Velosef Prescribing Information
Package insert / product label
Generic name: Cephradine
Dosage form: capsule, suspension
Drug class: First generation cephalosporins
Medically reviewed by Drugs.com. Last updated on Mar 25, 2024.
On This Page
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Velosef and other antibacterial drugs, Velosef should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
Velosef Description
Velosef (Cephradine) is a semisynthetic cephalosporin antibiotic; oral dosage forms include capsules containing 250 mg and 500 mg cephradine and cephradine for oral suspension containing, after constitution, 125 mg and 250 mg per 5 mL dose.
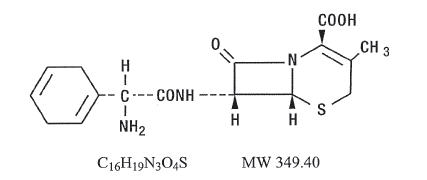
Cephradine is designated chemically as (6R, 7R)-7-[(R)-2-amino-2-(1,4-cyclohexadien-1-yl)acetamido]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid. Structural formula:
Inactive ingredients: Velosef Capsules—colorants (D&C Red No. 33 and Yellow No. 10; FD&C Blue No. 1, and, for ‘250’ only, Red No. 3), gelatin, lactose, magnesium stearate, talc, and titanium dioxide. Velosef for Oral Suspension—citric acid, colorants (FD&C Red No. 40 for ‘250’ only; FD&C Yellow No. 6 for ‘125’ only), flavors, guar gum, methylcellulose, sodium citrate, and sucrose.
Velosef - Clinical Pharmacology
Velosef (Cephradine) is acid stable. It is rapidly absorbed after oral administration in the fasting state. Following single doses of 250 mg, 500 mg, and 1 g in normal adult volunteers, average peak serum concentrations within one hour were approximately 9 mcg/mL, 16.5 mcg/mL, and 24.2 mcg/mL, respectively. In vitro studies by an ultracentrifugation technique show that at therapeutic serum antibiotic concentrations, cephradine is minimally bound (8 to 17 percent) to normal serum protein. Cephradine does not pass across the blood-brain barrier to any appreciable extent. The presence of food in the gastrointestinal tract delays absorption but does not affect the total amount of cephradine absorbed. Over 90 percent of the drug is excreted unchanged in the urine within six hours. Peak urine concentrations are approximately 1600 mcg/mL, 3200 mcg/mL, and 4000 mcg/mL following single doses of 250 mg, 500 mg, and 1 g, respectively.
Microbiology: In vitro tests demonstrate that the cephalosporins are bactericidal because of their inhibition of cell-wall synthesis. Cephradine is active against the following organisms in vitro:
- Group A beta-hemolytic streptococci
- Staphylococci, including coagulase-positive, coagulase-negative, and penicillinase-producing strains
- Streptococcus pneumoniae (formerly Diplococcus pneumoniae)
- Escherichia coli
- Proteus mirabilis
- Klebsiella species
- Hemophilus influenzae
Cephradine is not active against most strains of Enterobacter species, P. morganii, and P. vulgaris. It has no activity against Pseudomonas or Herellea species. When tested by in vitro methods, staphylococci exhibit cross-resistance between cephradine and methicillin-type antibiotics.
Note—Most strains of enterococci (Streptococcus faecalis) are resistant to cephradine.
Disc Susceptibility Tests: Quantitative methods that require measurement of zone diameters give the most precise estimates of antibiotic susceptibility. One recommended procedure (21 CFR § 460.1) uses cephalosporin class discs for testing susceptibility; interpretations correlate zone diameters of this disc test with MIC values for cephradine. With this procedure, a report from the laboratory of “resistant” indicates that the infecting organism is not likely to respond to therapy. A report of “intermediate susceptibility” suggests that the organism would be susceptible if the infection is confined to the urinary tract, as high antibiotic levels can be obtained in the urine, or if high dosage is used in other types of infection.
