Losartan Potassium and Hydrochlorothiazide Prescribing Information
Package insert / product label
Dosage form: tablet
Drug class: Angiotensin II inhibitors with thiazides
Medically reviewed by Drugs.com. Last updated on Jan 2, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
LOSARTAN POTASSIUM and HYDROCHLOROTHIAZIDE tablets, for oral use
Initial U.S. Approval: 1995
WARNING: FETAL TOXICITY
See full prescribing information for complete boxed warning.
When pregnancy is detected, discontinue losartan potassium and hydrochlorothiazide tablets as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. (5.1)
Indications and Usage for Losartan Potassium and Hydrochlorothiazide
Losartan potassium and hydrochlorothiazide tablets are a combination of losartan, an angiotensin II receptor blocker (ARB) and hydrochlorothiazide, a diuretic indicated for:
- Treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. (1.1)
- Reduction of the risk of stroke in patients with hypertension and left ventricular hypertrophy. There is evidence that this benefit does not apply to Black patients. (1.2)
Losartan Potassium and Hydrochlorothiazide Dosage and Administration
Dosage Forms and Strengths
Tablets (losartan potassium/hydrochlorothiazide content): 50/12.5 mg; 100/12.5 mg; and 100/25 mg. (3)
Contraindications
Warnings and Precautions
- Hypotension: Correct volume or salt depletion prior to administration of losartan potassium and hydrochlorothiazide tablets. (5.2)
- Monitor renal function and potassium in susceptible patients. (5.3)
- Observe for clinical signs of fluid or electrolyte imbalance. (5.5)
- Acute angle-closure glaucoma. (5.6)
- Exacerbation of systemic lupus erythematosus. (5.7)
Adverse Reactions/Side Effects
Most common adverse reactions (incidence ≥2% and greater than placebo) are dizziness, upper respiratory infection, cough, and back pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Teva at 1-888-838-2872 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- Agents increasing serum potassium: Risk of hyperkalemia. (7.1)
- Lithium: Risk of lithium toxicity. (7.2)
- Non-Steroidal Anti-Inflammatory Drugs (NSAIDs): increased risk of renal impairment and reduced diuretic, natriuretic, and antihypertensive effects. (7.3)
- Dual inhibition of the renin-angiotensin system: increased risk of renal impairment, hypotension, syncope, and hyperkalemia. (7.4)
- Antidiabetic drugs: dosage adjustment of antidiabetic may be required. (7.5)
- Cholestyramine and colestipol: Reduced absorption of thiazides. (7.5)
Use In Specific Populations
- Hepatic Impairment: Losartan potassium and hydrochlorothiazide tablets are not recommended for initial therapy, because the recommended starting dose is not available. (8.7)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 11/2023
Related/similar drugs
amlodipine, lisinopril, metoprolol, losartan, furosemide, hydrochlorothiazide
Full Prescribing Information
WARNING: FETAL TOXICITY
When pregnancy is detected, discontinue losartan potassium and hydrochlorothiazide tablets as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus [see Warnings and Precautions (5.1)].
1. Indications and Usage for Losartan Potassium and Hydrochlorothiazide
1.1 Hypertension
Losartan potassium and hydrochlorothiazide tablets are indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure lowers the risk of fatal and non-fatal cardiovascular (CV) events, primarily strokes and myocardial infarction. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including losartan and hydrochlorothiazide.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than 1 drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in Black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
This fixed dose combination is not indicated for initial therapy of hypertension, except when the hypertension is severe enough that the value of achieving prompt blood pressure control exceeds the risk of initiating combination therapy in these patients [see Clinical Studies (14) and Dosage and Administration (2.1)].
Losartan potassium and hydrochlorothiazide tablets may be administered with other antihypertensive agents.
1.2 Hypertensive Patients with Left Ventricular Hypertrophy
Losartan potassium and hydrochlorothiazide tablets are indicated to reduce the risk of stroke in patients with hypertension and left ventricular hypertrophy, but there is evidence that this benefit does not apply to Black patients [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3), and Dosage and Administration (2.2)].
2. Losartan Potassium and Hydrochlorothiazide Dosage and Administration
2.1 Hypertension
The usual starting dose of losartan potassium and hydrochlorothiazide tablets is 50/12.5 (losartan 50 mg/hydrochlorothiazide 12.5 mg) once daily. The dosage can be increased after 3 weeks of therapy to a maximum of 100/25 (losartan 100 mg/hydrochlorothiazide 25 mg) once daily as needed to control blood pressure [see Clinical Studies (14.2)].
Initiate a patient whose blood pressure is not adequately controlled with losartan 50 mg monotherapy with losartan potassium and hydrochlorothiazide tablets 50/12.5 once daily. If blood pressure remains uncontrolled after about 3 weeks of therapy, the dosage may be increased to two tablets of losartan potassium and hydrochlorothiazide tablets 50/12.5 once daily or one tablet of losartan potassium and hydrochlorothiazide tablets 100/25 once daily.
Initiate a patient whose blood pressure is not adequately controlled with losartan 100 mg monotherapy with losartan potassium and hydrochlorothiazide tablets 100/12.5 (losartan 100 mg/hydrochlorothiazide 12.5 mg) once daily. If blood pressure remains uncontrolled after about 3 weeks of therapy, increase the dose to two tablets of losartan potassium and hydrochlorothiazide tablets 50/12.5 once daily or one tablet of losartan potassium and hydrochlorothiazide tablets 100/25 once daily.
Initiate a patient whose blood pressure is inadequately controlled with hydrochlorothiazide 25 mg once daily, or is controlled but who experiences hypokalemia with this regimen, on losartan potassium and hydrochlorothiazide tablets 50/12.5 once daily, reducing the dose of hydrochlorothiazide without reducing the overall expected antihypertensive response. Evaluate the clinical response to losartan potassium and hydrochlorothiazide tablets 50/12.5 and, if blood pressure remains uncontrolled after about 3 weeks of therapy, increase the dose to two tablets of losartan potassium and hydrochlorothiazide tablets 50/12.5 once daily or one tablet of losartan potassium and hydrochlorothiazide tablets 100/25 once daily.
2.2 Hypertensive Patients with Left Ventricular Hypertrophy
In patients whose blood pressure is not adequately controlled on 50 mg losartan potassium, initiate treatment with losartan potassium and hydrochlorothiazide tablets 50/12.5. If additional blood pressure reduction is needed, increase the dose to losartan potassium and hydrochlorothiazide tablets 100/12.5, followed by losartan potassium and hydrochlorothiazide tablets 100/25. For further blood pressure reduction add other antihypertensives [see Clinical Studies (14)].
3. Dosage Forms and Strengths
- 50 mg/12.5 mg - Yellow, oval-shaped, film-coated tablet, debossed with “93” on one side and “7367” on the other.
- 100 mg/12.5 mg - White to off-white, oval-shaped, film-coated tablet, debossed with “93” on one side and “7369” on the other.
- 100 mg/25 mg - Light yellow, oval-shaped, film-coated tablet, debossed with “93” on one side and “7368” on the other.
4. Contraindications
Losartan potassium and hydrochlorothiazide tablets are contraindicated:
- In patients who are hypersensitive to any component of this product.
- In patients with anuria
- For coadministration with aliskiren in patients with diabetes
5. Warnings and Precautions
5.1 Fetal Toxicity
Losartan potassium and hydrochlorothiazide tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue losartan potassium and hydrochlorothiazide tablets as soon as possible.
Thiazides cross the placental barrier and appear in cord blood. Adverse reactions include fetal or neonatal jaundice, thrombocytopenia [see Use in Specific Populations (8.1)].
