Ultiva Prescribing Information
Package insert / product label
Generic name: remifentanil hydrochloride
Dosage form: injection, powder, lyophilized, for solution
Drug class: Opioids (narcotic analgesics)
Medically reviewed by Drugs.com. Last updated on Jan 3, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Drug Abuse and Dependence
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
ULTIVA® (remifentanil hydrochloride), for injection, for intravenous use, CII
Initial U.S. Approval: 1996
WARNING: SERIOUS AND LIFE-THREATENING RISKS FROM USE OF ULTIVA
See full prescribing information for complete boxed warning.
- •
- ULTIVA exposes users to the risks of addiction, abuse, and misuse. Assess patient’s risk before prescribing and reassess regularly for the development of these behaviors and conditions.(5.1)
- •
- Serious, life-threatening, or fatal respiratory depression may occur with use of ULTIVA, especially during initiation or following a dosage increase. To reduce the risk of respiratory depression, proper dosing and titration of ULTIVA are essential. (5.2)
- •
- Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing for use in patients for whom alternative treatment options are inadequate. (5.3, 7)
Recent Major Changes
Indications and Usage for Ultiva
ULTIVA is an opioid agonist indicated for intravenous administration:
- •
- As an analgesic agent for use during the induction and maintenance of general anesthesia for inpatient and outpatient procedures. (1)
- •
- For continuation as an analgesic into the immediate postoperative period in adult patients under the direct supervision of an anesthesia practitioner in a postoperative anesthesia care unit or intensive care setting. (1)
- •
- As an analgesic component of monitored anesthesia care in adult patients. (1)
Ultiva Dosage and Administration
- •
- Monitor patients closely for respiratory depression when initiating therapy and following dosage increases and adjust the dosage accordingly. (2.1)
- •
- Initial Dosage in Adults: See full prescribing information for recommended doses in adult patients. (2.2, 2.3)
- •
- Initial Dosage in Pediatric Patients: See full prescribing information for recommended doses in pediatric patients. (2.2)
- •
- Geriatric Patients: The starting doses should be decreased by 50% in elderly patients (> 65 years). (2.6)
Dosage Forms and Strengths
For injection: 1 mg, 2 mg, and 5 mg for intravenous administration after reconstitution and dilution (3)
Contraindications
Warnings and Precautions
- •
- Life-Threatening Respiratory Depression: Monitor closely, particularly during initiation and titration. (5.2)
- •
- Risks from Use as Postoperative Analgesia with Concomitant Benzodiazepines or other CNS Depressants: Hypotension, profound sedation, respiratory depression, coma, and death may result from the concomitant use of ULTIVA with benzodiazepines or other CNS depressants (5.3)
- •
- Opioid-Induced Hyperalgesia and Allodynia: Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. If OIH is suspected, carefully consider appropriately decreasing the dose of the current opioid analgesic or opioid rotation. (5.4)
- •
- Serotonin Syndrome: Potentially life-threatening condition could result from concomitant serotonergic drug administration. Discontinue ULTIVA if serotonin syndrome is suspected. (5.5)
- •
- Administration: Continuous infusions of ULTIVA should be administered only by an infusion device. (5.6)
- •
- Skeletal Muscle Rigidity: is related to the dose and speed of administration. Muscle rigidity induced by ULTIVA should be managed in the context of the patient's clinical condition. (5.7)
- •
- Potential Inactivation by Nonspecific Esterases in Blood Products: ULTIVA should not be administered into the same IV tubing with blood due to potential inactivation by nonspecific esterases in blood products. (5.8)
- •
- Bradycardia: Monitor heart rate during dosage initiation and titration. It is responsive to ephedrine or anticholinergic drugs (5.9)
- •
- Hypotension: Monitor blood pressure during dosage initiation and titration. It is responsive to decreases in the administration of ULTIVA or to IV fluids or catecholamine administration (5.10)
- •
- Intraoperative Awareness: Inoperative awareness has been reported in patients under 55 years of age when ULTIVA has been administered with propofol infusion rates of ≤ 75 mcg/kg/min (5.11)
- •
- Risks of Use in Spontaneously Breathing Patients with Increased Intracranial Pressure, Brain Tumors, Head Injury, or Impaired Consciousness: Monitor for sedation and respiratory depression. (5.12)
- •
- Risks of Use in Patients with Biliary Tract Disease: Monitor patients with biliary tract disease, including acute pancreatitis, for worsening symptoms. (5.13)
- •
- Increased Risk of Seizures in Patients with Seizure Disorders: Monitor patients with a history of seizure disorders for worsened seizure control during ULTIVA therapy. (5.14)
- •
- Rapid Offset of Action: Standard monitoring should be maintained in the postoperative period to ensure adequate recovery without stimulation. (5.15)
Adverse Reactions/Side Effects
Most common adverse reactions (incidence ≥ 1%) were respiratory depression, bradycardia, hypotension, and skeletal muscle rigidity. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Mylan at 1-877-446-3679 (1-877-4-INFO-RX) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- •
- Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics: May reduce the analgesic effect of ULTIVA and/or precipitate withdrawal symptoms. If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. (7)
Use In Specific Populations
- •
- Pregnancy: May cause fetal harm. (8.1)
- •
- Labor or Delivery: Respiratory depression and other opioid effects may occur in newborns whose mothers are given ULTIVA shortly before delivery. (8.1)
- •
- Lactation: Infants exposed to ULTIVA through breast milk should be monitored for excess sedation and respiratory depression. (8.2)
- •
- Pediatric Use: ULTIVA has not been studied in pediatric patients for use as a postoperative analgesic or as an analgesic component of monitored anesthesia care. (8.4)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2023
Full Prescribing Information
WARNING: SERIOUS AND LIFE-THREATENING RISKS FROM USE OF ULTIVA
Addiction, Abuse, and Misuse
Because the use of ULTIVA exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death, assess each patient’s risk prior to prescribing and reassess all patients regularly for the development of these behaviors and conditions [see Warnings and Precautions (5.1)].
Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression may occur with use of ULTIVA, especially during initiation or following a dosage increase. To reduce the risk of respiratory depression, proper dosing and titration of ULTIVA are essential [see Warnings and Precautions (5.2)].
Risks From Concomitant Use With Benzodiazepines Or Other CNS Depressants
Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of ULTIVA and benzodiazepines or other CNS depressants for use in patients for whom alternative treatment options are inadequate [see Warnings and Precautions (5.3), Drug Interactions (7)].
1. Indications and Usage for Ultiva
ULTIVA is indicated for intravenous (IV) administration:
- •
- As an analgesic agent for use during the induction and maintenance of general anesthesia for inpatient and outpatient procedures.
- •
- For continuation as an analgesic into the immediate postoperative period in adult patients under the direct supervision of an anesthesia practitioner in a postoperative anesthesia care unit or intensive care setting.
- •
- As an analgesic component of monitored anesthesia care in adult patients.
2. Ultiva Dosage and Administration
2.1 Important Dosage and Administration Instructions
Monitor patients closely for respiratory depression when initiating therapy and following dosage increases with ULTIVA and adjust the dosage accordingly [see Warnings and Precautions (5.2)].
ULTIVA is for IV use only. Continuous infusions of ULTIVA should be administered only by an infusion device. The injection site should be close to the venous cannula and all IV tubing should be cleared at the time of discontinuation of infusion.
ULTIVA should not be administered without dilution.
Consider an alternative to ULTIVA for patients taking mixed agonist/antagonist and partial agonist opioid analgesics due to reduced analgesic effect or potential withdrawal symptoms. If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue ULTIVA if patient is not responding appropriately to treatment.
Discard unused portion.
2.2 General Anesthesia
ULTIVA is not recommended as the sole agent in general anesthesia because loss of consciousness cannot be assured and because of a high incidence of apnea, muscle rigidity, and tachycardia. ULTIVA is synergistic with other anesthetics; therefore, clinicians may need to reduce doses of thiopental, propofol, isoflurane, and midazolam by up to 75% with the coadministration of ULTIVA. The administration of ULTIVA must be individualized based on the patient's response.
Induction of Anesthesia
ULTIVA should be administered at an infusion rate of 0.5 to 1 mcg/kg/min with a hypnotic or volatile agent for the induction of anesthesia. If endotracheal intubation is to occur less than 8 minutes after the start of the infusion of ULTIVA, then an initial dose of 1 mcg/kg may be administered over 30 to 60 seconds.
ULTIVA should not be used as a sole agent for induction of anesthesia because loss of consciousness cannot be assured and because of a high incidence of apnea, muscle rigidity, and tachycardia.
Maintenance of Anesthesia
After endotracheal intubation, the infusion rate of ULTIVA should be decreased in accordance with the dosing guidelines in Tables 1 (adults, predominately ASA physical status I, II, or III) and 2 (pediatric patients).
- •
- Due to the fast onset and short duration of action of ULTIVA, the rate of administration during anesthesia can be titrated upward in 25% to 100% increments in adult patients or up to 50% increments in pediatric patients, or downward in 25% to 50% decrements every 2 to 5 minutes to attain the desired level of µ-opioid effect.
- •
- In response to light anesthesia or transient episodes of intense surgical stress, supplemental bolus doses of 1 mcg/kg may be administered every 2 to 5 minutes.
- •
- At infusion rates > 1 mcg/kg/min, increases in the concomitant anesthetic agents should be considered to increase the depth of anesthesia. [See Clinical Pharmacology: Specific Populations: Pediatric Population (12.3) and Dosage and Administration, Table 2 (2.2).]
|
|||
|
Phase |
Continuous IV Infusion of ULTIVA (mcg/kg/min) |
Range of ULTIVA (mcg/kg/min) |
Supplemental IV Bolus Dose of ULTIVA (mcg/kg) |
|
Induction of Anesthesia (through intubation) |
0.5 – 1* | ||
|
Maintenance of anesthesia with: | |||
|
Nitrous oxide (66%) |
0.4 |
0.1 – 2 |
1 |
|
Isoflurane (0.4 to 1.5 MAC) |
0.25 |
0.05 – 2 |
1 |
|
Propofol (100 to 200 mcg/kg/min) |
0.25 |
0.05 – 2 |
1 |
|
Continuation as an analgesic into the immediate postoperative period |
0.1 |
0.025 – 0.2 |
not recommended |
Table 2 summarizes the recommended doses in pediatric patients, predominantly ASA physical status I, II, or III. In pediatric patients, remifentanil was administered with nitrous oxide or nitrous oxide in combination with halothane, sevoflurane, or isoflurane. The use of atropine may blunt the potential for bradycardia that can occur upon administration of ULTIVA.
|
|||
|
Phase |
Continuous IV Infusion of ULTIVA (mcg/kg/min) |
Range of ULTIVA (mcg/kg/min) |
Supplemental IV Bolus Dose of ULTIVA (mcg/kg) |
|
Maintenance of anesthesia in patients aged 1 to 12 years old with*: | |||
|
Halothane (0.3 to 1.5 MAC) |
0.25 |
0.05 – 1.3 |
1 |
|
Sevoflurane (0.3 to 1.5 MAC) |
0.25 |
0.05 – 1.3 |
1 |
|
Isoflurane (0.4 to 1.5 MAC) |
0.25 |
0.05 – 1.3 |
1 |
|
Maintenance of anesthesia for patients from birth to 2 months of age with: | |||
|
Nitrous oxide (70%)† |
0.4 |
0.4 – 1.0 |
1‡ |
2.3 Continuation as an Analgesic into the Immediate Postoperative Period Under the Direct Supervision of an Anesthesia Practitioner
Infusions of ULTIVA may be continued into the immediate postoperative period for select patients for whom later transition to longer acting analgesics may be desired.
- •
- ULTIVA has not been studied in pediatric patients for use in the immediate postoperative period.
- •
- The use of bolus injections of ULTIVA to treat pain during the postoperative period is not recommended.
- •
- When used as an IV analgesic in the immediate postoperative period, ULTIVA should be initially administered by continuous infusion at a rate of 0.1 mcg/kg/min.
- •
- The infusion rate may be adjusted every 5 minutes in 0.025 mcg/kg/min increments to balance the patient's level of analgesia and respiratory rate.
- •
- Infusion rates greater than 0.2 mcg/kg/min are associated with respiratory depression (respiratory rate less than 8 breaths/min).
Due to the rapid offset of action of ULTIVA, no residual analgesic activity will be present within 5 to 10 minutes after discontinuation. For patients undergoing surgical procedures where postoperative pain is generally anticipated, alternative analgesics should be administered prior to discontinuation of ULTIVA. The choice of analgesic should be appropriate for the patient's surgical procedure and the level of follow-up care [see Clinical Studies (14)].
2.4 Analgesic Component of Monitored Anesthesia Care
It is strongly recommended that supplemental oxygen be supplied to the patient whenever ULTIVA is administered.
- •
- ULTIVA has not been studied for use in children in monitored anesthesia care.
Single Dose
A single IV dose of 0.5 to 1 mcg/kg over 30 to 60 seconds of ULTIVA may be given 90 seconds before the placement of the local or regional anesthetic block [see Warnings and Precautions (5.7)].
Continuous Infusion
When used alone as an IV analgesic component of monitored anesthesia care, ULTIVA should be initially administered by continuous infusion at a rate of 0.1 mcg/kg/min beginning 5 minutes before placement of the local or regional anesthetic block.