Related/similar drugs
prednisone, amoxicillin, doxycycline, azithromycin, cephalexin, ciprofloxacin, metronidazole
Indications and Usage for Velosef
Velosef (Cephradine) Capsules and Velosef for Oral Suspension are indicated in the treatment of the following infections when caused by susceptible strains of the designated microorganisms:
RESPIRATORY TRACT INFECTIONS (e.g., tonsillitis, pharyngitis, and lobar pneumonia) caused by group A beta-hemolytic streptococci and S. pneumoniae (formerly D. pneumonia).
(Penicillin is the usual drug of choice in the treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever. Velosef is generally effective in the eradication of streptococci from the nasopharynx; substantial data establishing the efficacy of Velosef in the subsequent prevention of rheumatic fever are not available at present.)
OTITIS MEDIA caused by group A beta-hemolytic streptococci, S. pneumoniae (formerly D. pneumoniae), H. influenzae, and staphylococci.
SKIN AND SKIN STRUCTURE INFECTIONS caused by staphylococci (penicillin-susceptible and penicillin-resistant) and beta-hemolytic streptococci.
URINARY TRACT INFECTIONS, including prostatitis, caused by E. coli, P. mirabilis, Klebsiella species, and enterococci (S. faecalis). The high concentrations of cephradine achievable in the urinary tract will be effective against many strains of enterococci for which disc susceptibility studies indicate relative resistance. It is to be noted that among beta-lactam antibiotics, ampicillin is the drug of choice for enterococcal urinary tract (S. faecalis) infection.
Note—Culture and susceptibility tests should be initiated prior to and during therapy.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Velosef and other antibacterial drugs, Velosef should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Following clinical improvement achieved with parenteral therapy, oral cephradine may be utilized for continuation of treatment of persistent or severe conditions where prolonged therapy is indicated.
Contraindications
Cephradine is contraindicated in patients with known hypersensitivity to the cephalosporin group of antibiotics.
Warnings
In penicillin-sensitive patients, cephalosporin derivatives should be used with great caution. There is clinical and laboratory evidence of partial cross-allergenicity of the penicillins and the cephalosporins, and there are instances of patients who have had reactions to both drug classes (including anaphylaxis after parenteral use).
Any patient who has demonstrated some form of allergy, particularly to drugs, should receive antibiotics, including cephradine, cautiously and then only when absolutely necessary.
Pseudomembranous colitis has been reported with the use of cephalosporins (and other broad spectrum antibiotics); therefore, it is important to consider its diagnosis in patients who develop diarrhea in association with antibiotic use. Treatment with broad spectrum antibiotics alters normal flora of the colon and may permit overgrowth of clostridia. Studies indicate a toxin produced by Clostridium difficile is one primary cause of antibiotic-associated colitis. Cholestyramine and colestipol resins have been shown to bind the toxin in vitro. Mild cases of colitis may respond to drug discontinuance alone. Moderate to severe cases should be managed with fluid, electrolyte and protein supplementation as indicated. When the colitis is not relieved by drug discontinuance or when it is severe, oral vancomycin is the treatment of choice for antibiotic-associated pseudomembranous colitis produced by C. difficile. Other causes of colitis should also be considered.
Precautions
General
Prescribing Velosef in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Patients should be followed carefully so that any side effects or unusual manifestations of drug idiosyncrasy may be detected. If a hypersensitivity reaction occurs, the drug should be discontinued and the patient treated with the usual agents, e.g., pressor amines, antihistamines, or corticosteroids.
Administer cephradine with caution in the presence of markedly impaired renal function. In patients with known or suspected renal impairment, careful clinical observation and appropriate laboratory studies should be made prior to and during therapy as cephradine accumulates in the serum and tissues. See DOSAGE AND ADMINISTRATION section for information on treatment of patients with impaired renal function.
Cephradine should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Prolonged use of antibiotics may promote the overgrowth of nonsusceptible organisms. Should superinfection occur during therapy, appropriate measures should be taken.