5.2 Hypotension in Volume- or Salt-Depleted Patients
In patients with an activated renin-angiotensin system, such as volume- or salt-depleted patients (e.g., those being treated with high doses of diuretics), symptomatic hypotension may occur after initiation of treatment with losartan potassium and hydrochlorothiazide tablets. Correct volume or salt depletion prior to administration of losartan potassium and hydrochlorothiazide tablets. Do not use losartan potassium and hydrochlorothiazide tablets as initial therapy in patients with intravascular volume depletion.
5.3 Impaired Renal Function
Changes in renal function including acute renal failure can be caused by drugs that inhibit the renin-angiotensin system and by diuretics. Patients whose renal function may depend in part on the activity of the renin-angiotensin system (e.g., patients with renal artery stenosis, chronic kidney disease, severe congestive heart failure, or volume depletion) may be at particular risk of developing acute renal failure on losartan potassium and hydrochlorothiazide tablets. Monitor renal function periodically in these patients. Consider withholding or discontinuing therapy in patients who develop a clinically significant decrease in renal function on losartan potassium and hydrochlorothiazide tablets [see Drug Interactions (7.3) and Use in Specific Populations (8.8)].
5.4 Hypersensitivity
Hypersensitivity reactions to hydrochlorothiazide may occur in patients with or without a history of allergy or bronchial asthma but are more likely in patients with such a history [see Adverse Reactions (6.2)].
5.5 Electrolyte and Metabolic Effects
In double-blind clinical trials of various doses of losartan potassium and hydrochlorothiazide, the incidence of hypertensive patients who developed hypokalemia (serum potassium <3.5 mEq/L) was 6.7% versus 3.5% for placebo; the incidence of hyperkalemia (serum potassium >5.7 mEq/L) was 0.4% versus 0% for placebo.
Losartan potassium and hydrochlorothiazide tablets contain hydrochlorothiazide which can cause hypokalemia, hyponatremia and hypomagnesemia. Hypomagnesemia can result in hypokalemia which may be difficult to treat despite potassium repletion. Losartan potassium and hydrochlorothiazide tablets also contain losartan which can cause hyperkalemia. Monitor serum electrolytes periodically [see Drug Interactions (7.1)].
Concomitant use of other drugs that may increase serum potassium may lead to hyperkalemia [see Drug Interactions (7.1)].
Hydrochlorothiazide may alter glucose tolerance and raise serum levels of cholesterol and triglycerides.
Hyperuricemia may occur or frank gout may be precipitated in patients receiving thiazide therapy. Because losartan decreases uric acid, losartan in combination with hydrochlorothiazide attenuates the diuretic-induced hyperuricemia.
Hydrochlorothiazide decreases urinary calcium excretion and may cause elevations of serum calcium. Monitor calcium levels.
5.6 Acute Myopia and Secondary Angle-Closure Glaucoma
Hydrochlorothiazide, a sulfonamide, can cause an idiosyncratic reaction, resulting in acute transient myopia and acute angle-closure glaucoma. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma can lead to permanent vision loss. The primary treatment is to discontinue hydrochlorothiazide as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
6. Adverse Reactions/Side Effects
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Losartan potassium-hydrochlorothiazide has been evaluated for safety in 858 patients treated for essential hypertension and 3889 patients treated for hypertension and left ventricular hypertrophy. Most adverse reactions have been mild and transient in nature and have not required discontinuation of therapy. In controlled clinical trials, discontinuation of therapy due to clinical adverse events was required in only 2.8% and 2.3% of patients treated with the combination and placebo, respectively.
In these double-blind controlled clinical trials, adverse reactions occurring in greater than 2% of subjects treated with losartan-hydrochlorothiazide and at a greater rate than placebo were: back pain (2.1% vs 0.6%), dizziness (5.7% vs 2.9%), and upper respiratory infection (6.1% vs 4.6%).
The following additional adverse reactions have been reported in clinical trials with losartan potassium and hydrochlorothiazide tablets and/or the individual components:
Blood and the lymphatic system disorders: Anemia, aplastic anemia, hemolytic anemia, leukopenia, agranulocytosis.
Metabolism and nutrition disorders: Anorexia, hyperglycemia, hyperuricemia, electrolyte imbalance including hyponatremia and hypokalemia.
Psychiatric disorders: Insomnia, restlessness.
Nervous system disorders: Dysgeusia, headache, migraine, paraesthesias.
Eye disorders: Xanthopsia, transient blurred vision.
Cardiac disorders: Palpitation, tachycardia.
Vascular disorders: Dose-related orthostatic effects, necrotizing angiitis (vasculitis, cutaneous vasculitis).
Respiratory, thoracic and mediastinal disorders: Nasal congestion.
Gastrointestinal disorders: Dyspepsia, abdominal pain, gastric irritation, cramping, nausea, vomiting, pancreatitis, sialoadenitis.
Hepato-biliary disorders: Jaundice (intrahepatic cholestatic jaundice).
Skin and subcutaneous tissue disorders: Rash, pruritus, purpura, toxic epidermal necrolysis, urticaria, photosensitivity, cutaneous lupus erythematosus.
Musculoskeletal and connective tissue disorders: Muscle cramps, muscle spasm.
Renal and urinary disorders: Glycosuria, renal dysfunction, interstitial nephritis, renal failure.
Reproductive system and breast disorders: Erectile dysfunction/impotence.
General disorders and administration site conditions: Chest pain, malaise, weakness.
Investigations: Liver function abnormalities.
Cough
Persistent dry cough has been associated with ACE-inhibitor use and in practice can be a cause of discontinuation of ACE-inhibitor therapy. Two prospective, parallel-group, double-blind, randomized, controlled trials were conducted to assess the effects of losartan on the incidence of cough in hypertensive patients who had experienced cough while receiving ACE-inhibitor therapy. Patients who had typical ACE-inhibitor cough when challenged with lisinopril, whose cough disappeared on placebo, were randomized to losartan 50 mg, lisinopril 20 mg, or either placebo (one study, n=97) or 25 mg hydrochlorothiazide (n=135). The double-blind treatment period lasted up to 8 weeks. The incidence of cough is shown in Table 1 below.
|
Study 1* |
HCTZ |
Losartan |
Lisinopril |
|
Cough |
25% |
17% |
69% |
|
Study 2† |
Placebo |
Losartan |
Lisinopril |
|
Cough |
35% |
29% |
62% |
These studies demonstrate that the incidence of cough associated with losartan therapy, in a population that all had cough associated with ACE-inhibitor therapy, is similar to that associated with hydrochlorothiazide or placebo therapy.
Cases of cough, including positive re-challenges, have been reported with the use of losartan in postmarketing experience.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of losartan potassium and hydrochlorothiazide tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency reliably or to establish a causal relationship to drug exposure.
Digestive: Hepatitis has been reported rarely in patients treated with losartan.
Hematologic: Thrombocytopenia.
Hypersensitivity: Angioedema, including swelling of the larynx and glottis, causing airway obstruction and/or swelling of the face, lips, pharynx, and/or tongue. Vasculitis, including Henoch-Schönlein purpura; anaphylactic reactions which can present as respiratory distress (including pneumonitis and pulmonary edema).
Musculoskeletal: Rhabdomyolysis.
Skin: Erythroderma.
Non-melanoma Skin Cancer: Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of >50,000 mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
7. Drug Interactions
7.1 Agents Increasing Serum Potassium
Coadministration of losartan with other drugs that raise serum potassium levels may result in hyperkalemia. Monitor serum potassium in such patients.
7.2 Lithium
Increases in serum lithium concentrations and lithium toxicity have been reported with concomitant use of angiotensin II receptor antagonists or thiazide diuretics. Monitor lithium levels in patients receiving losartan potassium and hydrochlorothiazide tablets and lithium.
7.3 Non-Steroidal Anti-Inflammatory Agents Including Selective Cyclooxygenase-2 Inhibitors
Losartan Potassium
In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors, with angiotensin II receptor antagonists (including losartan) may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving losartan and NSAID therapy.
The antihypertensive effect of angiotensin II receptor antagonists, including losartan, may be attenuated by NSAIDs, including selective COX-2 inhibitors.
