ZACTRAN
This page contains information on ZACTRAN for veterinary use.The information provided typically includes the following:
- ZACTRAN Indications
- Warnings and cautions for ZACTRAN
- Direction and dosage information for ZACTRAN
ZACTRAN
This treatment applies to the following species: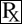 Company: Boehringer Ingelheim Animal Health
Company: Boehringer Ingelheim Animal Health
(gamithromycin)
Approved by FDA under NADA # 141-328
150 mg/mL ANTIMICROBIAL
For subcutaneous injection in beef and non-lactating dairy cattle only. Not for use in female dairy cattle 20 months of age or older or in calves to be processed for veal.
ZACTRAN Caution
Federal (USA) law restricts this drug to use by or on the order of a licensed veterinarian.
READ ENTIRE BROCHURE CAREFULLY BEFORE USING THIS PRODUCT.
Description
ZACTRAN® Injection for Cattle is a ready to use sterile parenteral solution containing gamithromycin, a macrolide sub-class, 7a-azalide antimicrobial. Each mL of ZACTRAN contains 150 mg of gamithromycin as the free base, 1 mg of monothioglycerol and 40 mg of succinic acid in a glycerol formal vehicle.
The chemical name of gamithromycin is 1 - Oxa - 7 - azacyclopentadecan - 15 - one,13 - [(2,6 - dideoy - 3 - C - methyl - 3 - O - methyl - alpha - L - ribo - hexopyranosyl)oxy] - 2 - ethyl - 3,4,10 - trihydroxy 3,5,8,10,12,14 - hexamethyl - 7 - propyl - 11 - {[3,4,6 - trideoxy - 3 - (dimethylamino) - beta - D - xylo - hexopyranosyl]oxy} - , [(2R*,3S*,4R*,5S*,8R*,10R*,11R*,12S*,13S*,14R*)]-and the structure is shown below.
ZACTRAN Indications
ZACTRAN is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida, Histophilus somni and Mycoplasma bovis in beef and non-lactating dairy cattle. ZACTRAN is also indicated for the control of respiratory disease in beef and non-lactating dairy cattle at high risk of developing BRD associated with Mannheimia haemolytica and Pasteurella multocida.
ZACTRAN Dosage And Administration
Administer ZACTRAN one time as a subcutaneous injection in the neck at 6 mg/kg (2 mL/110 lb) body weight (BW). If the total dose exceeds 10 mL, divide the dose so that no more than 10 mL is administered at each injection site.
|
Body Weight (lb) |
Dose Volume (mL) |
|
110 |
2 |
|
220 |
4 |
|
330 |
6 |
|
440 |
8 |
|
550 |
10 |
|
660 |
12 |
|
770 |
14 |
|
880 |
16 |
|
990 |
18 |
|
1100 |
20 |
Animals should be appropriately restrained to achieve the proper route of administration. Use sterile equipment. Inject under the skin in front of the shoulder (see illustration).
Contraindications
As with all drugs, the use of ZACTRAN is contraindicated in animals previously found to be hypersensitive to this drug.
Warning
FOR USE IN CATTLE ONLY.
NOT FOR USE IN HUMANS.
KEEP THIS AND ALL DRUGS OUT OF REACH OF CHILDREN.
NOT FOR USE IN CHICKENS OR TURKEYS.
The Safety Data Sheet (SDS) contains more detailed occupational safety information. To report suspected adverse drug events, for technical assistance, or to obtain a copy of the SDS, contact Boehringer Ingelheim Animal Health USA Inc. at 1-888-637-4251. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS, or online at www.fda.gov/reportanimalae.
 |
RESIDUE WARNINGS: Do not treat cattle within 35 days of slaughter. Because a discard time in milk has not been established, do not use in female dairy cattle 20 months of age or older. A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in calves to be processed for veal. |
 |
Precautions
The effects of ZACTRAN on bovine reproductive performance, pregnancy, and lactation have not been determined. Subcutaneous injection of ZACTRAN may cause a transient local tissue reaction in some cattle that may result in trim loss of edible tissues at slaughter.