- •
- Because of the risk for hypoventilation, the infusion rate of ULTIVA should be decreased to 0.05 mcg/kg/min following placement of the block.
- •
- Thereafter, rate adjustments of 0.025 mcg/kg/min at 5 minute intervals may be used to balance the patient's level of analgesia and respiratory rate.
- •
- Rates greater than 0.2 mcg/kg/min are generally associated with respiratory depression (respiratory rates less than 8 breaths/min).
- •
- Bolus doses of ULTIVA administered simultaneously with a continuous infusion of ULTIVA to spontaneously breathing patients are not recommended.
Table 3 summarizes the recommended doses for monitored anesthesia care in adult patients, predominately ASA physical status I, II, or III.
|
Method |
Timing |
ULTIVA Alone |
ULTIVA + 2 mg |
|
Single IV Dose |
Given 90 seconds before |
1 mcg/kg over 30 to 60 seconds |
0.5 mcg/kg over 30 to 60 seconds |
|
Continuous IV Infusion |
Beginning 5 minutes |
0.1 mcg/kg/min |
0.05 mcg/kg/min |
|
After local anesthetic |
0.05 mcg/kg/min |
0.025 mcg/kg/min |
2.5 Discontinuation
Upon discontinuation of ULTIVA, the IV tubing should be cleared to prevent the inadvertent administration of ULTIVA at a later time.
For patients undergoing surgical procedures where postoperative pain is generally anticipated, alternative analgesics should be administered prior to discontinuation of ULTIVA. The choice of analgesic should be appropriate for the patient's surgical procedure and the level of follow-up care [see Clinical Studies (14)].
2.6 Dosage Modifications in Geriatric Patients
The starting doses of ULTIVA should be decreased by 50% in elderly patients (> 65 years). ULTIVA should then be cautiously titrated to effect [see Use in Specific Populations (8.5)].
2.7 Dosage Modifications in Pediatric Patients
See Table 2 for dosing recommendations for use of ULTIVA in pediatric patients from birth to 12 years of age for maintenance of anesthesia. [See Clinical Pharmacology: Specific Populations: Pediatric Population (12.3) and Dosage and Administration, Table 2 and Maintenance of Anesthesia (2.2).]
ULTIVA has not been studied in pediatric patients for use in the immediate postoperative period or for use as a component of monitored anesthesia care.
2.8 Dosage Modifications in Coronary Artery Bypass Surgery
Table 4 summarizes the recommended doses for induction, maintenance, and continuation as an analgesic into the ICU in adult patients, predominantly ASA physical status III or IV. To avoid hypotension during the induction phase, it is important to consider the concomitant medication regimens. [See Clinical Studies: Coronary Artery Bypass Surgery (14.5).]
| Phase | Continuous IV Infusion of ULTIVA (mcg/kg/min) | Range of Infusion Dose ULTIVA (mcg/kg/min) | Supplemental IV Bolus Dose of ULTIVA (mcg/kg) |
|---|---|---|---|
|
|||
|
Induction of Anesthesia |
1 | ||
|
Maintenance of Anesthesia |
1 |
0.125 to 4 |
0.5 to 1 |
|
Continuation as an analgesic |
1 |
0.05 to 1 | |
2.9 Dosage Modifications in Obese Patients
The starting doses of ULTIVA should be based on ideal body weight (IBW) in obese patients (greater than 30% over their IBW) [see Use in Specific Populations (8.6)].
2.10 Dosage Modifications in Preanesthetic Medication
The need for premedication and the choice of anesthetic agents must be individualized. In clinical studies, patients who received ULTIVA frequently received a benzodiazepine premedication.
2.11 Preparation for Administration
To reconstitute solution, add 1 mL of diluent per mg of remifentanil. Shake well to dissolve. When reconstituted as directed, the solution contains approximately 1 mg of remifentanil activity per 1 mL.
- •
- ULTIVA should be diluted to a recommended final concentration of 20, 25, 50, or 250 mcg/mL prior to administration (see Table 5). ULTIVA should not be administered without dilution.
|
Final Concentration |
Amount of ULTIVA in |
Final Volume After |
|
20 mcg/mL |
1 mg |
50 mL |
|
2 mg |
100 mL |
|
|
5 mg |
250 mL |
|
|
25 mcg/mL |
1 mg |
40 mL |
|
2 mg |
80 mL |
|
|
5 mg |
200 mL |
|
|
50 mcg/mL |
1 mg |
20 mL |
|
2 mg |
40 mL |
|
|
5 mg |
100 mL |
|
|
250 mcg/mL |
5 mg |
20 mL |
Continuous IV infusions of ULTIVA should be administered only by an infusion device. Infusion rates of ULTIVA can be individualized for each patient using Table 6:
|
Drug Delivery Rate (mcg/kg/min) |
Infusion Delivery Rate (mL/kg/h) |
|||
|
20 mcg/mL |
25 mcg/mL |
50 mcg/mL |
250 mcg/mL |
|
|
0.0125 |
0.038 |
0.03 |
0.015 |
not |
|
0.025 |
0.075 |
0.06 |
0.03 |
not |
|
0.05 |
0.15 |
0.12 |
0.06 |
0.012 |
|
0.075 |
0.23 |
0.18 |
0.09 |
0.018 |
|
0.1 |
0.3 |
0.24 |
0.12 |
0.024 |
|
0.15 |
0.45 |
0.36 |
0.18 |
0.036 |
|
0.2 |
0.6 |
0.48 |
0.24 |
0.048 |
|
0.25 |
0.75 |
0.6 |
0.3 |
0.06 |
|
0.5 |
1.5 |
1.2 |
0.6 |
0.12 |
|
0.75 |
2.25 |
1.8 |
0.9 |
0.18 |
|
1.0 |
3.0 |
2.4 |
1.2 |
0.24 |
|
1.25 |
3.75 |
3.0 |
1.5 |
0.3 |
|
1.5 |
4.5 |
3.6 |
1.8 |
0.36 |
|
1.75 |
5.25 |
4.2 |
2.1 |
0.42 |
|
2.0 |
6.0 |
4.8 |
2.4 |
0.48 |
When ULTIVA is used as an analgesic component of monitored analgesia care, a final concentration of 25 mcg/mL is recommended. When ULTIVA is used for pediatric patients 1 year of age and older, a final concentration of 20 or 25 mcg/mL is recommended. Table 7 is a guideline for milliliter-per-hour delivery for a solution of 20 mcg/mL with an infusion device.
|
Infusion Rate |
Patient Weight (kg) |
||||||
|
5 |
10 |
20 |
30 |
40 |
50 |
60 |
|
|
0.0125 |
0.188 |
0.375 |
0.75 |
1.125 |
1.5 |
1.875 |
2.25 |
|
0.025 |
0.375 |
0.75 |
1.5 |
2.25 |
3.0 |
3.75 |
4.5 |
|
0.05 |
0.75 |
1.5 |
3.0 |
4.5 |
6.0 |
7.5 |
9.0 |
|
0.075 |
1.125 |
2.25 |
4.5 |
6.75 |
9.0 |
11.25 |
13.5 |
|
0.1 |
1.5 |
3.0 |
6.0 |
9.0 |
12.0 |
15.0 |
18.0 |
|
0.15 |
2.25 |
4.5 |
9.0 |
13.5 |
18.0 |
22.5 |
27.0 |
|
0.2 |
3.0 |
6.0 |
12.0 |
18.0 |
24.0 |
30.0 |
36.0 |
|
0.25 |
3.75 |
7.5 |
15.0 |
22.5 |
30.0 |
37.5 |
45.0 |
|
0.3 |
4.5 |
9.0 |
18.0 |
27.0 |
36.0 |
45.0 |
54.0 |
|
0.35 |
5.25 |
10.5 |
21.0 |
31.5 |
42.0 |
52.5 |
63.0 |
|
0.4 |
6.0 |
12.0 |
24.0 |
36.0 |
48.0 |
60.0 |
72.0 |
Table 8 is a guideline for milliliter-per-hour delivery for a solution of 25 mcg/mL with an infusion device.
|
Infusion Rate |
Patient Weight (kg) |
|||||||||
|
10 |
20 |
30 |
40 |
50 |
60 |
70 |
80 |
90 |
100 |
|
|
0.0125 |
0.3 |
0.6 |
0.9 |
1.2 |
1.5 |
1.8 |
2.1 |
2.4 |
2.7 |
3.0 |
|
0.025 |
0.6 |
1.2 |
1.8 |
2.4 |
3.0 |
3.6 |
4.2 |
4.8 |
5.4 |
6.0 |
|
0.05 |
1.2 |
2.4 |
3.6 |
4.8 |
6.0 |
7.2 |
8.4 |
9.6 |
10.8 |
12.0 |
|
0.075 |
1.8 |
3.6 |
5.4 |
7.2 |
9.0 |
10.8 |
12.6 |
14.4 |
16.2 |
18.0 |
|
0.1 |
2.4 |
4.8 |
7.2 |
9.6 |
12.0 |
14.4 |
16.8 |
19.2 |
21.6 |
24.0 |
|
0.15 |
3.6 |
7.2 |
10.8 |
14.4 |
18.0 |
21.6 |
25.2 |
28.8 |
32.4 |
36.0 |
|
0.2 |
4.8 |
9.6 |
14.4 |
19.2 |
24.0 |
28.8 |
33.6 |
38.4 |
43.2 |
48.0 |
Table 9 is a guideline for milliliter-per-hour delivery for a solution of 50 mcg/mL with an infusion device.
|
Infusion Rate |
Patient Weight (kg) |
|||||||
|
30 |
40 |
50 |
60 |
70 |
80 |
90 |
100 |
|
|
0.025 |
2.1 |
2.4 |
2.7 |
3.0 |
||||
|
0.05 |
2.4 |
3.0 |
3.6 |
4.2 |
4.8 |
5.4 |
6.0 |
|
|
0.075 |
2.7 |
3.6 |
4.5 |
5.4 |
6.3 |
7.2 |
8.1 |
9.0 |
|
0.1 |
3.6 |
4.8 |
6.0 |
7.2 |
8.4 |
9.6 |
10.8 |
12.0 |
|
0.15 |
5.4 |
7.2 |
9.0 |
10.8 |
12.6 |
14.4 |
16.2 |
18.0 |
|
0.2 |
7.2 |
9.6 |
12.0 |
14.4 |
16.8 |
19.2 |
21.6 |
24.0 |
|
0.25 |
9.0 |
12.0 |
15.0 |
18.0 |
21.0 |
24.0 |
27.0 |
30.0 |
|
0.5 |
18.0 |
24.0 |
30.0 |
36.0 |
42.0 |
48.0 |
54.0 |
60.0 |
|
0.75 |
27.0 |
36.0 |
45.0 |
54.0 |
63.0 |
72.0 |
81.0 |
90.0 |
|
1.0 |
36.0 |
48.0 |
60.0 |
72.0 |
84.0 |
96.0 |
108.0 |
120.0 |
|
1.25 |
45.0 |
60.0 |
75.0 |
90.0 |
105.0 |
120.0 |
135.0 |
150.0 |
|
1.5 |
54.0 |
72.0 |
90.0 |
108.0 |
126.0 |
144.0 |
162.0 |
180.0 |
|
1.75 |
63.0 |
84.0 |
105.0 |
126.0 |
147.0 |
168.0 |
189.0 |
210.0 |
|
2.0 |
72.0 |
96.0 |
120.0 |
144.0 |
168.0 |
192.0 |
216.0 |
240.0 |
Table 10 is a guideline for milliliter-per-hour delivery for a solution of 250 mcg/mL with an infusion device.
|
Infusion Rate |
Patient Weight (kg) |
|||||||
|
30 |
40 |
50 |
60 |
70 |
80 |
90 |
100 |
|
|
0.1 |
0.72 |
0.96 |
1.20 |
1.44 |
1.68 |
1.92 |
2.16 |
2.40 |
|
0.15 |
1.08 |
1.44 |
1.80 |
2.16 |
2.52 |
2.88 |
3.24 |
3.60 |
|
0.2 |
1.44 |
1.92 |
2.40 |
2.88 |
3.36 |
3.84 |
4.32 |
4.80 |
|
0.25 |
1.80 |
2.40 |
3.00 |
3.60 |
4.20 |
4.80 |
5.40 |
6.00 |
|
0.5 |
3.60 |
4.80 |
6.00 |
7.20 |
8.40 |
9.60 |
10.80 |
12.00 |
|
0.75 |
5.40 |
7.20 |
9.00 |
10.80 |
12.60 |
14.40 |
16.20 |
18.00 |
|
1.0 |
7.20 |
9.60 |
12.00 |
14.40 |
16.80 |
19.20 |
21.60 |
24.00 |
|
1.25 |
9.00 |
12.00 |
15.00 |
18.00 |
21.00 |
24.00 |
27.00 |
30.00 |
|
1.5 |
10.80 |
14.40 |
18.00 |
21.60 |
25.20 |
28.80 |
32.40 |
36.00 |
|
1.75 |
12.60 |
16.80 |
21.00 |
25.20 |
29.40 |
33.60 |
37.80 |
42.00 |
|
2.0 |
14.40 |
19.20 |
24.00 |
28.80 |
33.60 |
38.40 |
43.20 |
48.00 |
2.12 Compatibility and Stability
Reconstitution and Dilution Prior to Administration
ULTIVA is stable for 24 hours at room temperature after reconstitution and further dilution to concentrations of 20 to 250 mcg/mL with the IV fluids listed below.