Hydrochlorothiazide
The administration of a non-steroidal anti-inflammatory agent including a selective COX-2 inhibitor can reduce the diuretic, natriuretic, and antihypertensive effects of loop, potassium-sparing and thiazide diuretics. Therefore, when losartan potassium and hydrochlorothiazide tablets and non-steroidal anti-inflammatory agents including selective COX-2 inhibitors are used concomitantly, observe closely to determine if the desired effect of the diuretic is obtained.
In patients receiving diuretic therapy, coadministration of NSAIDs with angiotensin receptor blockers, including losartan, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving hydrochlorothiazide, losartan, and NSAID therapy.
7.4 Dual Blockade of the Renin-Angiotensin System (RAS)
Dual blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, syncope, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy.
The Veterans Affairs Nephropathy in Diabetes (VA NEPHRON-D) trial enrolled 1448 patients with type 2 diabetes, elevated urinary-albumin-to-creatinine ratio, and decreased estimated glomerular filtration rate (GFR 30 to 89.9 mL/min), randomized them to lisinopril or placebo on a background of losartan therapy and followed them for a median of 2.2 years. Patients receiving the combination of losartan and lisinopril did not obtain any additional benefit compared to monotherapy for the combined endpoint of decline in GFR, end-stage renal disease, or death, but experienced an increased incidence of hyperkalemia and acute kidney injury compared with the monotherapy group.
Closely monitor blood pressure, renal function, and electrolytes in patients on losartan potassium and hydrochlorothiazide tablets and other agents that affect the RAS.
Do not coadminister aliskiren with losartan potassium and hydrochlorothiazide tablets in patients with diabetes. Avoid use of aliskiren with losartan potassium and hydrochlorothiazide tablets in patients with renal impairment (GFR <60 mL/min).
7.5 The Use of Hydrochlorothiazide with Other Drugs
When administered concurrently, the following drugs may interact with thiazide diuretics [see Clinical Pharmacology (12.3)]:
Antidiabetic drugs (oral agents and insulin) — dosage adjustment of the antidiabetic drug may be required.
Cholestyramine and colestipol resins — Absorption of hydrochlorothiazide is impaired in the presence of anionic exchange resins. Single doses of either cholestyramine or colestipol resins bind the hydrochlorothiazide and reduce its absorption from the gastrointestinal tract by up to 85 and 43 percent, respectively. Stagger the dosage of hydrochlorothiazide and the resin such that hydrochlorothiazide is administered at least 4 hours before or 4 to 6 hours after the administration of the resin.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Losartan potassium and hydrochlorothiazide tablets can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the renin-angiotensin system from other antihypertensive agents. When pregnancy is detected, discontinue losartan potassium and hydrochlorothiazide tablets as soon as possible (see Clinical Considerations).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-associated Maternal and/or Embryo/Fetal Risk
Hypertension in pregnancy increases the maternal risk for pre-eclampsia, gestational diabetes, premature delivery, and delivery complications (e.g., need for cesarean section, post-partum hemorrhage). Hypertension increases the fetal risk for intrauterine growth restriction and intrauterine death. Pregnant women with hypertension should be carefully monitored and managed accordingly.
Fetal/Neonatal Adverse Reactions
Losartan:
Use of drugs that act on the renin-angiotensin system in the second and third trimesters of pregnancy can result in the following: oligohydramnios, reduced fetal renal function leading to anuria and renal failure, fetal lung hypoplasia, skeletal deformations, including skull hypoplasia, hypotension, and death.
In the unusual case that there is no appropriate alternative to therapy with drugs affecting the renin-angiotensin system for a particular patient, apprise the mother of the potential risk to the fetus. Perform serial ultrasound examinations to assess the intra-amniotic environment. If oligohydramnios is observed, discontinue losartan potassium and hydrochlorothiazide tablets, unless it is considered lifesaving for the mother. Fetal testing may be appropriate, based on the week of gestation. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. Closely observe neonates with histories of in utero exposure to losartan potassium and hydrochlorothiazide tablets for hypotension, oliguria, and hyperkalemia. In neonates with a history of in utero exposure to losartan potassium and hydrochlorothiazide tablets, if oliguria or hypotension occurs, support blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and replacing renal function.
Hydrochlorothiazide:
Thiazides can cross the placenta, and concentrations reached in the umbilical vein approach those in the maternal plasma. Hydrochlorothiazide, like other diuretics, can cause placental hypoperfusion. It accumulates in the amniotic fluid, with reported concentrations up to 19 times higher than in umbilical vein plasma. Use of thiazides during pregnancy is associated with a risk of fetal or neonatal jaundice or thrombocytopenia. Since they do not alter the course of pre-eclampsia, these drugs should not be used to treat hypertension in pregnant women. The use of hydrochlorothiazide for other indications in pregnancy should be avoided.
Data
Animal Data
There was no evidence of teratogenicity in rats or rabbits treated with a maximum losartan potassium dose of 10 mg/kg/day in combination with 2.5 mg/kg/day of hydrochlorothiazide. At these dosages, respective exposures (AUCs) of losartan, its active metabolite, and hydrochlorothiazide in rabbits were approximately 5, 1.5, and 1.0 times those achieved in humans with 100 mg losartan in combination with 25 mg hydrochlorothiazide. AUC values for losartan, its active metabolite and hydrochlorothiazide, extrapolated from data obtained with losartan administered to rats at a dose of 50 mg/kg/day in combination with 12.5 mg/kg/day of hydrochlorothiazide, were approximately 6, 2, and 2 times greater than those achieved in humans with 100 mg of losartan in combination with 25 mg of hydrochlorothiazide. Fetal toxicity in rats, as evidenced by a slight increase in supernumerary ribs, was observed when females were treated prior to and throughout gestation with 10 mg/kg/day losartan in combination with 2.5 mg/kg/day hydrochlorothiazide. As also observed in studies with losartan alone, adverse fetal and neonatal effects, including decreased body weight, renal toxicity, and mortality, occurred when pregnant rats were treated during late gestation and/or lactation with 50 mg/kg/day losartan in combination with 12.5 mg/kg/day hydrochlorothiazide. Respective AUCs for losartan, its active metabolite and hydrochlorothiazide at these dosages in rats were approximately 35, 10 and 10 times greater than those achieved in humans with the administration of 100 mg of losartan in combination with 25 mg hydrochlorothiazide. When hydrochlorothiazide was administered without losartan to pregnant mice and rats during their respective periods of major organogenesis, at doses up to 3000 and 1000 mg/kg/day, respectively, there was no evidence of harm to the fetus.
8.2 Lactation
Risk Summary
It is not known whether losartan is excreted in human milk, but significant levels of losartan and its active metabolite were shown to be present in rat milk. Thiazides appear in human milk. Because of the potential for adverse effects on the nursing infant, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
Safety and effectiveness of losartan potassium and hydrochlorothiazide tablets in pediatric patients have not been established.
Neonates with a history of in utero exposure to losartan potassium and hydrochlorothiazide tablets: If oliguria or hypotension occurs, direct attention toward support of blood pressure and renal perfusion. Exchange transfusion or dialysis may be required as means of reversing hypotension and/or substituting for disordered renal function.
8.5 Geriatric Use
In a controlled clinical study for the reduction in the combined risk of cardiovascular death, stroke and myocardial infarction in hypertensive patients with left ventricular hypertrophy, 2857 patients (62%) were 65 years and over, while 808 patients (18%) were 75 years and over. In an effort to control blood pressure in this study, patients were coadministered losartan and hydrochlorothiazide 74% of the total time they were on study drug. No overall differences in effectiveness were observed between these patients and younger patients. Adverse events were somewhat more frequent in the elderly compared to non-elderly patients for both the losartan-hydrochlorothiazide and the control groups [see Clinical Pharmacology (12.3)].