Adverse Reactions
Transient animal discomfort and mild to moderate injection site swelling may be seen in cattle treated with ZACTRAN.
Clinical Pharmacology
The macrolide antimicrobials as a class are weak bases and as such concentrate in some cells (such as pulmonary leukocytes). Prolonged exposure of extracellular pulmonary pathogens to macrolides appears to reflect the slow release of drug from its intracellular reservoir to the site of action, the pulmonary epithelial lining fluid (ELF). It is the ELF that is relevant to the successful treatment and control of BRD. Gamithromycin is primarily bacteriostatic at therapeutic concentrations. However, in vitro bactericidal activity has been observed at concentrations of 10 µg/mL (Mueller-Hinton broth)
and after exposure to the 6-hour and 24-hour plasma samples derived from cattle dosed at 6 mg gamithromycin/kg BW.
Macrolides typically exhibit substantially higher concentrations in the alveolar macrophages and ELF as compared to concentrations observed in plasma. Gamithromycin concentrations in the ELF and ELF cells exceed the concentrations observed in the plasma. Postmortem gamithromycin concentrations in ELF exceed the MIC90 of M. haemolytica, H. somni and P. multocida through at least 72 hours after drug administration. Because M. haemolytica, P. multocida and H. somni are extracellular pathogens, drug concentrations in the ELF are considered to be clinically relevant. The postmortem area under the concentration-time curve (AUC) observed in lysed ELF cells (e.g., alveolar macrophages) are at least 300-times greater than that in the plasma. Although published studies suggest that inflammation can increase the release of drug from macrophages and neutrophils, these high concentrations in the alveolar macrophages should not be considered indicative of the magnitude or duration of response to the pathogens for which this product is indicated.
ZACTRAN administered subcutaneously in the neck of cattle at a single dosage of 6 mg/kg BW is rapidly and completely absorbed, with peak concentrations generally occurring within 1 hour after administration. Based upon plasma and lung homogenate data, the terminal half-life (T1/2) of gamithromycin is approximately 3 days. In vitro plasma protein binding studies show that 26% of the gamithromycin binds to plasma protein, resulting in free drug available for rapid and extensive distribution into body tissues. The free drug is rapidly cleared from the systemic circulation with a clearance rate of 712 mL/hr/kg and a volume of distribution of 25 L/kg. Dose proportionality was established based on AUC over a range of 3 mg/kg BW to 9 mg/kg BW. Biliary excretion of the unchanged drug is the major route of elimination.
Microbiology
The minimum inhibitory concentrations (MIC’s) of gamithromycin were determined for BRD isolates obtained from calves enrolled in BRD treatment field studies in the U.S. in 2004 using methods recommended by the Clinical and Laboratory Standards Institute (M31-A2). Isolates were obtained from pre-treatment nasopharyngeal swabs from each enrolled calf and from calves removed from the study due to BRD. The results are shown below in Table 1.
Table 1. Gamithromycin minimum inhibitory concentration (MIC) values* of indicated pathogens isolated from BRD treatment field studies in the U.S.
|
Indicated Pathogens |
Years of Isolation |
No. of isolates |
MIC50** (µg/mL) |
MIC90** (µg/mL) |
MIC range (µg/mL) |
|
M. haemolytica |
2004 |
89 |
1 |
1 |
0.5 to >32 |
|
P. multocida |
2004 |
79 |
0.5 |
1 |
0.12 to >32 |
|
H. somni |
2004 |
32 |
0.5 |
0.5 |
0.25 to 1 |
*The correlation between in vitro susceptibility data and clinical effectiveness is unknown.
