcinogenesis, mutagenesis, impairment of fertility:
Studies to evaluate the carcinogenic or mutagenic potential of this product, or its potential to affect fertility adversely, have not been performed.
Pregnancy:
Pregnancy Category C.
Animal reproduction studies have not been conducted with Dianeal® Low Calcium peritoneal dialysis solution. It is also not known whether Dianeal® Low Calcium peritoneal dialysis solution can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Dianeal® Low Calcium peritoneal dialysis solution should be given to a pregnant woman only if clearly needed.
Adverse Reactions/Side Effects
Adverse reactions to peritoneal dialysis include mechanical and solution related problems as well as the results of contamination of equipment or improper technique in catheter placement. Abdominal pain, bleeding, peritonitis, subcutaneous infection around the peritoneal catheter, catheter site infection, catheter blockage, difficulty in fluid removal, and ileus are among the complications of the procedure. Solution related adverse reactions may include peritonitis, electrolyte and fluid imbalances, hypovolemia, hypervolemia, hypotension, hypertension, disequilibrium syndrome, allergic symptoms, and muscle cramping.
Dianeal Low Calcium Peritoneal Dialysis Solution Dosage and Administration
Dianeal® Low Calcium solutions are intended for intraperitoneal administration only.
It is recommended that adult patients being placed on continuous ambulatory peritoneal dialysis should be appropriately trained in a program which is under supervision of a physician. Training materials are available from Baxter Healthcare Corporation, Deerfield, IL, 60015 USA to facilitate this training.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
The frequency of treatment, formulation, exchange volume, duration of dwell, and length of dialysis should be selected by the physician responsible for and supervising the treatment of the individual patient.
To avoid the risk of severe dehydration and hypovolemia and to minimize the loss of protein, it is advisable to select the peritoneal dialysis solution with the lowest level of osmolarity consistent with the fluid removal requirements for that exchange.
Heating the dialysis solution to 37°C (98.6°F) may decrease discomfort.
Additives may be incompatible. Do not store solution containing additives.
Approximately two liters of dialysis solution are instilled into the peritoneal cavity of adults and the peritoneal access device is then clamped. The solution remains in the cavity for dwell times of 4 to 8 hours during the day and 8 to 12 hours overnight. At the conclusion of each dwell period, the access device is opened, the solution drained and fresh solution instilled. The procedure is repeated 3 to 5 times per day, 6 to 7 days per week. Solution exchange frequency should be individualized for adequate biochemical and fluid volume control. The majority of exchanges will utilize 1.5% or 2.5% dextrose containing peritoneal dialysis solutions, with 4.25% dextrose containing solutions being used when extra fluid removal is required. Patient weight is used as the indicator of the need for fluid removal.
DIRECTIONS FOR USE
Use aseptic technique.
For complete system preparation, see directions accompanying ancillary equipment.
Preparation for Administration
1. Gather supplies.
2. Tear the container overpouch firmly down the side from top slit and remove. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity should diminish gradually.
3. Place container on work station.
4. Uncoil tubing.
5. Inspect the patient connector to ensure the pull ring is attached. Do not use if pull ring is not attached to the connector.
6. Inspect tubing and drain container for presence of solution. If solution is noted, discard units.
NOTE: Small water droplets are acceptable.
7. Squeeze container to check for leaks and broken solution frangible. Note solution flow past the frangible. Discard if container or solution frangible leaks, because sterility may be impaired.
If Supplemental Medication is Prescribed:
1. Inspect container to ensure resealable rubber medication port is in place. Discard if rubber injection port is not attached to container port.
2. Put on a mask.
3. Prepare medication port according to aseptic technique.
4. Using a syringe with a 1 inch long, 19 to 25 gauge needle, puncture resealable medication port and inject medication.
5. Position container with medication port facing upward. Squeeze and tap medication port to empty solution. Mix solution by vigorously agitating container.
Administration:
1. Put on mask and wash hands.
2. Insure patient transfer set is closed.
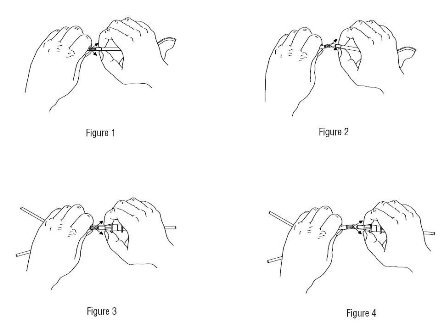
3. Break connector frangible (blue) by grasping the tubing above the top of the frangible and pulling forward and backward until the frangible separates from base. See Figures 1 and 2.
4. Remove pull ring from the patient connector.
5. Remove disconnect cap from patient transfer set. Immediately attach patient transfer set connector to the patient connector by twisting the connector until firmly secured.
6. Clamp solution line.
7. Break solution frangible (green) by grasping the tubing above the top of the frangible and pulling forward and backward until the frangible separates. See Figures 3 and 4.
8. Hang the new solution container.
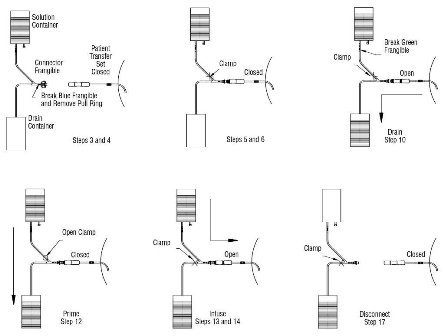
9. Place the drainage container below the level of the peritoneum.
10. Open transfer set clamp to drain solution from peritoneum. Warning: During solution drainage, fibrin strands may become attached to the connector frangible closure. Manipulation of the connector frangible closure in the tubing may free any fibrin obstruction that occurs.