8.6 Race
In the Losartan Intervention For Endpoint reduction in hypertension (LIFE) study, Black patients with hypertension and left ventricular hypertrophy treated with atenolol had a lower risk of stroke, the primary composite endpoint, as compared with Black patients treated with losartan (both cotreated with hydrochlorothiazide in the majority of patients). In the subgroup of Black patients (n=533, 6% of the LIFE study patients), there were 29 primary endpoints among 263 patients on atenolol (11%, 26 per 1000 patient-years) and 46 primary endpoints among 270 patients (17%, 42 per 1000 patient-years) on losartan. This finding could not be explained on the basis of differences in the populations other than race or on any imbalances between treatment groups. In addition, blood pressure reductions in both treatment groups were consistent between Black and non-Black patients. Given the difficulty in interpreting subset differences in large trials, it cannot be known whether the observed difference is the result of chance. However, the LIFE study provides no evidence that the benefits of losartan on reducing the risk of cardiovascular events in hypertensive patients with left ventricular hypertrophy apply to Black patients [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Initiation of losartan potassium and hydrochlorothiazide tablets is not recommended for patients with hepatic impairment because the appropriate starting dose of losartan, 25 mg, is not available.
8.8 Renal Impairment
Changes in renal function have been reported in susceptible individuals [see Dosage and Administration (2.1), Warnings and Precautions (5.4), and Clinical Pharmacology (12.3)]. Safety and effectiveness of losartan potassium and hydrochlorothiazide tablets in patients with severe renal impairment (creatinine clearance <30 mL/min) have not been established.
10. Overdosage
Losartan Potassium
Significant lethality was observed in mice and rats after oral administration of 1000 mg/kg and 2000 mg/kg, respectively, about 44 and 170 times the maximum recommended human dose on a mg/m2 basis.
Limited data are available in regard to overdosage in humans. The most likely manifestation of overdosage would be hypotension and tachycardia; bradycardia could occur from parasympathetic (vagal) stimulation. If symptomatic hypotension should occur, supportive treatment should be instituted.
Neither losartan nor its active metabolite can be removed by hemodialysis.
Hydrochlorothiazide
The oral LD50 of hydrochlorothiazide is greater than 10 g/kg in both mice and rats. The most common signs and symptoms observed are those caused by electrolyte depletion (hypokalemia, hypochloremia, hyponatremia) and dehydration resulting from excessive diuresis. If digitalis has also been administered, hypokalemia may accentuate cardiac arrhythmias. The degree to which hydrochlorothiazide is removed by hemodialysis has not been established.
11. Losartan Potassium and Hydrochlorothiazide Description
Losartan Potassium and Hydrochlorothiazide Tablets USP, 50 mg/12.5 mg, Losartan Potassium and Hydrochlorothiazide Tablets USP, 100 mg/12.5 mg, and Losartan Potassium and Hydrochlorothiazide Tablets USP, 100 mg/25 mg combine an angiotensin II receptor blocker acting on the AT1 receptor subtype and a diuretic, hydrochlorothiazide, USP.
Losartan potassium USP, a non-peptide molecule, is chemically described as 2-butyl-4-chloro-1-[p-(o-1H-tetrazol-5-ylphenyl)benzyl]imidazole-5-methanol monopotassium salt. Its structural formula is:
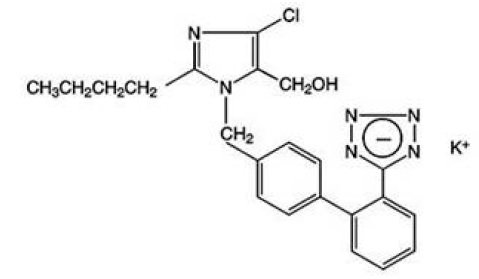
C22H22ClKN6O M.W. 461.01
Losartan potassium, USP is a white to off-white free-flowing crystalline powder. It is freely soluble in water, soluble in alcohols, and slightly soluble in common organic solvents, such as acetonitrile and methyl ethyl ketone.
Oxidation of the 5-hydroxymethyl group on the imidazole ring results in the active metabolite of losartan.
Hydrochlorothiazide, USP is 6-chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Its structural formula is:
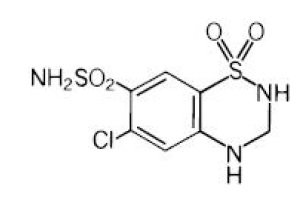
C7H8ClN3O4S2 M.W. 297.74
Hydrochlorothiazide, USP is a white, or practically white, crystalline powder, which is slightly soluble in water, but freely soluble in sodium hydroxide solution.
Losartan Potassium and Hydrochlorothiazide Tablets, USP are available for oral administration in three tablet combinations of losartan and hydrochlorothiazide, USP. Losartan Potassium and Hydrochlorothiazide Tablets USP, 50 mg/12.5 mg contain 50 mg of losartan potassium, USP and 12.5 mg of hydrochlorothiazide, USP. Losartan Potassium and Hydrochlorothiazide Tablets USP, 100 mg/12.5 mg contain 100 mg of losartan potassium, USP and 12.5 mg of hydrochlorothiazide, USP. Losartan Potassium and Hydrochlorothiazide Tablets USP, 100 mg/25 mg contain 100 mg of losartan potassium, USP and 25 mg of hydrochlorothiazide, USP. Inactive ingredients are: lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol-part hydrolyzed, pregelatinized corn starch, talc, and titanium dioxide. Additionally, 50 mg/12.5 mg tablets contain D&C Yellow #10 (Aluminum Lake) and FD&C Blue #1 (Aluminum Lake) and the 100 mg/25 mg tablets contain D&C Yellow #10 (Aluminum Lake).
Losartan Potassium and Hydrochlorothiazide Tablets USP, 50 mg/12.5 mg contain 4.24 mg (0.108 mEq) of potassium, Losartan Potassium and Hydrochlorothiazide Tablets USP, 100 mg/12.5 mg contain 8.48 mg (0.216 mEq) of potassium, and Losartan Potassium and Hydrochlorothiazide Tablets USP, 100 mg/25 mg contain 8.48 mg (0.216 mEq) of potassium.
12. Losartan Potassium and Hydrochlorothiazide - Clinical Pharmacology
12.1 Mechanism of Action
Losartan Potassium
Angiotensin II [formed from angiotensin I in a reaction catalyzed by angiotensin converting enzyme (ACE, kininase II)], is a potent vasoconstrictor, the primary vasoactive hormone of the renin-angiotensin system and an important component in the pathophysiology of hypertension. It also stimulates aldosterone secretion by the adrenal cortex. Losartan and its principal active metabolite block the vasoconstrictor and aldosterone-secreting effects of angiotensin II by selectively blocking the binding of angiotensin II to the AT1 receptor found in many tissues (e.g., vascular smooth muscle, adrenal gland). There is also an AT2 receptor found in many tissues but it is not known to be associated with cardiovascular homeostasis. Neither losartan nor its principal active metabolite exhibits any partial agonist activity at the AT1 receptor, and both have much greater affinity (about 1000-fold) for the AT1 receptor than for the AT2 receptor. In vitro binding studies indicate that losartan is a reversible, competitive inhibitor of the AT1 receptor. The active metabolite is 10 to 40 times more potent by weight than losartan and appears to be a reversible, non-competitive inhibitor of the AT1 receptor.
Neither losartan nor its active metabolite inhibits ACE (kininase II, the enzyme that converts angiotensin I to angiotensin II and degrades bradykinin), nor do they bind to or block other hormone receptors or ion channels known to be important in cardiovascular regulation.