Indicated surgical procedures should be performed in conjunction with antibiotic therapy.
Information for Patients
Caution diabetic patients that false results may occur with urine glucose tests (see PRECAUTIONS, Drug/Laboratory Test Interactions).
Patients should be counseled that antibacterial drugs including Velosef should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Velosef is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Velosef or other antibacterial drugs in the future.
Advise the patient to comply with the full course of therapy even if he begins to feel better and to take a missed dose as soon as possible. Inform the patient that this medication may be taken with food or milk since gastrointestinal upset may be a factor in compliance with the dosage regimen. The patient should report current use of any medicines and should be cautioned not to take other medications unless the physician knows and approves of their use (see PRECAUTIONS, Drug Interactions).
Laboratory Tests
In patients with known or suspected renal impairment, it is advisable to monitor renal function (see DOSAGE AND ADMINISTRATION).
Drug Interactions
When administered concurrently, the following drugs may interact with cephalosporins:
Other antibacterial agents—Bacteriostats may interfere with the bactericidal action of cephalosporins in acute infection; other agents, e.g., aminoglycosides, colistin, polymyxins, vancomycin, may increase the possibility of nephrotoxicity.
Diuretics (potent “loop diuretics,” e.g., furosemide and ethacrynic acid)—Enhanced possibility for renal toxicity.
Probenecid—Increased and prolonged blood levels of cephalosporins, resulting in increased risk of nephrotoxicity.
Drug/Laboratory Test Interactions
After treatment with cephradine, a false-positive reaction for glucose in the urine may occur with Benedict’s solution, Fehling’s solution, or with Clinitest® tablets, but not with enzyme-based tests such as Clinistix® and Tes-Tape®.
False-positive Coombs test results may occur in newborns whose mothers received a cephalosporin prior to delivery.
Cephalosporins have been reported to cause false-positive reactions in tests for urinary proteins which use sulfosalicylic acid, false elevations of urinary 17-ketosteroid values, and prolonged prothrombin times.
Carcinogenesis, Mutagenesis
Long-term studies in animals have not been performed to evaluate carcinogenic potential or mutagenesis.
Pregnancy Category B
Reproduction studies have been performed in mice and rats at doses up to four times the maximum indicated human dose and have revealed no evidence of impaired fertility or harm to the fetus due to cephradine. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
Since cephradine is excreted in breast milk during lactation, caution should be exercised when cephradine is administered to a nursing woman.
Pediatric Use
See DOSAGE AND ADMINISTRATION. Adequate information is unavailable on the efficacy of b.i.d. regimens in children under nine months of age.
Adverse Reactions/Side Effects
As with other cephalosporins, untoward reactions are limited essentially to gastrointestinal disturbances and, on occasion, to hypersensitivity phenomena. The latter are more likely to occur in individuals who have previously demonstrated hypersensitivity and those with a history of allergy, asthma, hay fever, or urticaria.
The following adverse reactions have been reported following the use of cephradine:
Gastrointestinal: Symptoms of pseudomembranous colitis can appear during antibiotic treatment. Nausea and vomiting have been reported rarely.
Skin and Hypersensitivity Reactions: Mild urticaria or skin rash, pruritus, and joint pains were reported by very few patients.
Hematologic: Mild, transient eosinophilia, leukopenia, and neutropenia have been reported.
Liver: Transient mild rise of SGOT, SGPT, and total bilirubin have been observed with no evidence of hepatocellular damage.
Renal: Transitory rises in BUN have been observed in some patients treated with cephalosporins; their frequency increases in patients over 50 years old. In adults for whom serum creatinine determinations were performed, the rise in BUN was not accompanied by a rise in serum creatinine.
Other adverse reactions have included dizziness and tightness in the chest and candidal vaginitis.
Velosef Dosage and Administration
Adults: For respiratory tract infections (other than lobar pneumonia) and skin and skin structure infections, the usual dose is 250 mg every 6 hours or 500 mg every 12 hours.