Sterile Water for Injection, USP
5% Dextrose Injection, USP
5% Dextrose and 0.9% Sodium Chloride Injection, USP
0.9% Sodium Chloride Injection, USP
0.45% Sodium Chloride Injection, USP
Lactated Ringer's and 5% Dextrose Injection, USP
ULTIVA is stable for 4 hours at room temperature after reconstitution and further dilution to concentrations of 20 to 250 mcg/mL with Lactated Ringer's Injection, USP.
ULTIVA has been shown to be compatible with these IV fluids when coadministered into a running IV administration set.
Compatibility with Other Therapeutic Agents
ULTIVA has been shown to be compatible with Diprivan® (propofol) Injection when coadministered into a running IV administration set. The compatibility of ULTIVA with other therapeutic agents has not been evaluated.
Incompatibilities
Nonspecific esterases in blood products may lead to the hydrolysis of remifentanil to its carboxylic acid metabolite. Therefore, administration of ULTIVA into the same IV tubing with blood is not recommended.
Note: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Product should be a clear, colorless liquid after reconstitution and free of visible particulate matter.
ULTIVA does not contain any antimicrobial preservative and thus care must be taken to assure the sterility of prepared solutions.
3. Dosage Forms and Strengths
For injection: 1 mg, 2 mg, and 5 mg:
|
3 mL Vial |
1 mg lyophilized powder |
|
5 mL Vial |
2 mg lyophilized powder |
|
10 mL Vial |
5 mg lyophilized powder |
4. Contraindications
ULTIVA is contraindicated:
- •
- For epidural or intrathecal administration due to the presence of glycine in the formulation [see Nonclinical Toxicology (13)].
- •
- In patients with hypersensitivity to remifentanil (e.g., anaphylaxis) [see Adverse Reactions (6.2)].
5. Warnings and Precautions
5.1 Addiction, Abuse, and Misuse
ULTIVA contains remifentanil, a Schedule II controlled substance. As an opioid, ULTIVA exposes users to the risks of addiction, abuse, and misuse [see Drug Abuse and Dependence (9)].
Opioids are sought for nonmedical use and are subject to diversion from legitimate prescribed use. Consider these risks when handling ULTIVA. Strategies to reduce these risks include proper product storage and control practices for a C-II drug. Contact local state professional licensing board or state-controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
5.2 Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression has been reported with the use of opioids, even when used as recommended. Respiratory depression, if not immediately recognized and treated, may lead to respiratory arrest and death.
ULTIVA should be administered only by persons specifically trained in the use of anesthetic drugs and the management of the respiratory effects of potent opioids, including respiration and cardiac resuscitation of patients in the age group being treated. Such training must include the establishment and maintenance of a patent airway and assisted ventilation. Resuscitative and intubation equipment, oxygen, and opioid antagonists must be readily available.
Respiratory depression in spontaneously breathing patients is generally managed by decreasing the rate of the infusion of ULTIVA by 50% or by temporarily discontinuing the infusion [see Overdosage (10)].
Carbon dioxide (CO2) retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids. While serious, life-threatening, or fatal respiratory depression can occur at any time during the use of ULTIVA, the risk is greatest during the initiation of therapy or following a dosage increase. Monitor patients closely for respiratory depression, especially when initiating therapy with and following dosage increases of ULTIVA.
ULTIVA should not be used in diagnostic or therapeutic procedures outside the monitored anesthesia care setting. Patients receiving monitored anesthesia care should be continuously monitored by persons not involved in the conduct of the surgical or diagnostic procedure. Oxygen saturation should be monitored on a continuous basis.
Patients with significant chronic obstructive pulmonary disease or cor pulmonale, and those with a substantially decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression are at increased risk of decreased respiratory drive including apnea, even at recommended dosages of ULTIVA. Elderly, cachectic, or debilitated patients may have altered pharmacokinetics or altered clearance compared to younger, healthier patients resulting in greater risk for respiratory depression. Monitor such patients closely including vital signs, particularly when initiating and titrating ULTIVA and when ULTIVA is given concomitantly with other drugs that depress respiration. To reduce the risk of respiratory depression, proper dosing and titration of ULTIVA are essential [see Dosage and Administration (2.11)].
5.3 Risks from Use as Postoperative Analgesia with Concomitant Benzodiazepines or Other CNS Depressants
When benzodiazepines or other CNS depressants are used with ULTIVA, pulmonary arterial pressure may be decreased. This fact should be considered by those who conduct diagnostic and surgical procedures where interpretation of pulmonary arterial pressure measurements might determine final management of the patient. When high dose or anesthetic dosages of ULTIVA are employed, even relatively small dosages of diazepam may cause cardiovascular depression.
When ULTIVA is used with CNS depressants, hypotension can occur. If it occurs, consider the possibility of hypovolemia and manage with appropriate parenteral fluid therapy. When operative conditions permit, consider repositioning the patient to improve venous return to the heart. Exercise care in moving and repositioning of patients because of the possibility of orthostatic hypotension. If volume expansion with fluids plus other countermeasures do not correct hypotension, consider administration of pressor agents other than epinephrine. Epinephrine may paradoxically decrease blood pressure in patients treated with a neuroleptic that blocks alpha adrenergic activity.
Hypotension, profound sedation, respiratory depression, coma, and death may result from the concomitant use of ULTIVA with benzodiazepines and/or other CNS depressants, including alcohol (e.g., non-benzodiazepine sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids). Patients should be advised to avoid alcohol for 24 hours after surgery [see Drug Interactions (7)].
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drug-related mortality compared to use of opioid analgesics alone. Because of similar pharmacological properties, it is reasonable to expect similar risk with the concomitant use of other CNS depressant drugs with opioid analgesics [see Drug Interactions (7)].
If the decision is made to manage postoperative pain with ULTIVA concomitantly with a benzodiazepine or other CNS depressant, start dosing with the lowest effective dosage and titrate based on clinical response. Monitor patients closely for signs and symptoms of respiratory depression, sedation, and hypotension. Fluids or other measures to counter hypotension should be available [see Drug Interactions (7)].
5.4 Opioid-Induced Hyperalgesia and Allodynia
Opioid-Induced Hyperalgesia (OIH) occurs when an opioid analgesic paradoxically causes an increase in pain, or an increase in sensitivity to pain. This condition differs from tolerance, which is the need for increasing doses of opioids to maintain a defined effect [see Dependence (9.3)]. Symptoms of OIH include (but may not be limited to) increased levels of pain upon opioid dosage increase, decreased levels of pain upon opioid dosage decrease, or pain from ordinarily non-painful stimuli (allodynia). These symptoms may suggest OIH only if there is no evidence of underlying disease progression, opioid tolerance, opioid withdrawal, or addictive behavior.
Cases of OIH have been reported, both with short-term and longer-term use of opioid analgesics. Though the mechanism of OIH is not fully understood, multiple biochemical pathways have been implicated. Medical literature suggests a strong biologic plausibility between opioid analgesics and OIH and allodynia. If a patient is suspected to be experiencing OIH, carefully consider appropriately decreasing the dose of the current opioid analgesic or opioid rotation (safely switching the patient to a different opioid moiety) [see Dosage and Administration (2), Warnings and Precautions (5.2)].
5.5 Serotonin Syndrome with Concomitant Use of Serotonergic Drugs
Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of ULTIVA with serotonergic drugs. Serotonergic drugs include selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that affect the serotonergic neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), and drugs that impair metabolism of serotonin (including MAO inhibitors, both those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue) [see Drug Interactions (7)]. This may occur within the recommended dosage range.
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination, rigidity), and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea) and can be fatal. The onset of symptoms generally occurs within several hours to a few days of concomitant use, but may occur later than that. Discontinue ULTIVA if serotonin syndrome is suspected.
5.6 Administration
Continuous infusions of ULTIVA should be administered only by an infusion device. IV bolus administration of ULTIVA should be used only during the maintenance of general anesthesia. In nonintubated patients, single doses of ULTIVA should be administered over 30 to 60 seconds.
Interruption of an infusion of ULTIVA will result in rapid offset of effect. Rapid clearance and lack of drug accumulation result in rapid dissipation of respiratory depressant and analgesic effects upon discontinuation of ULTIVA at recommended doses. Discontinuation of an infusion of ULTIVA should be preceded by the establishment of adequate postoperative analgesia.
Injections of ULTIVA should be made into IV tubing at or close to the venous cannula. Upon discontinuation of ULTIVA, the IV tubing should be cleared to prevent the inadvertent administration of ULTIVA at a later point in time. Failure to adequately clear the IV tubing to remove residual ULTIVA has been associated with the appearance of respiratory depression, apnea, and muscle rigidity upon the administration of additional fluids or medications through the same IV tubing.
5.7 Skeletal Muscle Rigidity
Skeletal muscle rigidity can be caused by ULTIVA and is related to the dose and speed of administration. ULTIVA may cause chest wall rigidity (inability to ventilate) after single doses of > 1 mcg/kg administered over 30 to 60 seconds, or after infusion rates > 0.1 mcg/kg/min. Single doses < 1 mcg/kg may cause chest wall rigidity when given concurrently with a continuous infusion of ULTIVA.
Muscle rigidity induced by ULTIVA should be managed in the context of the patient's clinical condition. Muscle rigidity occurring during the induction of anesthesia should be treated by the administration of a neuromuscular blocking agent and the concurrent induction medications and can be treated by decreasing the rate or discontinuing the infusion of ULTIVA or by administering a neuromuscular blocking agent. The neuromuscular blocking agents used should be compatible with the patient's cardiovascular status.
Muscle rigidity seen during the use of ULTIVA in spontaneously breathing patients may be treated by stopping or decreasing the rate of administration of ULTIVA. Resolution of muscle rigidity after discontinuing the infusion of ULTIVA occurs within minutes. In the case of life-threatening muscle rigidity, a rapid onset neuromuscular blocker or naloxone may be administered.
5.8 Potential Inactivation by Nonspecific Esterases in Blood Products
ULTIVA should not be administered into the same IV tubing with blood due to potential inactivation by nonspecific esterases in blood products.
5.9 Bradycardia
Bradycardia has been reported with ULTIVA and is responsive to ephedrine or anticholinergic drugs, such as atropine and glycopyrrolate.
5.10 Hypotension
Hypotension has been reported with ULTIVA and is responsive to decreases in the administration of ULTIVA or to IV fluids or catecholamine (ephedrine, epinephrine, norepinephrine, etc.) administration.
5.11 Intraoperative Awareness
Intraoperative awareness has been reported in patients under 55 years of age when ULTIVA has been administered with propofol infusion rates of ≤ 75 mcg/kg/min.
5.12 Risks of Use in Spontaneously Breathing Patients with Increased Intracranial Pressure, Brain Tumors, Head Injury, or Impaired Consciousness
In patients who may be susceptible to the intracranial effects of CO2 retention (e.g., those with evidence of increased intracranial pressure or brain tumors), ULTIVA may reduce respiratory drive, and the resultant CO2 retention can further increase intracranial pressure in spontaneously breathing patients. Monitor such patients for signs of sedation and respiratory depression, particularly when initiating therapy with ULTIVA.
Opioids may also obscure the clinical course in a patient with a head injury.
5.13 Risks of Use in Patients with Biliary Tract Disease
The remifentanil in ULTIVA may cause spasm of the sphincter of Oddi. Opioids may cause increases in serum amylase. Monitor patients with biliary tract disease, including acute pancreatitis, for worsening symptoms.
5.14 Increased Risk of Seizures in Patients with Seizure Disorders
The remifentanil in ULTIVA may increase the frequency of seizures in patients with seizure disorders, and may increase the risk of seizures occurring in other clinical settings associated with seizures. Monitor patients with a history of seizure disorders for worsened seizure control during ULTIVA therapy.
5.15 Rapid Offset of Action
Analgesic activity will subside within 5 to 10 minutes after discontinuation of administration of ULTIVA. However, respiratory depression may continue in some patients for up to 30 minutes after termination of infusion due to residual effects of concomitant anesthetics. Standard monitoring should be maintained in the postoperative period to ensure adequate recovery without stimulation. For patients undergoing surgical procedures where postoperative pain is generally anticipated, other analgesics should be administered prior to the discontinuation of ULTIVA.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are described, or described in greater detail, in other sections:
- •
- Addiction, Abuse, and Misuse [see Warnings and Precautions (5.1)]
- •
- Life-Threatening Respiratory Depression [see Warnings and Precautions (5.2)]
- •
- Interactions with Benzodiazepines or other CNS Depressants [see Warnings and Precautions (5.3)]
- •
- Opioid-Induced Hyperalgesia and Allodynia [see Warnings and Precautions (5.4)]
- •
- Serotonin Syndrome [see Warnings and Precautions (5.5)]
- •
- Skeletal Muscle Rigidity [see Warnings and Precautions (5.7)]
- •
- Bradycardia [see Warnings and Precautions (5.9)]
- •
- Hypotension [see Warnings and Precautions (5.10)]
- •
- Biliary Tract Disease [see Warnings and Precautions (5.13)]
- •
- Seizures [see Warnings and Precautions (5.14)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse event information is derived from controlled clinical studies that were conducted in a variety of surgical procedures of varying duration, using a variety of premedications and other anesthetics, and in patient populations with diverse characteristics including underlying disease.