Hydrochlorothiazide
Hydrochlorothiazide is a thiazide diuretic. Thiazides affect the renal tubular mechanisms of electrolyte reabsorption, directly increasing excretion of sodium and chloride in approximately equivalent amounts. Indirectly, the diuretic action of hydrochlorothiazide reduces plasma volume, with consequent increases in plasma renin activity, increases in aldosterone secretion, increases in urinary potassium loss, and decreases in serum potassium. The renin-aldosterone link is mediated by angiotensin II, so coadministration of an angiotensin II receptor antagonist tends to reverse the potassium loss associated with these diuretics. The mechanism of the antihypertensive effect of thiazides is unknown.
12.2 Pharmacodynamics
Losartan Potassium
Losartan inhibits the pressor effect of angiotensin II (as well as angiotensin I) infusions. A dose of 100 mg inhibits the pressor effect by about 85% at peak with 25 to 40% inhibition persisting for 24 hours. Removal of the negative feedback of angiotensin II causes a doubling to tripling in plasma renin activity and consequent rise in angiotensin II plasma concentration in hypertensive patients. Losartan does not affect the response to bradykinin, whereas ACE inhibitors increase the response to bradykinin. Aldosterone plasma concentrations fall following losartan administration. In spite of the effect of losartan on aldosterone secretion, very little effect on serum potassium was observed.
The effect of losartan is substantially present within one week but in some studies the maximal effect occurred in 3 to 6 weeks. In long-term follow-up studies (without placebo control) the effect of losartan appeared to be maintained for up to a year. There is no apparent rebound effect after abrupt withdrawal of losartan. There was essentially no change in average heart rate in losartan-treated patients in controlled trials.
Hydrochlorothiazide
After oral administration of hydrochlorothiazide, diuresis begins within 2 hours, peaks in about 4 hours, and lasts about 6 to 12 hours.
Drug Interactions
Hydrochlorothiazide
Alcohol, barbiturates, or narcotics — potentiation of orthostatic hypotension may occur.
Other antihypertensive drugs — additive effect or potentiation.
Skeletal muscle relaxants, nondepolarizing (e.g., tubocurarine) — possible increased responsiveness to the muscle relaxant.
Corticosteroids, ACTH, or glycyrrhizin (found in liquorice) — intensified electrolyte depletion, particularly hypokalemia.
Pressor amines (e.g., norepinephrine) — possible decreased response to pressor amines but not sufficient to preclude their use.
12.3 Pharmacokinetics
Losartan Potassium
Absorption: Following oral administration, losartan is well absorbed and undergoes substantial first-pass metabolism. The systemic bioavailability of losartan is approximately 33%. Mean peak concentrations of losartan and its active metabolite are reached in 1 hour and in 3 to 4 hours, respectively. While maximum plasma concentrations of losartan and its active metabolite are approximately equal, the AUC (area under the curve) of the metabolite is about 4 times as great as that of losartan. A meal slows absorption of losartan and decreases its Cmax but has only minor effects on losartan AUC or on the AUC of the metabolite (~10% decrease). The pharmacokinetics of losartan and its active metabolite are linear with oral losartan doses up to 200 mg and do not change over time.
Distribution: The volume of distribution of losartan and the active metabolite is about 34 liters and 12 liters, respectively. Both losartan and its active metabolite are highly bound to plasma proteins, primarily albumin, with plasma free fractions of 1.3% and 0.2%, respectively. Plasma protein binding is constant over the concentration range achieved with recommended doses. Studies in rats indicate that losartan crosses the blood-brain barrier poorly, if at all.
Metabolism: Losartan is an orally active agent that undergoes substantial first-pass metabolism by cytochrome P450 enzymes. It is converted, in part, to an active carboxylic acid metabolite that is responsible for most of the angiotensin II receptor antagonism that follows losartan treatment. About 14% of an orally-administered dose of losartan is converted to the active metabolite. In addition to the active carboxylic acid metabolite, several inactive metabolites are formed. In vitro studies indicate that cytochrome P450 2C9 and 3A4 are involved in the biotransformation of losartan to its metabolites.
Elimination: Total plasma clearance of losartan and the active metabolite is about 600 mL/min and 50 mL/min, respectively, with renal clearance of about 75 mL/min and 25 mL/min, respectively. The terminal half-life of losartan is about 2 hours and of the metabolite is about 6 to 9 hours. After single doses of losartan administered orally, about 4% of the dose is excreted unchanged in the urine and about 6% is excreted in urine as active metabolite. Biliary excretion contributes to the elimination of losartan and its metabolites. Following oral 14C-labeled losartan, about 35% of radioactivity is recovered in the urine and about 60% in the feces. Following an intravenous dose of 14C-labeled losartan, about 45% of radioactivity is recovered in the urine and 50% in the feces. Neither losartan nor its metabolite accumulate in plasma upon repeated once-daily dosing.
Hydrochlorothiazide
Hydrochlorothiazide is not metabolized but is eliminated rapidly by the kidney. When plasma levels have been followed for at least 24 hours, the plasma half-life has been observed to vary between 5.6 and 14.8 hours. At least 61 percent of the oral dose is eliminated unchanged within 24 hours. Hydrochlorothiazide crosses the placental but not the blood-brain barrier and is excreted in breast milk.
Specific Populations
Geriatric and Gender
Losartan pharmacokinetics have been investigated in the elderly (65 to 75 years) and in both genders. Plasma concentrations of losartan and its active metabolite are similar in elderly and young hypertensives. Plasma concentrations of losartan were about twice as high in female hypertensives as male hypertensives, but concentrations of the active metabolite were similar in males and females.
Race
Pharmacokinetic differences due to race have not been studied [see also Use in Specific Populations (8.6)].
Hepatic Insufficiency
Following oral administration in patients with mild to moderate alcoholic cirrhosis of the liver, plasma concentrations of losartan and its active metabolite were, respectively, 5 times and about 1.7 times those in young male volunteers. Compared to normal subjects, the total plasma clearance of losartan in patients with hepatic insufficiency was about 50% lower, and the oral bioavailability was about doubled. The lower starting dose of losartan recommended for use in patients with hepatic impairment cannot be given using losartan potassium and hydrochlorothiazide tablets. Its use in such patients as a means of losartan titration is, therefore, not recommended [see Warnings and Precautions (5.3) and Use in Specific Populations (8.7)].
Renal Insufficiency
Losartan
Following oral administration, plasma concentrations and AUCs of losartan and its active metabolite are increased by 50 to 90% in patients with mild (creatinine clearance of 50 to 74 mL/min) or moderate (creatinine clearance 30 to 49 mL/min) renal insufficiency. In this study, renal clearance was reduced by 55 to 85% for both losartan and its active metabolite in patients with mild or moderate renal insufficiency. Neither losartan nor its active metabolite can be removed by hemodialysis.
Hydrochlorothiazide
Following oral administration, the AUC for hydrochlorothiazide is increased by 70 and 700% for patients with mild and moderate renal insufficiency, respectively. In this study, renal clearance of hydrochlorothiazide decreased by 45 and 85% in patients with mild and moderate renal impairment, respectively.
Use the usual regimens of therapy with losartan potassium and hydrochlorothiazide tablets as long as the patient's creatinine clearance is greater than 30 mL/min. Safety and effectiveness of losartan potassium and hydrochlorothiazide tablets in patients with severe renal impairment (creatinine clearance less than 30 mL/min) have not been established [see Use in Specific Populations (8.8)].
Drug Interactions
Losartan Potassium
No clinically significant drug interactions have been found in studies of losartan potassium with hydrochlorothiazide, digoxin, warfarin, cimetidine and phenobarbital. However, rifampin has been shown to decrease the AUC of losartan and its active metabolite by 30% and 40%, respectively. Fluconazole, an inhibitor of cytochrome P450 2C9, decreased the AUC of the active metabolite by approximately 40%, but increased the AUC of losartan by approximately 70% following multiple doses. Conversion of losartan to its active metabolite after intravenous administration is not affected by ketoconazole, an inhibitor of P450 3A4.The AUC of active metabolite following oral losartan was not affected by erythromycin, an inhibitor of P450 3A4, but the AUC of losartan was increased by 30%.
