Niacin Tablets Prescribing Information
Package insert / product label
Dosage form: tablet
Drug classes: Miscellaneous antihyperlipidemic agents, Vitamins
Medically reviewed by Drugs.com. Last updated on Feb 1, 2024.
On This Page
Niacin Tablets Description
Niacin or nicotinic acid, a water-soluble B‑complex vitamin and antihyperlipidemic agent, is 3‑pyridinecarboxylic acid. It is a white, crystalline powder, sparingly soluble in water. It has the following structural formula:
| MW=123.11 | 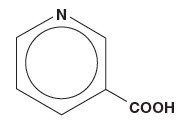 | C6H5NO2 |
Each Niacin Tablet, for oral administration, contains 500 mg of nicotinic acid. In addition, each tablet contains the following inactive ingredients: croscarmellose sodium, hydrogenated vegetable oil, magnesium stearate and microcrystalline cellulose.
Niacin Tablets - Clinical Pharmacology
The role of low-density lipoprotein (LDL) cholesterol in atherogenesis is supported by pathological observations, clinical studies, and many animal experiments. Observational epidemiological studies have clearly established that high total or LDL cholesterol and low high-density lipoprotein (HDL) cholesterol are risk factors for coronary heart disease. The Coronary Drug Project1, completed in 1975, was designed to assess the safety and efficacy of nicotinic acid and other lipid-altering drugs in men 30 to 64 years old with a history of myocardial infarction (MI). Over an observation period of five years, nicotinic acid showed a statistically significant benefit in decreasing nonfatal, recurrent myocardial infarctions. The incidence of definite, nonfatal MI was 8.9% for the 1,119 patients randomized to nicotinic acid versus 12.2% for the 2,789 patients who received placebo (p< 0.004). Though total mortality was similar in the two groups at five years (24.4% with nicotinic acid versus 25.4% with placebo; p=N.S.), in a fifteen year cumulative follow-up there were 11% (69) fewer deaths in the nicotinic acid group compared to the placebo cohort (52.0% versus 58.2%; p=0.0004)2.
The Cholesterol-Lowering Atherosclerosis Study (CLAS) was a randomized, placebo-controlled, angiographic trial testing combined colestipol and nicotinic acid therapy in 162 non-smoking males with previous coronary bypass surgery3. The primary, per subject cardiac endpoint was global coronary artery change score. After two years, 61% of patients in the placebo cohort showed disease progression by global change score (N=82), compared with only 38.8% of drug-treated subjects (N=80), when both native arteries and grafts were considered (p<0.005). In a follow-up to this trial in a subgroup of 103 patients treated for four years, again, significantly fewer patients in the drug-treated group demonstrated progression than in the placebo cohort (48% versus 85%, respectively; p< 0.0001)4.
The Familial Atherosclerosis Treatment Study (FATS) in 146 men ages 62 and younger with apolipoprotein B levels ≥125 mg/dL, established coronary artery disease, and family histories of vascular disease, assessed change in severity of disease in the proximal coronary arteries by quantitative arteriography5. Patients were given dietary counseling and randomized to treatment with either conventional therapy with double placebo (or placebo plus colestipol if the LDL cholesterol was elevated); lovastatin plus colestipol; or nicotinic acid plus colestipol. In the conventional therapy group, 46% of patients had disease progression (and no regression) in at least one of nine proximal coronary segments. In contrast, progression (as the only change) was seen in only 25% in the nicotinic acid plus colestipol group. Though not an original endpoint of the trial, clinical events (death, myocardial infarction, or revascularization for worsening angina) occurred in 10 of 52 patients who received conventional therapy, compared with 2 of 48 who received nicotinic acid plus colestipol.