**The lowest MIC to encompass 50% and 90% of the most susceptible isolates, respectively.
Effectiveness
The effectiveness of ZACTRAN for the treatment of BRD associated with Mannheimia haemolytica, Pasteurella multocida and Histophilus somni was demonstrated in a field study conducted at four geographic locations in the United States. A total of 497 cattle exhibiting clinical signs of BRD were enrolled in the study. Cattle were administered ZACTRAN (6 mg/kg BW) or an equivalent volume of sterile saline as a subcutaneous injection once on Day 0. Cattle were observed daily for clinical signs of BRD and were evaluated for clinical success on Day 10. The percentage of successes in cattle treated with ZACTRAN (58%) was statistically significantly higher (p<0.05) than the percentage of successes in the cattle treated with saline (19%).
The effectiveness of ZACTRAN for the treatment of BRD associated with M. bovis was demonstrated independently at two U.S. study sites. A total of 502 cattle exhibiting clinical signs of BRD were enrolled in the studies. Cattle were administered ZACTRAN (6 mg/kg BW) or an equivalent volume of sterile saline as a subcutaneous injection once on Day 0. At each site, the percentage of successes in cattle treated with ZACTRAN on Day 10 was statistically significantly higher than the percentage of successes in the cattle treated with saline (74.4% vs. 24% [p <0.001], and 67.4% vs. 46.2% [p = 0.002]). In addition, in the group of calves treated with gamithromycin that were confirmed positive for M. bovis (pre-treatment nasopharyngeal swabs), there were more calves at each site (45 of 57 calves, and 5 of 6 calves) classified as successes than as failures.
The effectiveness of ZACTRAN for the control of respiratory disease in cattle at high risk of developing BRD associated with Mannheimia haemolytica and Pasteurella multocida was demonstrated in two independent studies conducted in the United States. A total of 467 crossbred beef cattle at high risk of developing BRD were enrolled in the study. ZACTRAN (6 mg/kg BW) or an equivalent volume of sterile saline was administered as a single subcutaneous injection within one day after arrival. Cattle were observed daily for clinical signs of BRD and were evaluated for clinical success on Day 10 post-treatment. In each of the two studies, the percentage of successes in the cattle treated with ZACTRAN (86% and 78%) was statistically significantly higher (p = 0.0019 and p = 0.0016) than the percentage of successes in the cattle treated with saline (36% and 58%).
Animal Safety
In a target animal safety study in healthy, six-month old beef cattle, ZACTRAN was administered by subcutaneous injection at 6, 18, and 30 mg/kg bodyweight (1, 3, and 5 times the labeled dose) on Day 0, 5, and 10 (3 times the labeled administration frequency). Injection site discomfort (neck twisting, attempts to scratch or lick the injection site, and pawing at the ground) was observed in calves in the 18 mg/kg BW and 30 mg/kg BW groups at 10 minutes post-treatment following each injection. Mild to moderate injection site swelling and pathology changes consistent with inflammation were observed in the gamithromycin-treated groups. Other than injection site reactions, no clinically relevant treatment-related effects were observed.
Storage Conditions
Store at or below 77°F (25°C) with excursions between 59-86°F (15-30°C). Use within 18 months of first puncture.
HOW SUPPLIED
ZACTRAN is available in three ready-to-use bottle sizes. The 100, 250 and 500 mL bottles contain sufficient solution that will treat 10, 25 and 50 head of 550 lb (250 kg) cattle respectively.
Marketed by Boehringer Ingelheim Animal Health USA Inc., Duluth, GA 30096
Made in Austria
®ZACTRAN is a registered trademark of the Boehringer Ingelheim Group.
© 2020 Boehringer Ingelheim Animal Health USA Inc. All rights reserved.
M088802/04 US Code 6412 Rev. 07/2020
CPN: 1028348.0
3239 SATELLITE BLVD., BLDG 500, DULUTH, GA, 30096
| Telephone: | 800-325-9167 | |
| Customer Service: | 888-637-4251 | |
| Technical Service: | 888-637-4251 | |
| Fax: | 816-236-2717 | |
| Website: | www.boehringer-ingelheim.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |
Copyright © 2024 Animalytix LLC. Updated: 2024-02-27