The pharmacodynamic consequences of concomitant use of losartan and inhibitors of P450 2C9 have not been examined. Subjects who do not metabolize losartan to active metabolite have been shown to have a specific, rare defect in cytochrome P450 2C9. These data suggest that the conversion of losartan to its active metabolite is mediated primarily by P450 2C9 and not P450 3A4.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Losartan Potassium-Hydrochlorothiazide
No carcinogenicity studies have been conducted with the losartan potassium-hydrochlorothiazide combination.
Losartan potassium-hydrochlorothiazide when tested at a weight ratio of 4:1, was negative in the Ames microbial mutagenesis assay and the V-79 Chinese hamster lung cell mutagenesis assay. In addition, there was no evidence of direct genotoxicity in the in vitro alkaline elution assay in rat hepatocytes and in vitro chromosomal aberration assay in Chinese hamster ovary cells at noncytotoxic concentrations.
Losartan potassium, coadministered with hydrochlorothiazide, had no effect on the fertility or mating behavior of male rats at dosages up to 135 mg/kg/day of losartan and 33.75 mg/kg/day of hydrochlorothiazide. These dosages have been shown to provide respective systemic exposures (AUCs) for losartan, its active metabolite and hydrochlorothiazide that are approximately 60, 60 and 30 times greater than those achieved in humans with 100 mg of losartan potassium in combination with 25 mg of hydrochlorothiazide. In female rats, however, the coadministration of doses as low as 10 mg/kg/day of losartan and 2.5 mg/kg/day of hydrochlorothiazide was associated with slight but statistically significant decreases in fecundity and fertility indices. AUC values for losartan, its active metabolite and hydrochlorothiazide, extrapolated from data obtained with losartan administered to rats at a dose of 50 mg/kg/day in combination with 12.5 mg/kg/day of hydrochlorothiazide, were approximately 6, 2, and 2 times greater than those achieved in humans with 100 mg of losartan in combination with 25 mg of hydrochlorothiazide.
Losartan Potassium
Losartan potassium was not carcinogenic when administered at maximally tolerated dosages to rats and mice for 105 and 92 weeks, respectively. Female rats given the highest dose (270 mg/kg/day) had a slightly higher incidence of pancreatic acinar adenoma. The maximally tolerated dosages (270 mg/kg/day in rats, 200 mg/kg/day in mice) provided systemic exposures for losartan and its pharmacologically active metabolite that were approximately 160 and 90 times (rats) and 30 and 15 times (mice) the exposure of a 50 kg human given 100 mg per day.
Losartan potassium was negative in the microbial mutagenesis and V-79 mammalian cell mutagenesis assays and in the in vitro alkaline elution and in vitro and in vivo chromosomal aberration assays. In addition, the active metabolite showed no evidence of genotoxicity in the microbial mutagenesis, in vitro alkaline elution, and in vitro chromosomal aberration assays.
Fertility and reproductive performance were not affected in studies with male rats given oral doses of losartan potassium up to approximately 150 mg/kg/day. The administration of toxic dosage levels in females (300/200 mg/kg/day) was associated with a significant (p<0.05) decrease in the number of corpora lutea/female, implants/female, and live fetuses/female at C-section. At 100 mg/kg/day only a decrease in the number of corpora lutea/female was observed. The relationship of these findings to drug-treatment is uncertain since there was no effect at these dosage levels on implants/pregnant female, percent post-implantation loss, or live animals/litter at parturition. In nonpregnant rats dosed at 135 mg/kg/day for 7 days, systemic exposure (AUCs) for losartan and its active metabolite were approximately 66 and 26 times the exposure achieved in man at the maximum recommended human daily dosage (100 mg).
Hydrochlorothiazide
Two-year feeding studies in mice and rats conducted under the auspices of the National Toxicology Program (NTP) uncovered no evidence of a carcinogenic potential of hydrochlorothiazide in female mice (at doses of up to approximately 600 mg/kg/day) or in male and female rats (at doses of up to approximately 100 mg/kg/day). The NTP, however, found equivocal evidence for hepatocarcinogenicity in male mice.
Hydrochlorothiazide was not genotoxic in vitro in the Ames mutagenicity assay of Salmonella typhimurium strains TA 98, TA 100, TA 1535, TA 1537, and TA 1538 and in the Chinese Hamster Ovary (CHO) test for chromosomal aberrations, or in vivo in assays using mouse germinal cell chromosomes, Chinese hamster bone marrow chromosomes, and the Drosophila sex-linked recessive lethal trait gene. Positive test results were obtained only in the in vitro CHO Sister Chromatid Exchange (clastogenicity) and in the Mouse Lymphoma Cell (mutagenicity) assays, using concentrations of hydrochlorothiazide from 43 to 1300 mcg/mL, and in the Aspergillus nidulans non-disjunction assay at an unspecified concentration.
Hydrochlorothiazide had no adverse effects on the fertility of mice and rats of either sex in studies wherein these species were exposed, via their diet, to doses of up to 100 and 4 mg/kg, respectively, prior to mating and throughout gestation.
14. Clinical Studies
14.1 Losartan Monotherapy
Reduction in the Risk of Stroke: The LIFE study was a multinational, double-blind study comparing losartan and atenolol in 9193 hypertensive patients with ECG-documented left ventricular hypertrophy. Patients with myocardial infarction or stroke within six months prior to randomization were excluded. Patients were randomized to receive once daily losartan 50 mg or atenolol 50 mg. If goal blood pressure (<140/90 mmHg) was not reached, hydrochlorothiazide (12.5 mg) was added first and, if needed, the dose of losartan or atenolol was then increased to 100 mg once daily. If necessary, other antihypertensive treatments (e.g., increase in dose of hydrochlorothiazide therapy to 25 mg or addition of other diuretic therapy, calcium channel blockers, alpha-blockers, or centrally acting agents, but not ACE inhibitors, angiotensin II antagonists, or beta-blockers) were added to the treatment regimen to reach the goal blood pressure.
In efforts to control blood pressure, the patients in both arms of the LIFE study were coadministered hydrochlorothiazide the majority of time they were on study drug (73.9% and 72.4% of days in the losartan and atenolol arms, respectively).
Of the randomized patients, 4963 (54%) were female and 533 (6%) were Black. The mean age was 67 with 5704 (62%) age ≥65. At baseline, 1195 (13%) had diabetes, 1326 (14%) had isolated systolic hypertension, 1469 (16%) had coronary heart disease, and 728 (8%) had cerebrovascular disease. Baseline mean blood pressure was 174/98 mmHg in both treatment groups. The mean length of follow-up was 4.8 years. At the end of study or at the last visit before a primary endpoint, 77% of the group treated with losartan and 73% of the group treated with atenolol were still taking study medication. Of the patients still taking study medication, the mean doses of losartan and atenolol were both about 80 mg/day, and 15% were taking atenolol or losartan as monotherapy, while 77% were also receiving hydrochlorothiazide (at a mean dose of 20 mg/day in each group). Blood pressure reduction measured at trough was similar for both treatment groups but blood pressure was not measured at any other time of the day. At the end of study or at the last visit before a primary endpoint, the mean blood pressures were 144.1/81.3 mmHg for the group treated with losartan and 145.4/80.9 mmHg for the group treated with atenolol [the difference in SBP of 1.3 mmHg was significant (p<0.001), while the difference of 0.4 mmHg in DBP was not significant (p=0.098)].
The primary endpoint was the first occurrence of cardiovascular death, nonfatal stroke, or nonfatal myocardial infarction. Patients with nonfatal events remained in the trial, so that there was also an examination of the first event of each type even if it was not the first event (e.g., a stroke following an initial myocardial infarction would be counted in the analysis of stroke). Treatment with losartan resulted in a 13% reduction (p=0.021) in risk of the primary endpoint compared to the atenolol group; this difference was primarily the result of an effect on fatal and nonfatal stroke. Treatment with losartan reduced the risk of stroke by 25% relative to atenolol (p=0.001).