Nicotinic acid (but not nicotinamide) in gram doses produces an average 10% to 20% reduction in total and LDL cholesterol, a 30% to 70% reduction in triglycerides, and an average 20% to 35% increase in HDL cholesterol. The magnitude of individual lipid and lipoprotein responses may be influenced by the severity and type of underlying lipid abnormality. The increase in total HDL is associated with a shift in the distribution of HDL sub-fractions (as defined by ultra-centrifugation) with an increase in the HDL2:HDL3 ratio and an increase in apolipoprotein A‑I content. The mechanism by which nicotinic acid exerts these effects is not entirely understood, but may involve several actions, including a decrease in esterification of hepatic triglycerides. Nicotinic acid treatment also decreases the serum levels of apolipoprotein B‑100 (apo B), the major protein component of the very low-density lipoprotein (VLDL) and LDL fractions, and of lipoprotein a [Lp(a)], a variant form of LDL independently associated with coronary risk. The effect of nicotinic acid-induced changes in lipids/lipoproteins on cardiovascular morbidity or mortality in individuals without pre-existing coronary disease has not been established.
Pharmacokinetics
Following an oral dose, the pharmacokinetic profile of nicotinic acid is characterized by rapid absorption from the gastrointestinal tract and a short plasma elimination half-life. At a 1 gram dose, peak plasma concentrations of 15 to 30 µg/mL are reached within 30 to 60 minutes. Approximately 88% of an oral pharmacologic dose is eliminated by the kidneys as unchanged drug and nicotinuric acid, its primary metabolite. The plasma elimination half-life of nicotinic acid ranges from 20 to 45 minutes.
Indications and Usage for Niacin Tablets
I. Therapy with lipid-altering agents should be only one component of multiple risk factor intervention in those individuals at significantly increased risk for atherosclerotic vascular disease due to hypercholesterolemia. Nicotinic acid, alone or in combination with a bile-acid binding resin, is indicated as an adjunct to diet for the reduction of elevated total and LDL cholesterol levels in patients with primary hypercholesterolemia (Types IIa and IIb)†, when the response to a diet restricted in saturated fat and cholesterol and other nonpharmacologic measures alone has been inadequate (see also the NCEP treatment guidelines6). Prior to initiating therapy with nicotinic acid, secondary causes for hypercholesterolemia (e.g., poorly controlled diabetes mellitus, hypothyroidism, nephrotic syndrome, dysproteinemias, obstructive liver disease, other drug therapy, alcoholism) should be excluded, and a lipid profile performed to measure total cholesterol, HDL cholesterol, and triglycerides.
II. Nicotinic acid is also indicated as adjunctive therapy for the treatment of adult patients with very high serum triglyceride levels (Types IV and V hyperlipidemia)† who present a risk of pancreatitis and who do not respond adequately to a determined dietary effort to control them. Such patients typically have serum triglyceride levels over 2,000 mg/dL and have elevations of VLDL cholesterol as well as fasting chylomicrons (Type V hyperlipidemia)†. Subjects who consistently have total serum or plasma triglycerides below 1,000 mg/dL are unlikely to develop pancreatitis. Therapy with nicotinic acid may be considered for those subjects with triglyceride elevations between 1,000 and 2,000 mg/dL who have a history of pancreatitis or of recurrent abdominal pain typical of pancreatitis. Some Type IV patients with triglycerides under 1,000 mg/dL may, through dietary or alcoholic indiscretion, convert to a Type V pattern with massive triglyceride elevations accompanying fasting chylomicronemia, but the influence of nicotinic acid therapy on the risk of pancreatitis in such situations has not been adequately studied. Drug therapy is not indicated for patients with Type I hyperlipoproteinemia, who have elevations of chylomicrons and plasma triglycerides, but who have normal levels of VLDL. Inspection of plasma refrigerated for 14 hours is helpful in distinguishing Types I, IV, and V hyperlipoproteinemia7.
| Lipoproteins | Lipid Elevations | ||
|---|---|---|---|
| Type | Elevated | Major | Minor |
| C = cholesterol, TG = triglycerides LDL = low-density lipoprotein VLDL = very low-density lipoprotein IDL = intermediate-density lipoprotein |
|||
| I (rare) | Chylomicrons | TG | ↑→ C |
| IIa | LDL | C | ..... |
| IIb | LDL, VLDL | C | TG |
| III (rare) | IDL | C/TG | ..... |
| IV | VLDL | TG | ↑→ C |
| V (rare) | Chylomicrons, VLDL | TG | ↑→ C |
Contraindications
Nicotinic acid is contraindicated in patients with a known hypersensitivity to any component of this medication; significant or unexplained hepatic dysfunction; active peptic ulcer disease; or arterial bleeding.
