The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
Correctol
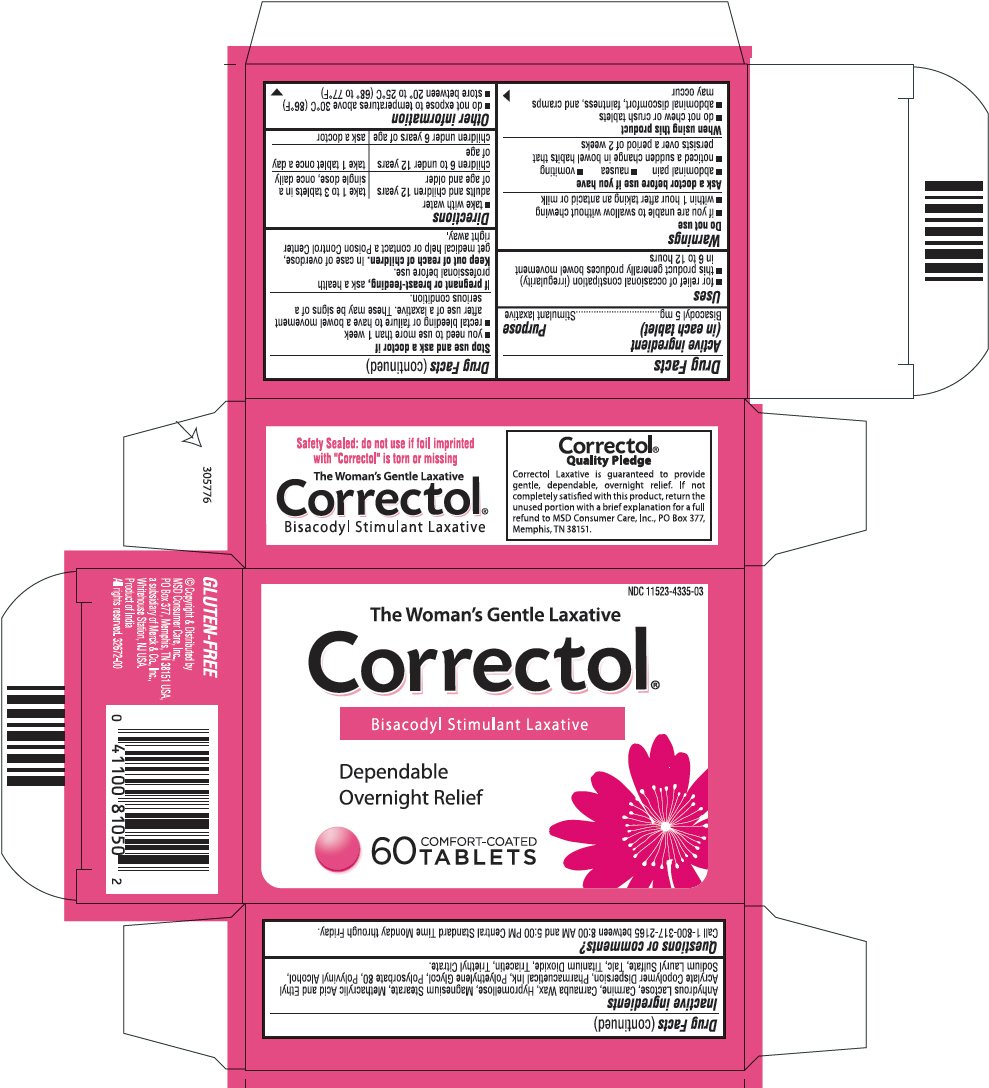
Dosage form: tablet, coated
Ingredients: Bisacodyl 5mg
Labeler: MSD Consumer Care, Inc.
NDC code: 11523-4335
Drug Facts
Bisacodyl 5mg
Stimulant Laxative
- for relief of occasional constipation (irregularity)
- this product generally produces bowel movement in 6 to 12
- if you are unable to swallow without chewing
- within 1 hour after taking antacid or milk
- abdominal pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that persists over a period of 2 weeks
- do not chew or crush tablets
- abdominal discomfort, faintness, and cramps may occur
- you need to use more than 1 week
- rectal bleeding or failure to have a bowel movement after use of a laxative. These may be signs of a serious condition
If pregnant or breast-feeding, ask a health professional before use
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away
- take with water
| adults and children 12 years of age and older | Take 1 to 3 tablets in a Single dose, once daily |
| children 6 to under 12 years of age | Take 1 tablet once a day |
| children under 6 years of age | ask a doctor |
- do not expose to temperatures above 30°F
- store between 20 to 25°C (68 to 77°F)
Anhydrous Lactose, Carmine, Carnauba wax, Hypromellose, Magnesium stearate, Methacrylic Acid and Ethyl Acrylate Copolymer Dispersion, Pharmaceutical Ink, Polyethylene Glycol, Polysorbate 80, Polyvinyl Alcohol, Sodium Lauryl Sulfate, Talc, Titanium Dioxide, Triacetin, Triethyl Citrate
Call 1-800-317-2165 between 8:00 AM and 5:00 PM Central Standard Time Monday through Friday.
Distributed by
MSD Consumer Care, Inc.,
PO Box 377, Memphis, TN 38151 USA,
a subsidiary of Merck & Co., Inc.,
Whitehouse Station, NJ USA.
| CORRECTOL
bisacodyl tablet, coated |
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| Labeler - MSD Consumer Care, Inc. (968091715) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Rottendorf Pharma INC. (Germany) | 315974691 | MANUFACTURE(11523-4335) | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.