Adults
Approximately 2,770 adult patients were exposed to ULTIVA in controlled clinical studies. The frequencies of adverse events during general anesthesia with the recommended doses of ULTIVA are given in Table 11. Each patient was counted once for each type of adverse event.
|
||||||
|
Adverse Event |
Induction/Maintenance |
Postoperative Analgesia |
After Discontinuation |
|||
|
ULTIVA |
Alfentanil/ Fentanyl |
ULTIVA |
Morphine |
ULTIVA |
Alfentanil/Fentanyl |
|
|
Nausea |
8 (< 1%) |
0 |
61 (22%) |
15 (15%) |
339 (36%) |
202 (43%) |
|
Hypotension |
178 (19%) |
30 (6%) |
0 |
0 |
16 (2%) |
9 (2%) |
|
Vomiting |
4 (< 1%) |
1 (< 1%) |
22 (8%) |
5 (5%) |
150 (16%) |
91 (20%) |
|
Muscle rigidity |
98 (11%)‡ |
37 (8%) |
7 (2%) |
0 |
2 (< 1%) |
1 (< 1%) |
|
Bradycardia |
62 (7%) |
24 (5%) |
3 (1%) |
3 (3%) |
11 (1%) |
6 (1%) |
|
Shivering |
3 (< 1%) |
0 |
15 (5%) |
9 (9%) |
49 (5%) |
10 (2%) |
|
Fever |
1 (< 1%) |
0 |
2 (< 1%) |
0 |
44 (5%) |
9 (2%) |
|
Dizziness |
0 |
0 |
1 (< 1%) |
0 |
27 (3%) |
9 (2%) |
|
Visual disturbance |
0 |
0 |
0 |
0 |
24 (3%) |
14 (3%) |
|
Headache |
0 |
0 |
1 (< 1%) |
1 (1%) |
21 (2%) |
8 (2%) |
|
Respiratory depression |
1 (< 1%) |
0 |
19 (7%) |
4 (4%) |
17 (2%) |
20 (4%) |
|
Apnea |
0 |
1 (< 1%) |
9 (3%) |
2 (2%) |
2 (< 1%) |
1 (< 1%) |
|
Pruritus |
2 (< 1%) |
0 |
7 (2%) |
1 (1%) |
22 (2%) |
7 (2%) |
|
Tachycardia |
6 (< 1%) |
7 (2%) |
0 |
0 |
10 (1%) |
8 (2%) |
|
Postoperative pain |
0 |
0 |
7 (2%) |
0 |
4 (< 1%) |
5 (1%) |
|
Hypertension |
10 (1%) |
7 (2%) |
5 (2%) |
3 (3%) |
12 (1%) |
8 (2%) |
|
Agitation |
2 (< 1%) |
0 |
3 (1%) |
1 (1%) |
6 (< 1%) |
1 (< 1%) |
|
Hypoxia |
0 |
0 |
1 (< 1%) |
0 |
10 (1%) |
7 (2%) |
In the elderly population (> 65 years), the incidence of hypotension is higher, whereas the incidence of nausea and vomiting is lower.
|
Adverse Event n |
Induction Maintenance |
Postoperative Analgesia |
After Discontinuation |
|||||||||
|
ULTIVA |
Alfentanil/Fentanyl |
ULTIVA |
Morphine |
ULTIVA |
Alfentanil/Fentanyl |
|||||||
|
Male 326 |
Female 595 |
Male 183 |
Female 283 |
Male 85 |
Female 196 |
Male 36 |
Female 62 |
Male 332 |
Female 597 |
Male 183 |
Female 283 |
|
|
Nausea |
2% |
< 1% |
0 |
0 |
12% |
26% |
8% |
19% |
22% |
45% |
30% |
52% |
|
Hypotension |
29% |
14% |
7% |
6% |
0 |
0 |
0 |
0 |
2% |
2% |
2% |
2% |
|
Vomiting |
< 1% |
< 1% |
0 |
< 1% |
4% |
10% |
0 |
8% |
5% |
22% |
8% |
27% |
|
Muscle rigidity |
17% |
7% |
14% |
4% |
6% |
1% |
0 |
0 |
< 1% |
< 1% |
0 |
< 1% |
The frequencies of adverse events from the clinical studies at the recommended doses of ULTIVA in monitored anesthesia care are given in Table 13.
|
|||
|
Adverse Event |
ULTIVA |
ULTIVA + 2 mg Midazolam†
|
Propofol (0.5 mg/kg then 50 mcg/kg/min) |
|
Nausea |
70 (44%) |
19 (18%) |
20 (32%) |
|
Vomiting |
35 (22%) |
5 (5%) |
13 (21%) |
|
Pruritus |
28 (18%) |
16 (16%) |
0 |
|
Headache |
28 (18%) |
12 (12%) |
6 (10%) |
|
Sweating |
10 (6%) |
0 |
1 (2%) |
|
Shivering |
8 (5%) |
1 (< 1%) |
1 (2%) |
|
Dizziness |
8 (5%) |
5 (5%) |
1 (2%) |
|
Hypotension |
7 (4%) |
0 |
6 (10%) |
|
Bradycardia |
6 (4%) |
0 |
7 (11%) |
|
Respiratory depression |
4 (3%) |
1 (< 1%)* |
0 |
|
Muscle rigidity |
4 (3%) |
0 |
1 (2%) |
|
Chills |
2 (1%) |
0 |
2 (3%) |
|
Flushing |
2 (1%) |
0 |
0 |
|
Warm sensation |
2 (1%) |
0 |
0 |
|
Pain at study IV site |
2 (1%) |
0 |
11 (17%) |
Other Adverse Events in Adult Patients
The frequencies of less commonly reported adverse clinical events from all controlled general anesthesia and monitored anesthesia care studies are presented below.
Event frequencies are calculated as the number of patients who were administered ULTIVA and reported an event divided by the total number of patients exposed to ULTIVA in all controlled studies including cardiac dose-ranging and neurosurgery studies (n = 1,883 general anesthesia, n = 609 monitored anesthesia care).
Incidence Less than 1%
Digestive: constipation, abdominal discomfort, xerostomia, gastro-esophageal reflux, dysphagia, diarrhea, ileus.
Cardiovascular: various atrial and ventricular arrhythmias, heart block, ECG change consistent with myocardial ischemia, elevated CPK-MB level, syncope.
Musculoskeletal: muscle stiffness, musculoskeletal chest pain.
Respiratory: cough, dyspnea, bronchospasm, laryngospasm, rhonchi, stridor, nasal congestion, pharyngitis, pleural effusion, hiccup(s), pulmonary edema, rales, bronchitis, rhinorrhea.
Nervous: anxiety, involuntary movement, prolonged emergence from anesthesia, confusion, awareness under anesthesia without pain, rapid awakening from anesthesia, tremors, disorientation, dysphoria, nightmare(s), hallucinations, paresthesia, nystagmus, twitch, seizure, amnesia.
Body as a Whole: decreased body temperature, anaphylactic reaction, delayed recovery from neuromuscular block.
Skin: rash, urticaria.
Urogenital: urine retention, oliguria, dysuria, urine incontinence.
Infusion Site Reaction: erythema, pruritus, rash.
Metabolic and Nutrition: abnormal liver function, hyperglycemia, electrolyte disorders, increased CPK level.
Hematologic and Lymphatic: anemia, lymphopenia, leukocytosis, thrombocytopenia.
The frequencies of adverse events from the clinical studies at the recommended doses of ULTIVA in cardiac surgery are given in Tables 14, 15, and 16. These tables represent adverse events collected during discrete phases of cardiac surgery. Any event should be viewed as temporally associated with drug administration and the phase indicated should not be perceived as the only time the event might occur.
|
||||||
|
Induction/Intubation |
Maintenance |
|||||
|
Adverse Event |
ULTIVA |
Fentanyl |
Sufentanil |
ULTIVA |
Fentanyl |
Sufentanil |
|
Hypotension |
18 (8%) |
6 (3%) |
7 (17%) |
26 (11%) |
6 (3%) |
1 (2%) |
|
Bradycardia |
9 (4%) |
5 (3%) |
0 |
3 (1%) |
1 (< 1%) |
1 (2%) |
|
Hypertension |
3 (1%) |
2 (1%) |
2 (5%) |
8 (4%) |
6 (3%) |
1 (2%) |
|
Constipation |
9 (4%) |
1 (< 1%) |
3 (7%) |
0 |
0 |
1 (2%) |
|
Muscle rigidity |
2 (< 1%) |
2 (1%) |
0 |
5 (2%) |
8 (5%) |
0 |
|
Premature ventricular beats |
1 (< 1%) |
0 |
0 |
3 (1%) |
1 (< 1%) |
0 |
|
Myocardial ischemia |
0 |
0 |
0 |
7 (3%) |
8 (5%) |
1 (2%) |
|
Atrial fibrillation |
0 |
0 |
0 |
7 (3%) |
3 (2%) |
1 (2%) |
|
Decreased cardiac output |
0 |
0 |
0 |
5 (2%) |
1 (< 1%) |
1 (2%) |
|
Tachycardia |
0 |
1 (< 1%) |
0 |
4 (2%) |
2 (1%) |
0 |
|
Coagulation disorder |
0 |
0 |
0 |
4 (2%) |
0 |
1 (2%) |
|
Arrhythmia |
0 |
0 |
0 |
3 (1%) |
0 |
0 |
|
Ventricular fibrillation |
0 |
0 |
0 |
3 (1%) |
1 (< 1%) |
1 (2%) |
|
Postoperative complication |
0 |
0 |
0 |
3 (1%) |
0 |
0 |
|
Third degree heart block |
0 |
0 |
0 |
2 (< 1%) |
0 |
1 (2%) |
|
Hemorrhage |
0 |
0 |
0 |
2 (< 1%) |
0 |
1 (2%) |
|
Perioperative complication |
0 |
0 |
0 |
2 (< 1%) |
1 (< 1%) |
1 (2%) |
|
Involuntary movement(s) |
0 |
0 |
0 |
2 (< 1%) |
3 (2%) |
0 |
|
Thrombocytopenia |
0 |
0 |
1 (2%) |
0 |
0 |
0 |
|
Oliguria |
0 |
0 |
0 |
0 |
3 (2%) |
0 |
|
Anemia |
0 |
0 |
0 |
2 (< 1%) |
2 (1%) |
0 |
|
|||
|
Adverse Event |
ULTIVA |
Fentanyl |
Sufentanil |
|
Hypertension |
14 (6%) |
8 (5%) |
2 (5%) |
|
Hypotension |
12 (5%) |
3 (2%) |
1 (2%) |
|
Tachycardia |
9 (4%) |
5 (3%) |
0 |
|
Shivering |
8 (4%) |
3 (2%) |
1 (2%) |
|
Nausea |
8 (4%) |
3 (2%) |
0 |
|
Hemorrhage |
4 (2%) |
1 (< 1%) |
1 (2%) |
|
Postoperative complication |
4 (2%) |
5 (3%) |
2 (5%) |
|
Agitation |
4 (2%) |
1 (< 1%) |
1 (2%) |
|
Ache |
4 (2%) |
0 |
0 |
|
Decreased cardiac output |
3 (1%) |
0 |
0 |
|
Arrhythmia |
3 (1%) |
0 |
0 |
|
Muscle rigidity |
2 (< 1%) |
1 (< 1%) |
2 (5%) |
|
Bradycardia |
2 (< 1%) |
2 (1%) |
0 |
|
Vomiting |
1 (< 1%) |
2 (1%) |
0 |
|
Premature ventricular beats |
1 (< 1%) |
2 (1%) |
0 |
|
Anemia |
0 |
3 (2%) |
0 |
|
Somnolence |
0 |
0 |
1 (2%) |
|
Fever |
0 |
2 (1%) |
0 |
|
|||
|
Adverse Event |
ULTIVA |
Fentanyl |
Sufentanil |
|
Nausea |
90 (40%) |
63 (36%) |
16 (39%) |
|
Vomiting |
33 (15%) |
26 (15%) |
3 (7%) |
|
Fever |
30 (13%) |
15 (9%) |
0 |
|
Atrial fibrillation |
27 (12%) |
33 (19%) |
4 (10%) |
|
Constipation |
20 (9%) |
35 (20%) |
3 (7%) |
|
Pleural effusion |
11 (5%) |
2 (1%) |
2 (5%) |
|
Hypotension |
8 (4%) |
8 (5%) |
1 (2%) |
|
Tachycardia |
9 (4%) |
15 (9%) |
0 |
|
Postoperative complication |
10 (4%) |
6 (3%) |
2 (5%) |
|
Oliguria |
7 (3%) |
7 (4%) |
1 (2%) |
|
Confusion |
7 (3%) |
10 (6%) |
5 (12%) |
|
Ache |
6 (3%) |
2 (1%) |
0 |
|
Anxiety |
6 (3%) |
6 (3%) |
0 |
|
Headache |
6 (3%) |
2 (1%) |
0 |
|
Perioperative complication |
5 (2%) |
7 (4%) |
1 (2%) |
|
Anemia |
5 (2%) |
5 (3%) |
1 (2%) |
|
Agitation |
5 (2%) |
3 (2%) |
1 (2%) |
|
Diarrhea |
5 (2%) |
1 (< 1%) |
1 (2%) |
|
Edema |
4 (2%) |
6 (3%) |
0 |
|
Dizziness |
4 (2%) |
3 (2%) |
1 (2%) |
|
Postoperative infection |
5 (2%) |
7 (4%) |
0 |
|
Hypoxia |
4 (2%) |
5 (3%) |
0 |
|
Apnea |
4 (2%) |
1 (< 1%) |
1 (2%) |
|
Hypertension |
3 (1%) |
3 (2%) |
0 |
|
Shivering |
3 (1%) |
1 (< 1%) |
0 |
|
Heartburn |
3 (1%) |
3 (2%) |
0 |
|
Atrial flutter |
3 (1%) |
1 (< 1%) |
0 |
|
Arrhythmia |
3 (1%) |
5 (3%) |
0 |
|
Hallucinations |
3 (1%) |
3 (2%) |
0 |
|
Pneumonia |
3 (1%) |
3 (2%) |
1 (2%) |
|
Pharyngitis |
3 (1%) |
1 (< 1%) |
1 (2%) |
|
Decreased mental acuity |
3 (1%) |
1 (< 1%) |
0 |
|
Dyspnea |
3 (1%) |
1 (< 1%) |
0 |
|
Cough |
3 (1%) |
0 |
0 |
|
Decreased cardiac output |
1 (< 1%) |
0 |
3 (7%) |
|
Renal insufficiency |
1 (< 1%) |
5 (3%) |
0 |
|
Bradycardia |
1 (< 1%) |
1 (< 1%) |
1 (2%) |
|
Urine retention |
2 (< 1%) |
3 (2%) |
0 |
|
Cerebral infarction |
2 (< 1%) |
2 (1%) |
1 (2%) |
|
Premature ventricular beats |
2 (< 1%) |
3 (2%) |
0 |
|
Cerebral ischemia |
1 (< 1%) |
1 (< 1%) |
1 (2%) |
|
Paresthesia |
2 (< 1%) |
2 (1%) |
0 |
|
Seizure |
2 (< 1%) |
1 (< 1%) |
1 (2%) |
|
Sleep disorder |
1 (< 1%) |
1 (< 1%) |
1 (2%) |
|
Bronchospasm |
1 (< 1%) |
6 (3%) |
0 |
|
Atelectasis |
2 (< 1%) |
3 (2%) |
0 |
|
Respiratory depression |
2 (< 1%) |
3 (2%) |
0 |
|
Pulmonary edema |
1 (< 1%) |
2 (1%) |
0 |
|
Respiratory distress |
2 (< 1%) |
0 |
1 (2%) |
|
Hyperkalemia |
2 (< 1%) |
3 (2%) |
0 |
|
Electrolyte disorder |
0 |
3 (2%) |
0 |
|
Chest congestion |
0 |
3 (2%) |
0 |
|
Hemoptysis |
0 |
2 (1%) |
0 |
|
Facial ptosis |
0 |
2 (1%) |
0 |
|
Hemorrhage |
0 |
2 (1%) |
0 |
|
Hematuria |
0 |
1 (< 1%) |
1 (2%) |
|
Visual disturbance(s) |
0 |
1 (< 1%) |
1 (2%) |
|
Hypokalemia |
0 |
2 (1%) |
0 |
|
Exacerbation of renal failure |
0 |
0 |
1 (2%) |
|
Blood in stool |
0 |
0 |
1 (2%) |
|
First degree heart block |
0 |
0 |
1 (2%) |
|
Pericarditis |
0 |
0 |
1 (2%) |
Pediatrics
ULTIVA has been studied in 342 pediatric patients in controlled clinical studies for maintenance of general anesthesia. In the pediatric population (birth to 12 years), the most commonly reported events were nausea, vomiting, and shivering.