Warnings
Liver Dysfunction
Cases of severe hepatic toxicity, including fulminant hepatic necrosis have occurred in patients who have substituted sustained-release (modified-release, timed-release) nicotinic acid products for immediate-release (crystalline) nicotinic acid at equivalent doses.
Liver function tests should be performed on all patients during therapy with nicotinic acid. Serum transaminase levels, including ALT (SGPT), should be monitored before treatment begins, every six weeks to twelve weeks for the first year, and periodically thereafter (e.g., at approximately 6 month intervals). Special attention should be paid to patients who develop elevated serum transaminase levels, and in these patients, measurements should be repeated promptly and then performed more frequently. If the transaminase levels show evidence of progression, particularly if they rise to three times the upper limit of normal and are persistent, the drug should be discontinued. Liver biopsy should be considered if elevations persist beyond discontinuation of the drug.
Nicotinic acid should be used with caution in patients who consume substantial quantities of alcohol and/or have a past history of liver disease. Active liver diseases or unexplained transaminase elevations are contraindications to the use of nicotinic acid.
Skeletal Muscle
Rare cases of rhabdomyolysis have been associated with concomitant administration of lipid-altering doses (≥1 g/day) of nicotinic acid and HMG‑CoA reductase inhibitors. Physicians contemplating combined therapy with HMG‑CoA reductase inhibitors and nicotinic acid should carefully weigh the potential benefits and risks and should carefully monitor patients for any signs and symptoms of muscle pain, tenderness, or weakness, particularly during the initial months of therapy and during any periods of upward dosage titration of either drug. Periodic serum creatine phosphokinase (CPK) and potassium determinations should be considered in such situations, but there is no assurance that such monitoring will prevent the occurrence of severe myopathy.
Precautions
General
Before instituting therapy with nicotinic acid, an attempt should be made to control hyperlipidemia with appropriate diet, exercise, and weight reduction in obese patients, and to treat other underlying medical problems [see INDICATIONS AND USAGE ].
Patients with a past history of jaundice, hepatobiliary disease, or peptic ulcer should be observed closely during nicotinic acid therapy. Frequent monitoring of liver function tests and blood glucose should be performed to ascertain that the drug is producing no adverse effects on these organ systems. Diabetic patients may experience a dose-related rise in glucose intolerance, the clinical significance of which is unclear. Diabetic or potentially diabetic patients should be observed closely. Adjustment of diet and/or hypoglycemic therapy may be necessary.
Caution should also be used when nicotinic acid is used in patients with unstable angina or in the acute phase of myocardial infarction, particularly when such patients are also receiving vasoactive drugs such as nitrates, calcium channel blockers, or adrenergic blocking agents.
Elevated uric acid levels have occurred with nicotinic acid therapy, therefore use with caution in patients predisposed to gout.
Drug Interactions
Antihypertensive Therapy
Nicotinic acid may potentiate the effects of ganglionic blocking agents and vasoactive drugs resulting in postural hypotension.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Nicotinic acid administered to mice for a lifetime as a 1% solution in drinking water was not carcinogenic. The mice in this study received approximately 6 to 8 times a human dose of 3,000 milligrams/day as determined on a milligram/square meter basis. Nicotinic acid was negative for mutagenicity in the Ames test. No studies on impairment of fertility have been performed.
