Dextrose Injection Prescribing Information
Package insert / product label
Generic name: dextrose monohydrate
Dosage form: injection, solution
Drug class: Glucose elevating agents
Medically reviewed by Drugs.com. Last updated on Aug 2, 2024.
On This Page
NOTE: This solution is hypertonic - See WARNINGS and PRECAUTIONS.
LifeShield® Abboject® Syringe
Abboject® Syringe
Fliptop Container
Ansyr® II Plastic Syringe
Rx only
Dextrose Injection Description
50% Dextrose Injection, USP is a sterile, nonpyrogenic, hypertonic solution of dextrose in water for injection for intravenous injection as a fluid and nutrient replenisher.
Each mL of fluid contains 0.5 g dextrose, hydrous which delivers 3.4 kcal/gram. The solution has an osmolarity of 2.53 mOsmol/mL (calc.), pH 3.2 to 6.5 and may contain sodium hydroxide and/or hydrochloric acid for pH adjustment.
The solution contains no bacteriostat, antimicrobial agent or added buffer (except for pH adjustment) and is intended only for use as a single-dose injection. When smaller doses are required, the unused portion should be discarded with the entire unit.
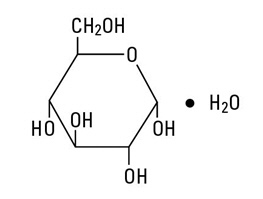
Dextrose, USP is chemically designated C6H12O6 ∙ H2O (D-glucose monohydrate), a hexose sugar freely soluble in water.
Dextrose, hydrous has the following structural formula:
Water for Injection, USP is chemically designated H2O.
The syringe is molded from a specially formulated polypropylene. Water permeates from inside the container at an extremely slow rate which will have an insignificant effect on solution concentration over the expected shelf life. Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the syringe material.
Related/similar drugs
acetylcysteine, ascorbic acid, biotin, multivitamin, niacin
Dextrose Injection - Clinical Pharmacology
When administered intravenously this solution restores blood glucose levels in hypoglycemia and provides a source of carbohydrate calories.
Carbohydrate in the form of dextrose may aid in minimizing liver glycogen depletion and exerts a protein-sparing action. Dextrose injection undergoes oxidation to carbon dioxide and water.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult requirement ranges from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na+) plays a major role in maintaining physiologic equilibrium.
Indications and Usage for Dextrose Injection
50% Dextrose Injection is indicated in the treatment of insulin hypoglycemia (hyperinsulinemia or insulin shock) to restore blood glucose levels.
The solution is also indicated, after dilution, for intravenous infusion as a source of carbohydrate calories in patients whose oral intake is restricted or inadequate to maintain nutritional requirements. Slow infusion of hypertonic solutions is essential to ensure proper utilization of dextrose and avoid production of hyperglycemia.
Contraindications
A concentrated dextrose solution should not be used when intracranial or intraspinal hemorrhage is present, nor in the presence of delirium tremens if the patient is already dehydrated.
Dextrose injection without electrolytes should not be administered simultaneously with blood through the same infusion set because of the possibility that pseudoagglutination of red cells may occur.
Warnings
50% Dextrose Injection is hypertonic and may cause phlebitis and thrombosis at the site of injection.
Significant hyperglycemia and possible hyperosmolar syndrome may result from too rapid administration. The physician should be aware of the symptoms of hyperosmolar syndrome, such as mental confusion and loss of consciousness, especially in patients with chronic uremia and those with known carbohydrate intolerance.
The intravenous administration of this solution can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, overhydration, congested states or pulmonary edema.
Additives may be incompatible. Consult with pharmacist if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
Precautions
Do not use unless the solution is clear and seal is intact. Discard unused portion.
Electrolyte deficits, particularly in serum potassium and phosphate, may occur during prolonged use of concentrated dextrose solutions. Blood electrolyte monitoring is essential and fluid and electrolyte imbalances should be corrected. Essential vitamins and minerals also should be provided as needed.
To minimize hyperglycemia and consequent glycosuria, it is desirable to monitor blood and urine glucose and if necessary, add insulin.
When a concentrated dextrose infusion is abruptly withdrawn, it is advisable to follow with the administration of 5% or 10% dextrose injection to avoid rebound hypoglycemia.
Solutions containing dextrose should be used with caution in patients with known subclinical or overt diabetes mellitus.
Care should be exercised to ensure that the needle is well within the lumen of the vein and that extravasation does not occur. If thrombosis should occur during administration, the injection should be stopped and corrective measures instituted.
Concentrated dextrose solutions should not be administered subcutaneously or intramuscularly.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Studies with solutions in polypropylene syringes have not been performed to evaluate carcinogenic potential, mutagenic potential or effects on fertility.
Adverse Reactions/Side Effects
Hyperosmolar syndrome, resulting from excessively rapid administration of concentrated dextrose may cause mental confusion and/or loss of consciousness.
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
Overdosage
In the event of overhydration or solute overload during therapy, re-evaluate the patient and institute appropriate corrective measures. See WARNINGS and PRECAUTIONS.
Dextrose Injection Dosage and Administration
For peripheral vein administration:
Injection of the solution should be made slowly.
The maximum rate at which dextrose can be infused without producing glycosuria is 0.5 g/kg of body weight/hour. About 95% of the dextrose is retained when infused at a rate of 0.8 g/kg/hr.
In insulin-induced hypoglycemia, intravenous injection of 10 to 25 grams of dextrose (20 to 50 mL of 50% dextrose) is usually adequate. Repeated doses and supportive treatment may be required in severe cases. A specimen for blood glucose determination should be taken before injecting the dextrose. In such emergencies, dextrose should be administered promptly without awaiting pretreatment test results.