The frequencies of adverse events during general anesthesia with the recommended doses of ULTIVA are given in Table 17. Each patient was counted once for each type of adverse event.
There were no adverse events ≥ 1% for any treatment group during the maintenance period in the pediatric patient general anesthesia studies.
|
Recovery |
Follow-up† |
|||||
|
Adverse Event |
ULTIVA |
Fentanyl |
Bupivacaine |
ULTIVA |
Fentanyl |
Bupivacaine |
|
Vomiting |
40 (12%) |
9 (9%) |
10 (12%) |
56 (16%) |
8 (8%) |
12 (14%) |
|
Nausea |
23 (8%) |
7 (7%) |
1 (1%) |
17 (6%) |
6 (6%) |
5 (6%) |
|
Shivering |
9 (3%) |
0 |
0 |
0 |
0 |
0 |
|
Rhonchi |
8 (3%) |
2 (2%) |
0 |
0 |
0 |
0 |
|
Postoperative complication |
5 (2%) |
2 (2%) |
0 |
4 (1%) |
0 |
0 |
|
Stridor |
4 (1%) |
2 (2%) |
0 |
0 |
0 |
0 |
|
Cough |
4 (1%) |
1 (< 1%) |
0 |
0 |
0 |
0 |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of remifentanil. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular: Asystole
Serotonin syndrome: Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of opioids with serotonergic drugs.
Anaphylaxis: Anaphylaxis has been reported with ingredients contained in ULTIVA.
Hyperalgesia and Allodynia: Cases of hyperalgesia and allodynia have been reported with opioid therapy of any duration [see Warnings and Precautions (5.4)].
Hypoglycemia: Cases of hypoglycemia have been reported in patients taking opioids. Most reports were in patients with at least one predisposing risk factor (e.g., diabetes).
7. Drug Interactions
Table 18 includes clinically significant drug interactions with ULTIVA.
|
Benzodiazepines and other Central Nervous System (CNS) Depressants |
|
|
Clinical Impact: |
Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants including alcohol, increases the risk of hypotension, respiratory depression, profound sedation, coma, and death. |
|
Intervention: |
Limit dosages and durations to the minimum required. Follow patients closely for signs of respiratory depression and sedation. Patients should be advised to avoid alcohol for 24 hours after surgery [see Warnings and Precautions (5.3)]. |
|
Examples: |
Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol. |
|
Serotonergic Drugs |
|
|
Clinical Impact: |
The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome [see Warnings and Precautions (5.5)]. |
|
Intervention: |
If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue ULTIVA if serotonin syndrome is suspected. |
|
Examples: |
Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that effect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
|
Monoamine Oxidase Inhibitors (MAOIs) |
|
|
Clinical Impact: |
MAOI interactions with opioids may manifest as serotonin syndrome [see Warnings and Precautions (5.5)] or opioid toxicity (e.g., respiratory depression, coma) [see Warnings and Precautions (5.2)]. If urgent use of ULTIVA is necessary, use test doses and frequent titration of small doses while closely monitoring blood pressure and signs and symptoms of CNS and respiratory depression. |
|
Intervention: |
The use of ULTIVA is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. |
|
Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics |
|
|
Clinical Impact: |
May reduce the analgesic effect of ULTIVA and/or precipitate withdrawal symptoms. |
|
Intervention: |
If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Consider discontinuing ULTIVA if patient is not responding appropriately to treatment and institute alternative analgesic treatment. |
|
Examples: |
butorphanol, nalbuphine, pentazocine, buprenorphine |
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Use of opioid analgesics for an extended period of time during pregnancy may cause neonatal opioid withdrawal syndrome. Available data with remifentanil hydrochloride in pregnant women are insufficient to inform a drug-associated risk for major birth defects and miscarriage. In animal reproduction studies, reduced fetal rat body weight and pup weights were reported at 2.2 times a human intravenous infusion of an induction dose of 1 mcg/kg with a maintenance dose of 2 mcg/kg/min for a surgical procedure lasting 3 hours. There were no malformations noted when remifentanil was administered via bolus injection to pregnant rats or rabbits during organogenesis at doses approximately 5 times and approximately equal, respectively, to a human intravenous infusion of an induction dose of 1 mcg/kg with a maintenance dose of 2 mcg/kg/min for a surgical procedure lasting 3 hours [see Data]. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Labor or Delivery
Opioids cross the placenta and may produce respiratory depression and psycho-physiologic effects in neonates. An opioid antagonist, such as naloxone, must be available for reversal of opioid-induced respiratory depression in the neonate. ULTIVA is not recommended for use in pregnant women during or immediately prior to labor, when other analgesic techniques are more appropriate. Opioid analgesics, including ULTIVA, can prolong labor through actions which temporarily reduce the strength, duration, and frequency of uterine contractions. However, this effect is not consistent and may be offset by an increased rate of cervical dilation, which tends to shorten labor. Monitor neonates exposed to opioid analgesics during labor for signs of excess sedation and respiratory depression.
Data
Human Data
In a human clinical trial, the average maternal remifentanil concentrations were approximately twice those seen in the fetus. In some cases, however, fetal concentrations were similar to those in the mother. The umbilical arteriovenous ratio of remifentanil concentrations was approximately 30% suggesting metabolism of remifentanil in the neonate.
Animal Data
Pregnant rats were treated from Gestation Day 6 to 15 with intravenous remifentanil doses of 0.5, 1.6, or 5 mg/kg/day (0.2, 0.7, or 2.2 times a human intravenous infusion of an induction dose of 1 mcg/kg with a maintenance dose of 2 mcg/kg/min based on body surface area for a surgical procedure lasting 3 hours based on body surface area, respectively). Reduced fetal weights were reported in the high dose group; however, no malformations were reported in surviving fetuses despite a non-dose dependent increase in maternal mortality.
Pregnant rabbits were treated from Gestation Day 6 to 18 with intravenous remifentanil doses of 0.1, 0.5, or 0.8 mg/kg/day (0.09, 0.4, or 0.7 times a human intravenous infusion of an induction dose of 1 mcg/kg with a maintenance dose of 2 mcg/kg/min based on body surface area for a surgical procedure lasting 3 hours based on body surface area, respectively). No malformations were reported in surviving fetuses despite clear maternal toxicity (decreased food consumption and body weights and increased mortality in all treatment groups).
Pregnant rats were treated from Gestation Day 6 to Lactation Day 21 with intravenous boluses of remifentanil 0.5, 1.6, or 5 mg/kg/day (0.2, 0.7, or 2.2 times a human intravenous infusion of an induction dose of 1 mcg/kg with a maintenance dose of 2 mcg/kg/min based on body surface area for a surgical procedure lasting 3 hours based on body surface area, respectively). Reduced birth weights were noted in the high-dose groups in the presence of maternal toxicity (increased mortality in all groups).
8.2 Lactation
Risk Summary
It is not known whether remifentanil is excreted in human milk. After receiving radioactive-labeled remifentanil, the radioactivity was present in the milk of lactating rats. Because fentanyl analogs are excreted in human milk, caution should be exercised when ULTIVA is administered to a nursing woman.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ULTIVA and any potential adverse effects on the breastfed infant from ULTIVA or from the underlying maternal condition.
8.4 Pediatric Use
The efficacy and safety of ULTIVA as an analgesic agent for use in the maintenance of general anesthesia in outpatient and inpatient pediatric surgery have been established in controlled clinical studies in pediatric patients from birth to 12 years [see Clinical Studies (14.4)].
The initial maintenance infusion regimen of ULTIVA evaluated in pediatric patients from birth to 2 months of age was 0.4 mcg/kg/min, the approved adult regimen for use with N2O. The clearance rate observed in neonates was highly variable and on average was 2 times higher than in the young healthy adult population. Therefore, while a starting infusion rate of 0.4 mcg/kg/min may be appropriate for some neonates, an increased infusion rate may be necessary to maintain adequate surgical anesthesia, and additional bolus doses may be required. The individual dose for each patient should be carefully titrated. [See Clinical Pharmacology: Specific Populations: Pediatric Population (12.3) and Dosage and Administration, Table 2 and Maintenance of Anesthesia (2.2).]
ULTIVA has not been studied in pediatric patients for use as a postoperative analgesic or as an analgesic component of monitored anesthesia care.
8.5 Geriatric Use
Respiratory depression is the chief risk for elderly patients treated with opioids and has occurred after large initial doses were administered to patients who were not opioid-tolerant or when opioids were co-administered with other agents that depress respiration. Titrate the dosage of ULTIVA slowly in geriatric patients and frequently reevaluate the patient for signs of central nervous system and respiratory depression [see Warnings and Precautions (5.2)]
Of the total number of subjects in clinical studies of ULTIVA, 486 were 65 and over (age range 66 to 90 years). While the effective biological half-life of remifentanil is unchanged, elderly patients have been shown to be twice as sensitive as the younger population to the pharmacodynamic effects of remifentanil. The recommended starting dose of ULTIVA should be decreased by 50% in patients over 65 years of age [see Clinical Pharmacology (12.3) and Dosage and Administration (2.2)]. Titrate the dosage of ULTIVA slowly in geriatric patients. [See Warnings and Precautions (5.5).]
The clearance of remifentanil is reduced (approximately 25%) in the elderly (> 65 years of age) compared to young adults (average 25 years of age). However, remifentanil blood concentrations fall as rapidly after termination of administration in the elderly as in young adults.