11. Close transfer set line clamp after drainage is complete.
12. Open solution line clamp and allow the new solution to flow into the drainage container for 5 seconds to prime line.
13. Clamp drain line.
14. Open transfer set clamp and allow the solution to flow into the peritoneum.
15. Close transfer set clamp when infusion is complete.
16. Prepare a new disconnect cap following the directions accompanying the cap.
17. Disconnect the patient transfer set from the UltraBag™ and attach a new disconnect cap to the transfer set.
Administration Procedure for the UltraBag™ Container Exchange
(The Steps Refer to the Corresponding Administration Steps)
How is Dianeal Low Calcium Peritoneal Dialysis Solution supplied
Dianeal® Low Calcium peritoneal dialysis solutions in UltraBag™ containers are available in nominal size flexible containers as shown in the table in the DESCRIPTION section.
All Dianeal® Low Calcium peritoneal dialysis solutions have overfills which are declared on container labeling.
Freezing of solutions may occur at temperatures below 0°C (32°F). Allow to thaw naturally in ambient conditions and thoroughly mix contents by shaking.
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. It is recommended the product be stored at room temperature (25°C/77°F).
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
Printed in USA
©Copyright 1992, Baxter Healthcare Corporation. All rights reserved.
07-19-26-995
2002/02
*BAR CODE POSITION ONLY
071926995
PRINCIPAL DISPLAY PANEL

Container Label
5B9766
NDC 0941-0424-52
2000 mL
(APPROX 80 mL EXCESS)
Baxter Logo
Dianeal
Low Calcium (2.5 mEq/L)
Peritoneal Dialysis Solution
with 1.5% Dextrose
EACH 100 mL CONTAINS 1.5 g DEXTROSE HYDROUS USP
538 mg SODIUM CHLORIDE USP 448 mg SODIUM
LACTATE 18.3 mg CALCIUM CHLORIDE USP 5.08 mg
MAGNESIUM CHLORIDE USP pH 5.2 (4.0 TO 6.5)
mEq/L SODIUM - 132 CALCIUM - 2.5 MAGNESIUM - 0.5
CHLORIDE - 95 LACTATE - 40
OSMOLARITY - 344 mOsmol/L (CALC)
STERILE NONPYROGENIC
POTASSIUM CHLORIDE TO BE ADDED ONLY
UNDER THE DIRECTION OF A PHYSICIAN
READ PACKAGE INSERT FOR FULL INFORMATION
FOR INTRAPERITONEAL ADMINISTRATION ONLY
DOSAGE AS DIRECTED BY A PHYSICIAN
CAUTIONS SQUEEZE AND INSPECT INNER BAG
WHICH MAINTAINS PRODUCT STERILITY DISCARD
IF LEAKS ARE FOUND
DO NOT USE UNLESS SOLUTION IS CLEAR
DISCARD UNUSED PORTION
Rx ONLY
STORE UNIT IN MOISTURE BARRIER OVERWRAP AT
ROOM TEMPERATURE (25° C/77°F) UNTIL READY TO
USE
AVOID EXCESSIVE HEAT SEE INSERT
UltraBag CONTAINER PL 146 PLASTIC
BAXTER DIANEAL ULTRABAG AND PL 146 ARE
TRADEMARKS OF BAXTER INTERNATIONAL INC
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
US PAT NOS 4340049 4346703
4439188 4573980
Low Calcium 1.5% Dextrose

Carton Label
5B9766
6-2000ML IN 2000ML
ULTRABAGTM CONT
1.5%
DIANEAL® LOW CALCIUM 1.5% DEX
PERITONEAL DIALYSIS SOLUTION
EXP
XXXXX
SECONDARY BAR CODE
(17) YYMM00 (10) XXXXX
LOT
XXXXX
PRIMARY BAR CODE
(01) 50309410424525
| DIANEAL LOW CALCIUM WITH DEXTROSE
dextrose monohydrate, sodium chloride, sodium lactate, calcium chloride, and magnesium chloride injection, solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| DIANEAL LOW CALCIUM WITH DEXTROSE
dextrose monohydrate, sodium chloride, sodium lactate, calcium chloride, and magnesium chloride injection, solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| DIANEAL LOW CALCIUM WITH DEXTROSE
dextrose monohydrate, sodium chloride, sodium lactate, calcium chloride, and magnesium chloride injection, solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Baxter Healthcare Corporation (005083209) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Healthcare Corporation | 059140764 | ANALYSIS(0941-0424, 0941-0430, 0941-0433) , LABEL(0941-0424, 0941-0430, 0941-0433) , MANUFACTURE(0941-0424, 0941-0430, 0941-0433) , PACK(0941-0424, 0941-0430, 0941-0433) , STERILIZE(0941-0424, 0941-0430, 0941-0433) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Baxter Healthcare Corporation | 194684502 | ANALYSIS(0941-0424, 0941-0430, 0941-0433) | |
More about lvp solution
Patient resources
Professional resources
- Dextrose and Sodium Chloride Injection prescribing information
- Dextrose in Lactated Ringer's (FDA)
- Dextrose in Ringer's (FDA)
Other brands
Normosol-R, Extraneal, Isolyte S, Isolyte S PH 7.4, ... +2 more