14.2 Losartan Potassium-Hydrochlorothiazide
The 3 controlled studies of losartan and hydrochlorothiazide included over 1300 patients assessing the antihypertensive efficacy of various doses of losartan (25, 50 and 100 mg) and concomitant hydrochlorothiazide (6.25, 12.5 and 25 mg). A factorial study compared the combination of losartan/hydrochlorothiazide 50/12.5 mg with its components and placebo. The combination of losartan/hydrochlorothiazide 50/12.5 mg resulted in an approximately additive placebo-adjusted systolic/diastolic response (15.5/9.0 mmHg for the combination compared to 8.5/5.0 mmHg for losartan alone and 7.0/3.0 mmHg for hydrochlorothiazide alone). Another study investigated the dose-response relationship of various doses of hydrochlorothiazide (6.25, 12.5 and 25 mg) or placebo on a background of losartan (50 mg) in patients not adequately controlled (Sitting Diastolic Blood Pressure [SiDBP] 93 to 120 mmHg) on losartan (50 mg) alone. The third study investigated the dose-response relationship of various doses of losartan (25, 50 and 100 mg) or placebo on a background of hydrochlorothiazide (25 mg) in patients not adequately controlled (SiDBP 93 to 120 mmHg) on hydrochlorothiazide (25 mg) alone. These studies showed an added antihypertensive response at trough (24 hours post-dosing) of hydrochlorothiazide 12.5 or 25 mg added to losartan 50 mg of 5.5/3.5 and 10.0/6.0 mmHg, respectively. Similarly, there was an added antihypertensive response at trough when losartan 50 or 100 mg was added to hydrochlorothiazide 25 mg of 9.0/5.5 and 12.5/6.5 mmHg, respectively. There was no significant effect on heart rate.
There was no difference in response for men and women or in patients over or under 65 years of age.
Black patients had a larger response to hydrochlorothiazide than non-Black patients and a smaller response to losartan. The overall response to the combination was similar for Black and non-Black patients.
Severe Hypertension (SiDBP ≥110 mmHg)
The safety and efficacy of losartan potassium and hydrochlorothiazide tablets as initial therapy for severe hypertension (defined as a mean SiDBP ≥110 mmHg confirmed on 2 separate occasions off all antihypertensive therapy) was studied in a 6-week double-blind, randomized, multicenter study. Patients were randomized to either losartan and hydrochlorothiazide (50/12.5 mg, once daily) or to losartan (50 mg, once daily) and followed for blood pressure response. Patients were titrated at 2-week intervals if their SiDBP did not reach goal (<90 mmHg). Patients on combination therapy were titrated from losartan 50 mg/hydrochlorothiazide 12.5 mg to losartan 50 mg/hydrochlorothiazide 12.5 mg (sham titration to maintain the blind) to losartan 100 mg/hydrochlorothiazide 25 mg. Patients on monotherapy were titrated from losartan 50 mg to losartan 100 mg to losartan 150 mg, as needed. The primary endpoint was a comparison at 4 weeks of patients who achieved goal diastolic blood pressure (trough SiDBP <90 mmHg).
The study enrolled 585 patients, including 264 (45%) females, 124 (21%) blacks, and 21 (4%) ≥65 years of age. The mean blood pressure at baseline for the total population was 171/113 mmHg. The mean age was 53 years. After 4 weeks of therapy, the mean SiDBP was 3.1 mmHg lower and the mean SiSBP was 5.6 mmHg lower in the group treated with losartan potassium and hydrochlorothiazide tablets. As a result, a greater proportion of the patients on losartan potassium and hydrochlorothiazide tablets reached the target diastolic blood pressure (17.6% for losartan potassium and hydrochlorothiazide tablets, 9.4% for losartan; p=0.006). Similar trends were seen when the patients were grouped according to gender, race or age (<, ≥ 65).
After 6 weeks of therapy, more patients who received the combination regimen reached target diastolic blood pressure than those who received the monotherapy regimen (29.8% versus 12.5%).
16. How is Losartan Potassium and Hydrochlorothiazide supplied
Losartan Potassium and Hydrochlorothiazide Tablets, USP are supplied as a film-coated tablet.
50 mg/12.5 mg - Yellow, oval-shaped, film-coated tablet, debossed with “93” on one side and “7367” on the other in bottles of 30 (NDC 0093-7367-56), 90 (NDC 0093-7367-98), and 1000 (NDC 0093-7367-10).
100 mg/12.5 mg - White to off-white, oval-shaped, film-coated tablet, debossed with “93” on one side and “7369” on the other in bottles of 30 (NDC 0093-7369-56), 90 (NDC 0093-7369-98), and 1000 (NDC 0093-7369-10).
100 mg/25 mg - Light yellow, oval-shaped, film-coated tablet, debossed with “93” on one side and “7368” on the other in bottles of 30 (NDC 0093-7368-56), 90 (NDC 0093-7368-98), and 1000 (NDC 0093-7368-10).
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Keep container tightly closed. Protect from light.
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
Keep this and all medications out of the reach of children.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Pregnancy: Advise female patients of childbearing age about the consequences of exposure to losartan potassium and hydrochlorothiazide tablets during pregnancy. Discuss treatment options with women planning to become pregnant. Tell patients to report pregnancies to their physicians as soon as possible [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
Symptomatic Hypotension: Advise patients that lightheadedness can occur, especially during the first days of therapy, and to report this symptom to a healthcare provider. Inform patients that dehydration from inadequate fluid intake, excessive perspiration, vomiting, or diarrhea may lead to an excessive fall in blood pressure. If syncope occurs, advise patients to contact their healthcare provider [see Warnings and Precautions (5.2)].
Potassium Supplements: Advise patients not to use potassium supplements or salt substitutes containing potassium without consulting their healthcare provider [see Drug Interactions (7.1)].
Acute Myopia and Secondary Angle-closure Glaucoma: Advise patients to discontinue losartan potassium and hydrochlorothiazide tablets and seek immediate medical attention if they experience symptoms of acute myopia or secondary angle-closure glaucoma [see Warnings and Precautions (5.6)].
Non-melanoma Skin Cancer: Instruct patients taking hydrochlorothiazide to protect skin from the sun and undergo regular skin cancer screening.
Dispense with Patient Package Insert available at: www.tevausa.com/PatientPI
Manufactured In Israel By:
Teva Pharmaceutical Ind. Ltd.
Kfar Saba, 4410202, Israel
Manufactured For:
Teva Pharmaceuticals
Parsippany, NJ 07054
Rev. O 11/2023
PATIENT INFORMATION
Dispense with Patient Package Insert available at: www.tevausa.com/Patient PI
Losartan Potassium (loe sar’ tan poe tas’ ee um) and Hydrochlorothiazide (hye droe klor oh thye' a zide) Tablets
50 mg/12.5 mg, 100 mg/12.5 mg, 100 mg/25 mg
Rx only
Read the Patient Information that comes with losartan potassium and hydrochlorothiazide tablets before you start taking them and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about your condition and treatment.
What is the most important information I should know about losartan potassium and hydrochlorothiazide tablets?
- Losartan potassium and hydrochlorothiazide tablets can cause harm or death to an unborn baby.
- Talk to your doctor about other ways to lower your blood pressure if you plan to become pregnant.
- If you get pregnant while taking losartan potassium and hydrochlorothiazide tablets tell your doctor right away.
What are losartan potassium and hydrochlorothiazide tablets?
Losartan potassium and hydrochlorothiazide tablets contain 2 prescription medicines, an angiotensin receptor blocker (ARB) and a diuretic (water pill). It is used to:
- lower high blood pressure (hypertension). Losartan potassium and hydrochlorothiazide tablets are not usually the first medicine used to treat high blood pressure.