This drug is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
8.6 Use in Morbidly Obese Patients
As for all potent opioids, caution is required with use in morbidly obese patients because of alterations in cardiovascular and respiratory physiology [see Dosage and Administration (2.2)].
9. Drug Abuse and Dependence
9.2 Abuse
ULTIVA contains remifentanil, a substance with high potential for misuse and abuse, which can lead to the development of substance use disorder, including addiction [see Warnings and Precautions (5.1)].
Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a healthcare provider or for whom it was not prescribed.
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects.
Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence.
Misuse and abuse of ULTIVA increases risk of overdosage, which may lead to central nervous system and respiratory depression, hypotension, seizures, and death. The risk is increased with concurrent abuse of ULTIVA with alcohol and/or other CNS depressants. Abuse of and addiction to opioids in some individuals may not be accompanied by concurrent tolerance and symptoms of physical dependence. In addition, abuse of opioids can occur in the absence of addiction.
All patients treated with opioids require careful and frequent reevaluation for signs of misuse, abuse, and addiction, because use of opioid analgesic products carries the risk of addiction even under appropriate medical use. Patients at high risk of ULTIVA abuse include those with a history of prolonged use of any opioid, including products containing remifentanil, those with a history of drug or alcohol abuse, or those who use ULTIVA in combination with other abused drugs.
“Drug-seeking” behavior is very common in persons with substance use disorders. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing, or referral, repeated “loss” of prescriptions, tampering with prescriptions, and reluctance to provide prior medical records or contact information for other treating healthcare provider(s). “Doctor shopping” (visiting multiple prescribers to obtain additional prescriptions) is common among people who abuse drugs and people with substance use disorder. Preoccupation with achieving adequate pain relief can be appropriate behavior in a patient with inadequate pain control.
ULTIVA, like other opioids, can be diverted for nonmedical use into illicit channels of distribution. Careful record-keeping of prescribing information, including quantity, frequency, and renewal requests, as required by state and federal law, is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic reevaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
9.3 Dependence
Both tolerance and physical dependence can develop during use of opioid therapy.
Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose).
Physical dependence is a state that develops as a result of a physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dosage reduction of a drug.
Withdrawal may be precipitated through the administration of drugs with opioid antagonist activity (e.g., naloxone), mixed agonist/antagonist analgesics (e.g., pentazocine, butorphanol, nalbuphine), or partial agonists (e.g., buprenorphine). Physical dependence may not occur to a clinically significant degree until after several days to weeks of continued use.
ULTIVA should not be abruptly discontinued in a physically-dependent patient [see Dosage and Administration (2)]. If ULTIVA is abruptly discontinued in a physically-dependent patient, a withdrawal syndrome may occur, typically characterized by restlessness, lacrimation, rhinorrhea, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate.
Infants born to mothers physically dependent on opioids will also be physically dependent and may exhibit respiratory difficulties and withdrawal signs [see Use in Specific Populations (8.1)].
10. Overdosage
Clinical Presentation
Acute overdose with remifentanil can be manifested by respiratory depression, somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and, in some cases, pulmonary edema, bradycardia, hypotension, hypoglycemia, partial or complete airway obstruction, atypical snoring, and death. Marked mydriasis rather than miosis may be seen with hypoxia in overdose situations [see Clinical Pharmacology (12.2)].
Treatment of Overdose
In case of overdose, priorities are the reestablishment of a patent and protected airway and institution of assisted or controlled ventilation, if needed. Employ other supportive measures (including oxygen and vasopressors) in the management of circulatory shock and pulmonary edema as indicated. Cardiac arrest or arrhythmias will require advanced life-support measures.
The opioid antagonists, naloxone or nalmefene, are specific antidotes to respiratory depression resulting from opioid overdose. For clinically significant respiratory or circulatory depression secondary to remifentanil overdose, stop the infusion or administer an opioid antagonist. Opioid antagonists should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to remifentanil overdose.
In an individual physically dependent on opioids, administration of the recommended usual dosage of the antagonist will precipitate an acute withdrawal syndrome. The severity of the withdrawal symptoms experienced will depend on the degree of physical dependence and the dose of the antagonist administered. If a decision is made to treat serious respiratory depression in the physically dependent patient, administration of the antagonist should be begun with care and by titration with smaller than usual doses of the antagonist.
11. Ultiva Description
ULTIVA (remifentanil hydrochloride) for injection is an opioid agonist. The chemical name is 3-[4-methoxycarbonyl-4-[(1-oxopropyl)phenylamino]-1-piperidine]propanoic acid methyl ester, hydrochloride salt. The molecular weight is 412.91. Its molecular formula is C20H28N2O5•HCl, and it has the following chemical structure.
ULTIVA is a sterile, nonpyrogenic, preservative-free, white to off-white lyophilized powder for intravenous (IV) administration after reconstitution and dilution. Each vial contains 1 mg, 2 mg, or 5 mg of remifentanil base; 15 mg glycine; and hydrochloric acid to buffer the solutions to a nominal pH of 3 after reconstitution. When reconstituted as directed, solutions of ULTIVA are clear and colorless and contain remifentanil hydrochloride (HCl) equivalent to 1 mg/mL of remifentanil base. The pH of reconstituted solutions of ULTIVA ranges from 2.5 to 3.5. Remifentanil hydrochloride has a pKa of 7.07. Remifentanil hydrochloride has an n-octanol:water partition coefficient of 17.9 at pH 7.3.
12. Ultiva - Clinical Pharmacology
12.1 Mechanism of Action
ULTIVA is a µ-opioid agonist with rapid onset and peak effect, and short duration of action. The µ-opioid activity of ULTIVA is antagonized by opioid antagonists such as naloxone.
Unlike other opioids, ULTIVA is rapidly metabolized by hydrolysis of the propanoic acid-methyl ester linkage by nonspecific blood and tissue esterases. ULTIVA is not a substrate for plasma cholinesterase (pseudocholinesterase) and, therefore, patients with atypical cholinesterase are expected to have a normal duration of action.
12.2 Pharmacodynamics
The analgesic effects of ULTIVA are rapid in onset and offset. Its effects and side effects are dose dependent and similar to other µ-opioids. ULTIVA in humans has a rapid blood-brain equilibration half-time of 1 ± 1 minutes (mean ± SD) and a rapid onset of action. The pharmacodynamic effects of ULTIVA closely follow the measured blood concentrations, allowing direct correlation between dose, blood levels, and response. Blood concentration decreases 50% in 3 to 6 minutes after a 1-minute infusion or after prolonged continuous infusion due to rapid distribution and elimination processes and is independent of duration of drug administration. Recovery from the effects of ULTIVA occurs rapidly (within 5 to 10 minutes). New steady-state concentrations occur within 5 to 10 minutes after changes in infusion rate. When used as a component of an anesthetic technique, ULTIVA can be rapidly titrated to the desired depth of anesthesia/analgesia (e.g., as required by varying levels of intraoperative stress) by changing the continuous infusion rate or by administering an IV bolus injection.
Effects on the Central Nervous System
Remifentanil produces respiratory depression by direct action on brain stem respiratory centers. The respiratory depression involves both a reduction in the responsiveness of the brain stem respiratory centers to increases in carbon dioxide tension and to electrical stimulation.
Remifentanil causes miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origins may produce similar findings). Marked mydriasis rather than miosis may be seen due to hypoxia in overdose situations.
Effects on the Gastrointestinal Tract and Other Smooth Muscle
Remifentanil causes a reduction in motility associated with an increase in smooth muscle tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone may be increased to the point of spasm resulting in constipation. Other opioid-induced effects may include a reduction in biliary and pancreatic secretions, spasm of sphincter of Oddi, and transient elevations in serum amylase.
Effects on the Cardiovascular System
Remifentanil produces peripheral vasodilation which may result in orthostatic hypotension or syncope. Manifestations of histamine release and/or peripheral vasodilation may include pruritus, flushing, red eyes and sweating and/or orthostatic hypotension. Caution must be used in hypovolemic patients, such as those suffering acute myocardial infarction, because remifentanil may cause or further aggravate their hypotension. Caution must also be used in patients with cor pulmonale who have received therapeutic doses of opioids.
Effects on the Endocrine System
Opioids inhibit the secretion of adrenocorticotropic hormone (ACTH), cortisol, and luteinizing hormone (LH) in humans. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon.
Effects on the Immune System
Opioids have been shown to have a variety of effects on components of the immune system in in vitro and animal models. The clinical significance of these findings is unknown. Overall, the effects of opioids appear to be modestly immunosuppressive.
Concentration–Efficacy Relationships
The minimum effective analgesic concentration will vary widely among patients, especially among patients who have been previously treated with opioid agonists [see Dosage and Administration (2.1, 2.2)]. The minimum effective analgesic concentration of remifentanil for any individual patient may increase over time due to an increase in pain, the development of a new pain syndrome and/or the development of analgesic tolerance.
Concentration–Adverse Reaction Relationships
There is a relationship between increasing remifentanil plasma concentration and increasing frequency of dose-related opioid adverse reactions such as nausea, vomiting, CNS effects, and respiratory depression. In opioid-tolerant patients, the situation may be altered by the development of tolerance to opioid-related adverse reactions [see Dosage and Administration (2.1, 2.2)].
Hemodynamics
In premedicated patients undergoing anesthesia, 1-minute infusions of < 2 mcg/kg of ULTIVA cause dose-dependent hypotension and bradycardia. While additional doses > 2 mcg/kg (up to 30 mcg/kg) do not produce any further decreases in heart rate or blood pressure, the duration of the hemodynamic change is increased in proportion to the blood concentrations achieved. Peak hemodynamic effects occur within 3 to 5 minutes of a single dose of ULTIVA or an infusion rate increase. Glycopyrrolate, atropine, and vagolytic neuromuscular blocking agents attenuate the hemodynamic effects associated with ULTIVA. When appropriate, bradycardia and hypotension can be reversed by reduction of the rate of infusion of ULTIVA, or the dose of concurrent anesthetics, or by the administration of fluids or vasopressors.
Respiration
ULTIVA depresses respiration in a dose-related fashion. Unlike other fentanyl analogs, the duration of action of ULTIVA at a given dose does not increase with increasing duration of administration, due to lack of drug accumulation. When ULTIVA and alfentanil were dosed to equal levels of respiratory depression, recovery of respiratory drive after 3-hour infusions was more rapid and less variable with ULTIVA (see Figure 1).
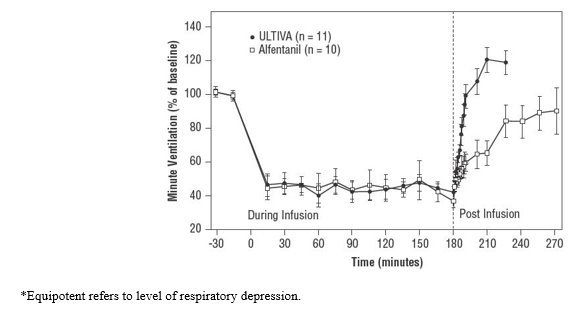
Figure 1: Recovery of Respiratory Drive After Equipotent* Doses of ULTIVA and Alfentanil Using CO2-Stimulated Minute Ventilation in Adult Volunteers (±1.5 SEM)
Spontaneous respiration occurs at blood concentrations of 4 to 5 ng/mL in the absence of other anesthetic agents; for example, after discontinuation of a 0.25 mcg/kg/min infusion of remifentanil, these blood concentrations would be reached in 2 to 4 minutes. In patients undergoing general anesthesia, the rate of respiratory recovery depends upon the concurrent anesthetic; N2O < propofol < isoflurane [see Clinical Studies: Recovery (14.2)].
Muscle Rigidity
Skeletal muscle rigidity can be caused by ULTIVA and is related to the dose and speed of administration. ULTIVA may cause chest wall rigidity (inability to ventilate) after single doses of > 1 mcg/kg administered over 30 to 60 seconds or infusion rates > 0.1 mcg/kg/min; peripheral muscle rigidity may occur at lower doses. Administration of doses < 1 mcg/kg may cause chest wall rigidity when given concurrently with a continuous infusion of ULTIVA.
Histamine Release
Assays of histamine in patients and normal volunteers have shown no elevation in plasma histamine levels after administration of ULTIVA in doses up to 30 mcg/kg over 60 seconds.
Analgesia
Infusions of 0.05 to 0.1 mcg/kg/min, producing blood concentrations of 1 to 3 ng/mL, are typically associated with analgesia with minimal decrease in respiratory rate. Supplemental doses of 0.5 to 1 mcg/kg, incremental increases in infusion rate > 0.05 mcg/kg/min, and blood concentrations exceeding 5 ng/mL (typically produced by infusions of 0.2 mcg/kg/min) have been associated with transient and reversible respiratory depression, apnea, and muscle rigidity.
Anesthesia
ULTIVA is synergistic with the activity of hypnotics (propofol and thiopental), inhaled anesthetics, and benzodiazepines [see Clinical Studies (14.1), Warnings and Precautions (5), and Dosage and Administration (2)].
Age
The pharmacodynamic activity of ULTIVA (as measured by the EC50 for development of delta waves on the EEG) increases with increasing age. The EC50 of remifentanil for this measure was 50% less in patients over 65 years of age when compared to healthy volunteers (25 years of age) [see Dosage and Administration (2.2)].