For lobar pneumonia, the usual dose is 500 mg every 6 hours or 1 g every 12 hours.
For uncomplicated urinary tract infections, the usual dose is 500 mg every 12 hours. In more serious urinary tract infections, including prostatitis, 500 mg every 6 hours or 1 g every 12 hours may be administered.
Larger doses (up to 1 g every 6 hours) may be given for severe or chronic infections.
Children: No adequate information is available on the efficacy of b.i.d. regimens in children under nine months of age. The usual dose in children over nine months of age is 25 to 50 mg/kg/day administered in equally divided doses every 6 or 12 hours. For otitis media due to H. influenzae, doses are from 75 to 100 mg/kg/day administered in equally divided doses every 6 or 12 hours, but should not exceed 4 g per day. Dosage for children should not exceed dosage recommended for adults.
All patients, regardless of age and weight: Larger doses (up to 1 g q.i.d.) may be given for severe or chronic infections.
As with antibiotic therapy in general, treatment should be continued for a minimum of 48 to 72 hours after the patient becomes asymptomatic or evidence of bacterial eradication has been obtained. In infections caused by group A beta-hemolytic streptococci, a minimum of 10 days of treatment is recommended to guard against the risk of rheumatic fever or glomerulonephritis. In the treatment of chronic urinary tract infection, frequent bacteriologic and clinical appraisal is necessary during therapy and may be necessary for several months afterwards. Persistent infections may require treatment for several weeks. Prolonged intensive therapy is recommended for prostatitis. Doses smaller than those indicated are not recommended.
Patients With Impaired Renal Function
Not on Dialysis: The following initial dosage schedule is suggested as a guideline based on creatinine clearance. Further modification in the dosage schedule may be required because of individual variations in absorption.
| Creatine Clearance | Dose | Time Interval |
|---|---|---|
| > 20 mL/min | 500 mg | 6 hours |
| 5–20 mL/min | 250 mg | 6 hours |
| < 50 mL/min | 250 mg | 12 hours |
How is Velosef supplied
Velosef Capsules (Cephradine Capsules USP)
| Velosef ‘250’ 250 mg/capsule: | bottles of 24 (NDC 0003-0113-24) and 100 (NDC 0003-0113-50) and 100 Unimatic® unit-dose packs (NDC 0003-0113-52). Capsule identification no. 113. |
| Velosef ‘500’ 500 mg/capsule: | bottles of 24 (NDC 0003-0114-26) and 100 (NDC 0003-0114-50) and 100 Unimatic unit-dose packs (NDC 0003-0114-52). Capsule identification no. 114. |
| Velosef for Oral Suspension (Cephradine for Oral Suspension USP) | |
| Velosef ‘125’ | When constituted as directed on the container label, a pleasant fruit-flavored suspension containing 125 mg per 5 mL, in bottle sizes for preparation of 100 mL (NDC 0003-1193-50) and 200 mL (NDC 0003-1193-80). |
| Velosef ‘250’ | When constituted as directed on the container label, a pleasant fruit-flavored suspension containing 250 mg per 5 mL, in bottle sizes for preparation of 100 mL (NDC 0003-1194-50) and 200 mL (NDC 0003-1194-80). |
Storage
Velosef Capsules—Keep tightly closed. Do not store above 86° F.
Velosef for Oral Suspension—Prior to constitution, store at room temperature; avoid excessive heat. After constitution, when stored at room temperature, discard unused portion after seven days; when stored in refrigerator, discard unused portion after 14 days. Keep tightly closed.
| VELOSEF
cephradine capsule, gelatin coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| VELOSEF
cephradine capsule, gelatin coated |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| VELOSEF
cephradine suspension |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| VELOSEF
cephradine suspension |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Bristol-Myers Squibb Company |
More about Velosef (cephradine)
- Check interactions
- Compare alternatives
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: first generation cephalosporins
- Breastfeeding