- lower the chance of stroke in patients with high blood pressure and a heart problem called left ventricular hypertrophy (LVH). Losartan potassium and hydrochlorothiazide tablets may not help Black patients with this problem.
Losartan potassium and hydrochlorothiazide tablets have not been studied in children less than 18 years old.
High Blood Pressure (hypertension). Blood pressure is the force in your blood vessels when your heart beats and when your heart rests. You have high blood pressure when the force is too much. The losartan ingredient in losartan potassium and hydrochlorothiazide tablets can help your blood vessels relax so your blood pressure is lower. The hydrochlorothiazide ingredient in losartan potassium and hydrochlorothiazide tablets works by making your kidneys pass more water and salt.
Left Ventricular Hypertrophy (LVH) is an enlargement of the walls of the left chamber of the heart (the heart’s main pumping chamber). LVH can happen from several things. High blood pressure is the most common cause of LVH.
Who should not take losartan potassium and hydrochlorothiazide tablets?
Do not take losartan potassium and hydrochlorothiazide tablets if you:
- are allergic to any ingredients in losartan potassium and hydrochlorothiazide tablets. See a complete list of ingredients in losartan potassium and hydrochlorothiazide tablets at the end of this leaflet.
- are not passing urine.
- have diabetes and are taking a medicine called aliskiren to reduce blood pressure.
What should I tell my doctor before taking losartan potassium and hydrochlorothiazide tablets?
Tell your doctor about all your medical conditions including if you:
- are pregnant or planning to become pregnant. See "What is the most important information I should know about losartan potassium and hydrochlorothiazide tablets?"
- are breastfeeding or plan to breastfeed. Losartan potassium and hydrochlorothiazide can pass into your milk and may harm your baby. You and your doctor should decide if you will take losartan potassium and hydrochlorothiazide tablets or breastfeed. You should not do both.
- have been vomiting (throwing up), having diarrhea, sweating a lot, or not drinking enough fluids. These could cause you to have low blood pressure.
- have liver problems
- have kidney problems
- have systemic lupus erythematosus (Lupus; SLE)
- have diabetes
- have gout
- have any allergies
- have had skin cancer or if you develop a new skin lesion during the treatment. Treatment with hydrochlorothiazide may increase the risk of some types of skin cancer (non-melanoma skin cancer). Discuss with your doctor how to protect your skin from sun exposure and how often you should undergo skin cancer screening.
Tell your doctor about all of the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Losartan potassium and hydrochlorothiazide tablets and certain other medicines may interact with each other. Especially tell your doctor if you are taking:
- potassium supplements
- salt substitutes containing potassium
- other medicines that may increase serum potassium
- water pills (diuretics)
- lithium (a medicine used to treat a certain kind of depression)
- medicines used to treat pain and arthritis, called non-steroidal anti-inflammatory drugs (NSAIDs), including COX-2 inhibitors
- other medicines to reduce blood pressure.
Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine.
How should I take losartan potassium and hydrochlorothiazide tablets?
- Take losartan potassium and hydrochlorothiazide tablets exactly as prescribed by your doctor. Your doctor may change your dose if needed.
- Losartan potassium and hydrochlorothiazide tablets can be taken with or without food.
- If you miss a dose, take it as soon as you remember. If it is close to your next dose, do not take the missed dose. Just take the next dose at your regular time.
- If you take too many losartan potassium and hydrochlorothiazide tablets, call your doctor or Poison Control Center, or go to the nearest hospital emergency room right away.
- Your doctor may do blood tests from time to time while you are taking losartan potassium and hydrochlorothiazide tablets.
What are the possible side effects of losartan potassium and hydrochlorothiazide tablets?
Losartan potassium and hydrochlorothiazide tablets may cause the following side effects that may be serious:
- Injury or death of unborn babies. See "What is the most important information I should know about losartan potassium and hydrochlorothiazide tablets?"
- Allergic reaction. Symptoms of an allergic reaction are swelling of the face, lips, throat, or tongue and may include severe shortness of breath and/or difficulty breathing. Get emergency medical help right away and stop taking losartan potassium and hydrochlorothiazide tablets.
- Low blood pressure (hypotension). Low blood pressure may cause you to feel faint or dizzy. Lie down if you feel faint or dizzy. Call your doctor right away.
- If you have kidney problems, you may see a worsening in how well your kidneys work. Call your doctor if you get swelling in your feet, ankles, or hands, or unexplained weight gain.
- A new or worsening condition called systemic lupus erythematosus (Lupus; SLE)
-
Eye problems. One of the medicines in losartan potassium and hydrochlorothiazide tablets can cause eye problems that, if left untreated, may lead to vision loss. Symptoms of eye problems can happen within hours to weeks of starting losartan potassium and hydrochlorothiazide tablets. Tell your doctor right away if you have:
- decrease in vision
- eye pain
- Sensitivity of the skin to the sun and risk of skin cancer.
The most common side effects of losartan potassium and hydrochlorothiazide tablets in people with high blood pressure are:
- "colds" (upper respiratory infection)
- dizziness
- stuffy nose
- back pain
Tell your doctor if you get any side effect that bothers you or that won't go away. This is not a complete list of side effects. For a complete list, ask your doctor or pharmacist.
How should I store losartan potassium and hydrochlorothiazide tablets?
- Store losartan potassium and hydrochlorothiazide tablets at room temperature at 68° to 77°F (20° to 25°C).
- Keep losartan potassium and hydrochlorothiazide tablets in a tightly closed container, and keep losartan potassium and hydrochlorothiazide tablets out of the light.
- Keep losartan potassium and hydrochlorothiazide tablets and all medicines out of the reach of children.
General information about losartan potassium and hydrochlorothiazide tablets
Medicines are sometimes prescribed for conditions that are not mentioned in patient information leaflets. Do not use losartan potassium and hydrochlorothiazide tablets for a condition for which they were not prescribed. Do not give losartan potassium and hydrochlorothiazide tablets to other people, even if they have the same symptoms that you have. They may harm them.
This leaflet summarizes the most important information about losartan potassium and hydrochlorothiazide tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information that is written for health professionals.
What are the ingredients in losartan potassium and hydrochlorothiazide tablets?
Active ingredients: losartan potassium, hydrochlorothiazide
Inactive ingredients:
lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol-part hydrolyzed, pregelatinized corn starch, talc, and titanium dioxide. Additionally, 50 mg/12.5 mg tablets contain D&C Yellow # 10 (Aluminum Lake) and FD&C Blue # 1 (Aluminum Lake) and the 100 mg/25 mg tablets contain D&C Yellow # 10 (Aluminum Lake).
Manufactured In Israel By:
Teva Pharmaceutical Ind. Ltd.
Kfar Saba, 4410202, Israel
Manufactured For:
Teva Pharmaceuticals
Parsippany, NJ 07054
Rev. I 11/2023
Package/Label Display Panel
NDC 0093-7367-56
Losartan Potassium and Hydrochlorothiazide Tablets, USP
50 mg/12.5 mg
Rx only
30 Tablets

| LOSARTAN POTASSIUM AND HYDROCHLOROTHIAZIDE
losartan potassium and hydrochlorothiazide tablet, film coated |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| LOSARTAN POTASSIUM AND HYDROCHLOROTHIAZIDE
losartan potassium and hydrochlorothiazide tablet, film coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| LOSARTAN POTASSIUM AND HYDROCHLOROTHIAZIDE
losartan potassium and hydrochlorothiazide tablet, film coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Teva Pharmaceuticals USA, Inc. (001627975) |
More about hydrochlorothiazide / losartan
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (60)
- Drug images
- Latest FDA alerts (9)
- Side effects
- Dosage information
- During pregnancy
- Drug class: angiotensin II inhibitors with thiazides
- En español
Patient resources
- Hydrochlorothiazide and losartan drug information
- Losartan and hydrochlorothiazide (Advanced Reading)
- Losartan and Hydrochlorothiazide