Sex
No differences have been shown in the pharmacodynamic activity (as measured by the EEG) of ULTIVA between men and women.
Drug Interactions
In animals the duration of muscle paralysis from succinylcholine is not prolonged by remifentanil.
Intraocular Pressure
There was no change in intraocular pressure after the administration of ULTIVA prior to ophthalmic surgery under monitored anesthesia care.
Cerebrodynamics
Under isoflurane-nitrous oxide anesthesia (PaCO2 < 30 mmHg), a 1-minute infusion of ULTIVA (0.5 or 1.0 mcg/kg) produced no change in intracranial pressure. Mean arterial pressure and cerebral perfusion decreased as expected with opioids. In patients receiving ULTIVA and nitrous oxide anesthesia, cerebrovascular reactivity to carbon dioxide remained intact. In humans, no epileptiform activity was seen on the EEG (n = 44) at remifentanil doses up to 8 mcg/kg/min.
12.3 Pharmacokinetics
After IV doses administered over 60 seconds, the pharmacokinetics of remifentanil fit a three-compartment model with a rapid distribution half-life of one minute, a slower distribution half-life of 6 minutes, and a terminal elimination half-life of 10 to 20 minutes. Since the terminal elimination component contributes less than 10% of the overall area under the concentration versus time curve (AUC), the effective biological half-life of ULTIVA is 3 to 10 minutes. This is similar to the 3- to 10-minute half-life measured after termination of prolonged infusions (up to 4 hours; see Figure 2) and correlates with recovery times observed in the clinical setting after infusions up to 12 hours. Concentrations of remifentanil are proportional to the dose administered throughout the recommended dose range. The pharmacokinetics of remifentanil are unaffected by the presence of renal or hepatic impairment.
Distribution
The initial volume of distribution (Vd) of remifentanil is approximately 100 mL/kg and represents distribution throughout the blood and rapidly perfused tissues. Remifentanil subsequently distributes into peripheral tissues with a steady-state volume of distribution of approximately 350 mL/kg. These two distribution volumes generally correlate with total body weight (except in severely obese patients when they correlate better with ideal body weight [IBW]). Remifentanil is approximately 70% bound to plasma proteins of which two-thirds is binding to alpha-1-acid-glycoprotein.
Elimination
The clearance of remifentanil in young, healthy adults is approximately 40 mL/min/kg. Clearance generally correlates with total body weight (except in severely obese patients when it correlates better with IBW). The high clearance of remifentanil combined with a relatively small volume of distribution produces a short elimination half-life of approximately 3 to 10 minutes (see Figure 2). This value is consistent with the time taken for blood or effect site concentrations to fall by 50% (context-sensitive half-times) which is approximately 3 to 6 minutes. Unlike other fentanyl analogs, the duration of action does not increase with prolonged administration.
Titration to Effect
The rapid elimination of remifentanil permits the titration of infusion rate without concern for prolonged duration. In general, every 0.1 mcg/kg/min change in the IV infusion rate will lead to a corresponding 2.5 ng/mL change in blood remifentanil concentration within 5 to 10 minutes. In intubated patients only, a more rapid increase (within 3 to 5 minutes) to a new steady state can be achieved with a 1.0 mcg/kg bolus dose in conjunction with an infusion rate increase.
Metabolism
Remifentanil is an esterase-metabolized opioid. A labile ester linkage renders this compound susceptible to hydrolysis by nonspecific esterases in blood and tissues. This hydrolysis results in the production of the carboxylic acid metabolite (3-[4-methoxycarbonyl-4-[(1-oxopropyl)phenylamino]-1-piperidine]propanoic acid), and represents the principal metabolic pathway for remifentanil (> 95%). The carboxylic acid metabolite is essentially inactive (1/4600 as potent as remifentanil in dogs). Remifentanil is not metabolized by plasma cholinesterase (pseudocholinesterase) and is not appreciably metabolized by the liver or lung.
Specific Populations
Age: Geriatric Population
The clearance of remifentanil is reduced (approximately 25%) in the elderly (> 65 years of age) compared to young adults (average 25 years of age). However, remifentanil blood concentrations fall as rapidly after termination of administration in the elderly as in young adults.
Age: Pediatric Population
In pediatric patients, 5 days to 17 years of age (n = 47), the clearance and volume of distribution of remifentanil were increased in younger children and declined to young healthy adult values by age 17. The average clearance of remifentanil in neonates (less than 2 months of age) was approximately 90.5 ± 36.8 mL/min/kg (mean ± SD) while in adolescents (13 to 16 years) this value was 57.2 ± 21.1 mL/min/kg. The total (steady-state) volume of distribution in neonates was 452 ± 144 mL/kg versus 223 ± 30.6 mL/kg in adolescents. The half-life of remifentanil was the same in neonates and adolescents. Clearance of remifentanil was maintained at or above normal adult values in patients 5 days to 17 years of age.
Sex
There is no significant difference in the pharmacokinetics of remifentanil in male and female patients after correcting for differences in weight.
Hepatic Impairment
The pharmacokinetics of remifentanil and its carboxylic acid metabolite are unchanged in patients with severe hepatic impairment.
Renal Impairment
The pharmacokinetic profile of ULTIVA is not changed in patients with end stage renal disease (creatinine clearance < 10 mL/min). In anephric patients, the half-life of the carboxylic acid metabolite increases from 90 minutes to 30 hours. The metabolite is removed by hemodialysis with a dialysis extraction ratio of approximately 30%.
Drug Interaction Studies
Remifentanil clearance is not altered by concomitant administration of thiopental, isoflurane, propofol, or temazepam during anesthesia. In vitro studies with atracurium, mivacurium, esmolol, echothiophate, neostigmine, physostigmine, and midazolam revealed no inhibition of remifentanil hydrolysis in whole human blood by these drugs.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term studies in animals to evaluate the carcinogenic potential of remifentanil have not been conducted.
Mutagenesis
Mutagenicity was observed with remifentanil in the in vitro mouse lymphoma assay in the presence but not
absence of metabolic activation. Remifentanil did not induce gene mutation in the in vitro bacterial reverse mutation assay (Ames test) and was not genotoxic in the in vivo rat hepatocyte unscheduled DNA synthesis assay. No clastogenic effect was seen in cultured Chinese hamster ovary cells or in the in vivo mouse micronucleus test.
Impairment of Fertility
Remifentanil has been shown to reduce fertility in male rats when tested after 70+ days of daily IV administration of 0.5 mg/kg, which is approximately 0.2 times a human intravenous infusion of an induction dose of 1 mcg/kg with a maintenance dose of 2 mcg/kg/min in terms of mg/m2 of body surface area for a surgical procedure lasting 3 hours or 40 times a single bolus human dose of 2 mcg/kg, in terms of mg/m2 of body surface area.
The fertility of female rats was not affected at IV doses as high as 1 mg/kg which is 0.4 times a human intravenous infusion of an induction dose of 1 mcg/kg with a maintenance dose of 2 mcg/kg/min in terms of mg/m2 of body surface area for a surgical procedure lasting 3 hours or approximately 80 times a single bolus human dose of 2 mcg/kg, in terms of mg/m2 of body surface area, when administered for at least 15 days before mating.
14. Clinical Studies
ULTIVA was evaluated in 3,341 patients undergoing general anesthesia (n = 2,706) and monitored anesthesia care (n = 639). These patients were evaluated in the following settings: inpatient (n = 2,079) which included cardiovascular (n = 426), and neurosurgical (n = 61), and outpatient (n = 1,349). Four-hundred and eighty-six (486) elderly patients (age range 66 to 90 years) and 410 pediatric patients (age range birth to 12 years) received ULTIVA. Of the general anesthesia patients, 682 also received ULTIVA as an IV analgesic agent during the immediate postoperative period.
14.1 Induction and Maintenance of General Anesthesia - Inpatient/Outpatient
The efficacy of ULTIVA was investigated in 1,562 patients in 15 randomized, controlled trials as the analgesic component for the induction and maintenance of general anesthesia. Eight of these studies compared ULTIVA to alfentanil and two studies compared ULTIVA to fentanyl. In these studies, doses of ULTIVA up to the ED90 were compared to recommended doses (approximately ED50) of alfentanil or fentanyl.
Induction of Anesthesia
ULTIVA was administered with isoflurane, propofol, or thiopental for the induction of anesthesia (n = 1,562). The majority of patients (80%) received propofol as the concurrent agent. ULTIVA reduced the propofol and thiopental requirements for loss of consciousness. Compared to alfentanil and fentanyl, a higher relative dose of ULTIVA resulted in fewer responses to intubation (see Table 19). Overall, hypotension occurred in 5% of patients receiving ULTIVA compared to 2% of patients receiving the other opioids.
ULTIVA has been used as a primary agent for the induction of anesthesia; however, it should not be used as a sole agent because loss of consciousness cannot be assured and because of a high incidence of apnea, muscle rigidity, and tachycardia. The administration of an induction dose of propofol or thiopental or a paralyzing dose of a muscle relaxant prior to or concurrently with ULTIVA during the induction of anesthesia markedly decreased the incidence of muscle rigidity from 20% to < 1%.
|
Opioid |
Initial Dose |
Pre-Intubation Infusion Rate |
No. (%) Muscle Rigidity |
No. (%) |
No. (%) Response to Intubation |
|
Study 1: | |||||
|
ULTIVA (35) |
1 |
0.1 |
1 (3%) |
0 |
27 (77%) |
|
ULTIVA (35) |
1 |
0.4 |
3 (9%) |
0 |
11 (31%)† |
|
Alfentanil (35) |
20 |
1.0 |
2 (6%) |
0 |
26 (74%) |
|
Study 2: | |||||
|
ULTIVA (116) |
1 |
0.5 |
9 (8%) |
5 (4%) |
17 (15%)† |
|
Alfentanil (118) |
25 |
1.0 |
6 (5%) |
5 (4%) |
33 (28%) |
|
Study 3: | |||||
|
ULTIVA (134) |
1 |
0.5 |
2 (1%) |
4 (3%) |
25 (19%) |
|
Alfentanil (66) |
20 |
2.0 |
0 |
0 |
19 (29%) |
|
Study 4: | |||||
|
ULTIVA (98) |
1 |
0.2 |
11 (11%)† |
2 (2%) |
35 (36%) |
|
ULTIVA (91) |
2‡ |
0.4 |
11 (12%)† |
2 (2%) |
12 (13%)† |
|
Fentanyl (97) |
3 |
NA |
1 (1%) |
1 (1%) |
29 (30%) |
Use During Maintenance of Anesthesia
ULTIVA was investigated in 929 patients in seven well controlled general surgery studies in conjunction with nitrous oxide, isoflurane, or propofol in both inpatient and outpatient settings. These studies demonstrated that ULTIVA could be dosed to high levels of opioid effect and rapidly titrated to optimize analgesia intraoperatively without delaying or prolonging recovery.
Compared to alfentanil and fentanyl, these higher relative doses (ED90) of ULTIVA resulted in fewer responses to intraoperative stimuli (see Table 20) and a higher frequency of hypotension (16% compared to 5% for the other opioids). ULTIVA was infused to the end of surgery, while alfentanil was discontinued 5 to 30 minutes before the end of surgery as recommended. The mean final infusion rates of ULTIVA were between 0.25 and 0.48 mcg/kg/min.
|
Opioid Treatment Group/(No. of Patients) |
Concurrent Anesthetic |
Post-Intubation Infusion |
No. (%) With Intraoperative Hypotension |
No. (%) With Response |
No. (%) With Signs of Light Anesthesia |
No. (%) With Response to Skin Closure |
|
Study 1: | ||||||
|
ULTIVA (35) |
0.1 |
0 |
20 (57%) |
33 (94%) |
6 (17%) |
|
|
ULTIVA (35) |
Nitrous oxide |
0.4 |
0 |
3 (9%)† |
12 (34%)† |
2 (6%)† |
|
Alfentanil (35) |
1.0 |
0 |
24 (69%) |
33 (94%) |
12 (34%) |
|
|
Study 2: | ||||||
|
ULTIVA (116) |
Isoflurane + |
0.25 |
35 (30%)† |
9 (8%)† |
66 (57%)† |
19 (16%) |
|
Alfentanil (118) |
Nitrous oxide |
0.5 |
12 (10%) |
20 (17%) |
85 (72%) |
25 (21%) |
|
Study 3: | ||||||
|
ULTIVA (134) |
Propofol |
0.5 |
3 (2%) |
14 (11%)† |
70 (52%)† |
25 (19%) |
|
Alfentanil (66) |
2.0 |
2 (3%) |
21 (32%) |
47 (71%) |
13 (20%) |
|
|
Study 4: | ||||||
|
ULTIVA (98) |
0.2 |
13 (13%) |
12 (12%)† |
67 (68%)† |
7 (7%) |
|
|
ULTIVA (91) |
Isoflurane |
0.4 |
16 (18%)† |
4 (4%)† |
44 (48%)† |
3 (3%)† |
|
Fentanyl (97) |
1.5-3 mcg/kg prn |
7 (7%) |
32 (33%) |
84 (87%) |
11 (11%) |
|
In three randomized, controlled studies (n = 407) during general anesthesia, ULTIVA attenuated the signs of light anesthesia within a median time of 3 to 6 minutes after bolus doses of 1 mcg/kg with or without infusion rate increases of 50% to 100% (up to a maximum rate of 2 mcg/kg/min).