Pregnancy
Pregnancy Category C
Animal reproduction studies have not been conducted with nicotinic acid. It is also not known whether nicotinic acid at doses typically used for lipid disorders can cause fetal harm when administered to pregnant women or whether it can affect reproductive capacity. If a woman receiving nicotinic acid for primary hypercholesterolemia (Types IIa or IIb) becomes pregnant, the drug should be discontinued. If a woman being treated with nicotinic acid for hypertriglyceridemia (Types IV or V) conceives, the benefits and risks of continued drug therapy should be assessed on an individual basis.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from lipid-altering doses of nicotinic acid, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Adverse Reactions/Side Effects
Cardiovascular: Atrial fibrillation and other cardiac arrhythmias, orthostasis, hypotension.
Gastrointestinal: Dyspepsia, vomiting, diarrhea, peptic ulceration, jaundice, abnormal liver function tests.
Skin: Mild to severe cutaneous flushing, pruritus, hyperpigmentation, acanthosis nigricans, dry skin.
Metabolic: Decreased glucose tolerance, hyperuricemia, gout.
Eye: Toxic amblyopia, cystoid macular edema.
Nervous System / Psychiatric: Headache.
Niacin Tablets Dosage and Administration
The usual adult dosage of nicotinic acid is 1 to 2 grams two or three times a day. Doses should be individualized according to the patient's response. Start with one-half tablet (250 mg) as a single daily dose following the evening meal. The frequency of dosing and total daily dose can be increased every four to seven days until the desired LDL cholesterol and/or triglyceride level is achieved or the first-level therapeutic dose of 1.5 to 2 grams/day is reached. If the patient's hyperlipidemia is not adequately controlled after 2 months at this level, the dosage can then be increased at two to four week intervals to 3 grams/day (1 gram three times per day). In patients with marked lipid abnormalities, a higher dose is occasionally required, but generally should not exceed 6 grams/day.
Flushing of the skin appears frequently and can be minimized by pretreatment with aspirin or non-steroidal anti-inflammatory drugs. Tolerance to this flushing develops rapidly over the course of several weeks. Flushing, pruritus, and gastrointestinal distress are also greatly reduced by slowly increasing the dose of nicotinic acid and avoiding administration on an empty stomach.
Sustained-release (modified-release, timed-release) nicotinic acid preparations should not be substituted for equivalent doses of immediate-release (crystalline) nicotinic acid.
How is Niacin Tablets supplied
Niacin Tablets, USP 500 mg.
Each tablet is a white, capsule-shaped, scored, uncoated tablet, debossed "US" to the left and "67" to the right of the score, with "500" strength on the unscored side.
Niacin is available in bottles of 90 tablets (NDC 71741-047-90).
References
- The Coronary Drug Project Research Group. Clofibrate and Niacin in Coronary Heart Disease. JAMA 1975; 231:360–81.
- Canner PL et al. Fifteen Year Mortality in Coronary Drug Project Patients: Long-Term Benefit with Niacin. JACC 1986; 8(6):1245–55.
- Blankenhorn DH et al. Beneficial Effects of Combined Colestipol-Niacin Therapy on Coronary Atherosclerosis and Coronary Venous Bypass Grafts. JAMA 1987; 257(23):3233–40.
- Cashin-Hemphill et al. Beneficial Effects of Colestipol-Niacin on Coronary Atherosclerosis. JAMA 1990; 264(23):3013–17.
- Brown G et al. Regression of Coronary Artery Disease as a Result of Intensive Lipid-Lowering Therapy in Men with High Levels of Apolipoprotein B. NEJM 1990; 323:1289–98.
- Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol. Arch Int Med 1988; 148:36–69.
- Nikkila EA: Familial lipoprotein lipase deficiency and related disorders of chylomicron metabolism. In Stanbury JB et al. (eds.): The Metabolic Basis of Inherited Disease, 5th ed., McGraw-Hill, 1983, Chap. 30, pp. 622–642.
| NIACIN
niacin tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Redmont Pharmaceuticals, LLC (080843607) |
Frequently asked questions
More about niacin
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (116)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Support group
- Drug class: miscellaneous antihyperlipidemic agents
Patient resources
- Niacin drug information
- Niacin Capsules and Tablets
- Niacin Controlled-Release Capsules and Controlled-Release Tablets