For central venous administration:
For total parenteral nutrition 50% Dextrose Injection, USP is administered by slow intravenous infusion (a) after admixture with amino acid solutions via an indwelling catheter with the tip positioned in a large central vein, preferably the superior vena cava, or (b) after dilution with sterile water for injection. Dosage should be adjusted to meet individual patient requirements.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such evaluation.
The maximum rate of dextrose administration which does not result in glycosuria is the same as cited above.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. See CONTRAINDICATIONS.
How is Dextrose Injection supplied
50% Dextrose Injection, USP is supplied in single-dose containers as follows:
| Unit of Sale and Product Description | Strength
(Concentration) | NDC |
|---|---|---|
|
Bundle of 10 |
25 g/50 mL |
0409-4902-34 |
|
Bundle of 10 |
25 g/50 mL |
0409-0505-25 |
|
Tray of 25 |
25 g/50 mL |
0409-6648-02 |
|
Bundle of 10 |
25 g/50 mL |
0409-7517-16 |
Abboject® is a trademark of Abbott Laboratories.
LifeShield® is the trademark of ICU Medical, Inc. and is used under license.
For Medical Information about 50% Dextrose Injection, please visit www.pfizermedinfo.com or call 1‑800-438-1985.
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
LAB-1027-7.0
Revised: July 2024
PRINCIPAL DISPLAY PANEL - 50 mL Syringe Label - 0409-7517-66
50% DEXTROSE
50 mL Single-dose
Rx only
NDC 0409-7517-66
50% DEXTROSE Injection, USP
25 g/50 mL (0.5 g/mL)
For intravenous use. Usual dosage: See insert. Sterile, nonpyrogenic.
2.53 mOsmol/mL (calc.). pH 4.2 (3.2 to 6.5)
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
RL-7117
LOT ##-###-AA
EXP DMMMYYYY
PRINCIPAL DISPLAY PANEL - 50 mL Syringe Carton - 0409-7517-66
50 mL
NDC 0409-7517-66
50%
DEXTROSE
Injection, USP
25 g/50 mL (0.5 g/mL)
Ansyr®II
Unit of Use Syringe
Rx only
LOT #####AA
EXP DMMMYYYY
Hospira
◀ PRESS AND PULL TO OPEN ▶
PRINCIPAL DISPLAY PANEL - 50 mL Syringe Label - 0409-4902-64
50% DEXTROSE
50 mL Single-dose
Rx only
NDC 0409-4902-64
50% DEXTROSE Inj., USP
25 g/50 mL (0.5 g/mL)
For intravenous use. Usual dosage: See insert.
Sterile, nonpyrogenic. 2.53 mOsmol/mL (calc.) pH 4.2 (3.2 to 6.5)
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
RL–7116
LOT ##–###–AA
EXP DMMMYYYY
PRINCIPAL DISPLAY PANEL - 50 mL Syringe Carton - 0409-4902-64
50 mL
NDC 0409-4902-64
50%
DEXTROSE
Injection, USP
25 g/50 mL
(0.5 g/mL)
LIFESHIELD®
Glass
ABBOJECT®
Unit of Use Syringe
with male luer lock
adapter and
18-Gauge protected needle
Rx only
Hospira
LOT #####AA
EXP DMMMYYYY
◀ PRESS AND PULL TO OPEN
PRINCIPAL DISPLAY PANEL - 50 mL Vial Label - 0409-6648-16
50 mL Single-dose
50% Dextrose
Injection, USP
25 grams/50 mL
(0.5 g/mL)
Contains no more than 600 mcg/L of
aluminum.
Distributed by Hospira, Inc.
Lake Forest, IL 60045 USA
Hospira
PRINCIPAL DISPLAY PANEL - 50 mL Vial Tray - 0409-6648-02
50 mL Single-dose Fliptop Vial
Rx only
NDC 0409-6648-02
Contains 25 of NDC 0409-6648-16
50% Dextrose Injection, USP
25 grams/50 mL
(0.5 g/mL)
Hospira
PRINCIPAL DISPLAY PANEL - 50 mL Syringe Label - 0409-0505-15
50 mL Single-Dose Syringe
Rx only
NDC 0409-0505-15
50% DEXTROSE Inj., USP
25 g/50 mL (0.5 g/mL)
For intravenous use. Recommended Dosage: See Prescribing
Information.
Sterile, nonpyrogenic. 2.53 mOsmol/mL (calc.) pH 3.2 to 6.5
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
Hospira
| DEXTROSE
dextrose monohydrate injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| DEXTROSE
dextrose monohydrate injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| DEXTROSE
dextrose monohydrate injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| DEXTROSE
dextrose monohydrate injection, solution |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Hospira, Inc. (141588017) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 093132819 | ANALYSIS(0409-7517, 0409-4902, 0409-6648, 0409-0505) , MANUFACTURE(0409-7517, 0409-4902, 0409-6648, 0409-0505) , PACK(0409-7517, 0409-4902, 0409-6648, 0409-0505) , LABEL(0409-7517, 0409-4902, 0409-6648, 0409-0505) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira, Inc. | 827731089 | ANALYSIS(0409-7517, 0409-4902, 0409-6648, 0409-0505) | |
Frequently asked questions
More about glucose
- Check interactions
- Compare alternatives
- Reviews (3)
- Drug images
- Side effects
- Support group
- Drug class: glucose elevating agents
- En español
Patient resources
Professional resources
- Dextrose 50% Injection prescribing information
- Dextrose Injection 10% (FDA)
- Dextrose Injection 70% (FDA)
- Dextrose Injection Concentrated (FDA)
- Dextrose Injection Infants (FDA)