In an additional double-blind, randomized study (n = 103), a constant rate (0.25 mcg/kg/min) of ULTIVA was compared to doubling the rate to 0.5 mcg/kg/min approximately 5 minutes before the start of the major surgical stress event. Doubling the rate decreased the incidence of signs of light anesthesia from 67% to 8% in patients undergoing abdominal hysterectomy, and from 19% to 10% in patients undergoing radical prostatectomy. In patients undergoing laminectomy the lower dose was adequate.
14.2 Recovery
In 2,169 patients receiving ULTIVA for periods up to 16 hours, recovery from anesthesia was rapid, predictable, and independent of the duration of the infusion of ULTIVA. In the seven controlled, general surgery studies, extubation occurred in a median of 5 minutes (range: -3 to 17 minutes in 95% of patients) in outpatient anesthesia and 10 minutes (range: 0 to 32 minutes in 95% of patients) in inpatient anesthesia. Recovery in studies using nitrous oxide or propofol was faster than in those using isoflurane as the concurrent anesthetic. There was no case of remifentanil-induced delayed respiratory depression occurring more than 30 minutes after discontinuation of remifentanil [see Warnings and Precautions (5.15)].
In a double-blind, randomized study, administration of morphine sulfate (0.15 mg/kg) intravenously 20 minutes before the anticipated end of surgery to 98 patients did not delay recovery of respiratory drive in patients undergoing major surgery with remifentanil-propofol total IV anesthesia.
14.3 Spontaneous Ventilation Anesthesia
Two randomized, dose-ranging studies (n = 127) examined the administration of ULTIVA to outpatients undergoing general anesthesia with a laryngeal mask. Starting infusion rates of ULTIVA of ≤ 0.05 mcg/kg/min provided supplemental analgesia while allowing spontaneous ventilation with propofol or isoflurane. Bolus doses of ULTIVA during spontaneous ventilation lead to transient periods of apnea, respiratory depression, and muscle rigidity.
14.4 Pediatric Anesthesia
ULTIVA has been evaluated for maintenance of general anesthesia in 410 pediatric patients from birth to 12 years undergoing inpatient and outpatient procedures. Four clinical studies have been performed.
Study 1, an open-label, randomized, controlled clinical trial (n = 129), compared ULTIVA (n = 68) with alfentanil (n = 19), isoflurane (n = 22), or propofol (n = 20) in children 2 to 12 years of age undergoing strabismus surgery. After induction of anesthesia which included the administration of atropine, ULTIVA was administered as an initial infusion of 1 mcg/kg/min with 70% nitrous oxide. The infusion rate required during maintenance of anesthesia was 0.73 to 1.95 mcg/kg/min. Time to extubation and to purposeful movement was a median of 10 minutes (range 1 to 24 minutes).
Study 2, a double-blind, randomized, controlled trial (n = 222), compared ULTIVA (n = 119) to fentanyl (n = 103) in children 2 to 12 years of age undergoing tonsillectomy with or without adenoidectomy. After induction of anesthesia, patients received a 0.25 mcg/kg/min infusion of ULTIVA or fentanyl by IV bolus with nitrous oxide/oxygen (2:1) and either halothane or sevoflurane for maintenance of anesthesia. The mean infusion rate required during maintenance of anesthesia was 0.3 mcg/kg/min (range 0.2 to 1.3 mcg/kg/min). The continuous infusion rate was decreased to 0.05 mcg/kg/min approximately 10 minutes prior to the end of surgery. Time to spontaneous purposeful movement was a median of 8 minutes (range 1 to 19 minutes). Time to extubation was a median of 9 minutes (range 2 to 19 minutes).
Study 3, an open-label, randomized, controlled trial (n = 271), compared ULTIVA (n = 185) with a regional anesthetic technique (n = 86) in children 1 to 12 years of age undergoing major abdominal, urological, or orthopedic surgery. Patients received a 0.25 mcg/kg/min infusion of ULTIVA following a 1.0 mcg/kg bolus or bupivacaine by epidural infusion, along with isoflurane and nitrous oxide after the induction of anesthesia. The mean infusion rate required during maintenance of anesthesia was 0.25 mcg/kg/min (range 0 to 0.75 mcg/kg/min). Both treatments were effective in attenuating responses to skin incision during surgery. The hemodynamic profile of the ULTIVA group was consistent with an opioid-based general anesthetic technique. Time to spontaneous purposeful movement was a median of 15 minutes (range, 2 to 75 minutes) in the remifentanil group. Time to extubation was a median of 13 minutes (range, 4 to 31 minutes) in the remifentanil group.
Study 4, an open-label, randomized, controlled trial (n = 60), compared ULTIVA (n = 38) with halothane (n = 22) in ASA 1 or 2, full term neonates and infants ≤ 8 weeks of age weighing at least 2500 grams who were undergoing pyloromyotomy. After induction of anesthesia, which included the administration of atropine, patients received 0.4 mcg/kg/min of ULTIVA or 0.4% halothane with 70% nitrous oxide for initial maintenance of anesthesia and then both agents were adjusted according to clinical response. Bolus doses of 1 mcg/kg administered over 30 to 60 seconds were used to treat brief episodes of hypertension and tachycardia, and infusion rates were increased by 50% to treat sustained hypertension and tachycardia. The range of infusion rates of ULTIVA required during maintenance of anesthesia was 0.4 to 1 mcg/kg/min.
Seventy-one percent (71%) of ULTIVA patients required supplementary boluses or rate increases from the starting dose of 0.4 mcg/kg/min to treat hypertension, tachycardia, movement or somatic signs of light anesthesia. Twenty-four percent of the patients required an increase from the initial rate of 0.4 mcg/kg/min prior to incision and 26% of patients required an infusion rate between 0.8 and 1.0 mcg/kg/min, most often during gastric manipulation. The continuous infusion rate was decreased to 0.05 mcg/kg/min approximately 10 minutes before the end of surgery.
In the ULTIVA group, median time from discontinuation of anesthesia to spontaneous purposeful movement was 6.5 minutes (range, 1 to 13 minutes) and median time to extubation was 8.5 minutes (range, 1 to 14 minutes).
The initial maintenance infusion regimen of ULTIVA evaluated in pediatric patients from birth to 2 months of age was 0.4 mcg/kg/min, the approved adult regimen for use with N2O. The clearance rate observed in the neonatal population was highly variable and on average was two times higher than in the young healthy adult population. [See Clinical Pharmacology: Specific Populations: Pediatric Population (12.3) and Dosage and Administration, Table 2 (2.2).]
No pediatric patients receiving ULTIVA required naloxone during the immediate postoperative recovery period.
14.5 Coronary Artery Bypass Surgery
ULTIVA was originally administered to 225 subjects undergoing elective CABG surgery in two dose-ranging studies without active comparators. Subsequently, two double-blind, double-dummy clinical studies (N = 426) evaluated ULTIVA (n = 236) at recommended doses versus active comparators (n = 190).
The first comparator study, a multi-center, randomized, double-blind, double-dummy, parallel-group study (N = 369), compared ULTIVA (n = 201) with fentanyl (n = 168) in adult patients undergoing elective CABG surgery. Subjects received 1 to 3 mg midazolam and 0.05 mg/kg morphine IV as premedication. Anesthesia was induced with propofol 0.5 mg/kg (higher doses administered with ULTIVA were associated with excessive hypotension) over one minute plus 10-mg boluses every 10 seconds until loss of consciousness followed by either cisatracurium 0.2 mg/kg or vecuronium 0.15 mg/kg. Patients randomized to ULTIVA received a 1 mcg/kg/min infusion of ULTIVA followed by a placebo bolus administered over 3 minutes. In the active control group, a placebo IV infusion was started and a fentanyl bolus 10 mcg/kg was administered over 3 minutes. All subjects received isoflurane titrated initially to end tidal concentration of 0.5%. During maintenance, the group randomized to ULTIVA received as needed 0.5-1 mcg/kg/min IV rate increases (to a maximum of 4 mcg/kg/min) of ULTIVA and 1 mcg/kg IV boluses of ULTIVA. The active control group received 2 mcg/kg IV boluses of fentanyl and increases in placebo IV infusion rate.
The second comparator study, a multi-center, double-blind, randomized, parallel group study (N = 57), compared ULTIVA (n = 35) to fentanyl (n = 22) in adult patients undergoing elective CABG surgery with poor left ventricular function (ejection fraction < 0.35). Subjects received oral lorazepam 40 mcg/kg as premedication. Anesthesia was induced using etomidate until loss of consciousness, followed by a low-dose propofol infusion (3 mg/kg/hr) and pancuronium 0.15 mg/kg. Subjects in the group administered ULTIVA received a placebo bolus dose and a continuous infusion of ULTIVA 1 mcg/kg/min and subjects in the fentanyl group received a bolus loading dose of 15 mcg/kg and placebo continuous infusion. During maintenance, supplemental bolus doses of ULTIVA (0.5 mcg/kg) and infusion rate increases of 0.5 to 1 mcg/kg/min (maximum rate allowed was 4 mcg/kg/min) of ULTIVA were administered to one group; while the fentanyl group was given intermittent maintenance bolus doses of 2 mcg/kg and increases in the placebo infusion rate.
In these two studies, using a high dose opioid technique with ULTIVA as a component of a balanced or total intravenous anesthetic regimen, the remifentanil regimen effectively attenuated response to maximal sternal spread generally better than the dose and regimen studied for the active control (fentanyl). While this provides evidence for the efficacy of remifentanil as an analgesic in this setting, caution must be exercised in interpreting these results as evidence of superiority of remifentanil over the active control, since these studies did not make any attempt to evaluate and compare the optimal analgesic doses of either drug in this setting.
14.6 Neurosurgery
ULTIVA was administered to 61 patients undergoing craniotomy for removal of a supratentorial mass lesion. In these studies, ventilation was controlled to maintain a predicted PaCO2 of approximately 28 mmHg. In one study (n = 30) with ULTIVA and 66% nitrous oxide, the median time to extubation and to patient response to verbal commands was 5 minutes (range -1 to 19 minutes). Intracranial pressure and cerebrovascular responsiveness to carbon dioxide were normal [see Clinical Pharmacology (12.2)].
A randomized, controlled study compared ULTIVA (n = 31) to fentanyl (n = 32). ULTIVA (1 mcg/kg/min) and fentanyl (2 mcg/kg/min) were administered after induction with thiopental and pancuronium. A similar number of patients (6%) receiving ULTIVA and fentanyl had hypotension during induction. Anesthesia was maintained with nitrous oxide and ULTIVA at a mean infusion rate of 0.23 mcg/kg/min (range 0.1 to 0.4) compared with a fentanyl mean infusion rate of 0.04 mcg/kg/min (range 0.02 to 0.07). Supplemental isoflurane was administered as needed. The patients receiving ULTIVA required a lower mean isoflurane dose (0.07 MAC-hours) compared with 0.64 MAC-hours for the fentanyl patients (P = 0.04). ULTIVA was discontinued at the end of anesthesia, whereas fentanyl was discontinued at the time of bone flap replacement (a median time of 44 minutes before the end of surgery). Median time to extubation was similar (5 and 3.5 minutes, respectively, with ULTIVA and fentanyl). None of the patients receiving ULTIVA required naloxone compared to seven of the fentanyl patients (P = 0.01). Eighty-one percent (81%) of patients receiving ULTIVA recovered (awake, alert, and oriented) within 30 minutes after surgery compared with 59% of fentanyl patients (P = 0.06). At 45 minutes, recovery rates were similar (81% and 69% respectively for ULTIVA and fentanyl, P = 0.27). Patients receiving ULTIVA required an analgesic for headache sooner than fentanyl patients (median of 35 minutes compared with 136 minutes, respectively [P = 0.04]). No adverse cerebrovascular effects were seen in this study [see Clinical Pharmacology (12.2)].
14.7 Continuation of Analgesic Use into the Immediate Postoperative Period
Analgesia with ULTIVA in the immediate postoperative period (until approximately 30 minutes after extubation) was studied in 401 patients in four dose-finding studies and in 281 patients in two efficacy studies. In the dose-finding studies, the use of bolus doses of ULTIVA and incremental infusion rate increases ≥ 0.05 mcg/kg/min led to respiratory depression and muscle rigidity.
In two efficacy studies, ULTIVA 0.1 mcg/kg/min was started immediately after discontinuing anesthesia. Incremental infusion rate increases of 0.025 mcg/kg/min every 5 minutes were given to treat moderate to severe postoperative pain. In Study 1, 50% decreases in infusion rate were made if respiratory rate decreased below 12 breaths/min and in Study 2, the same decreases were made if respiratory rate was below 8 breaths/min. With this difference in criteria for infusion rate decrease, the incidence of respiratory depression was lower in Study 1 (4%) than in Study 2 (12%). In both studies, ULTIVA provided effective analgesia (no or mild pain with respiratory rate ≥ 8 breaths/min) in approximately 60% of patients at mean final infusion rates of 0.1 to 0.125 mcg/kg/min.
Study 2 was a double-blind, randomized, controlled study in which patients received either morphine sulfate (0.15 mg/kg administered 20 minutes before the anticipated end of surgery plus 2 mg bolus doses for supplemental analgesia) or ULTIVA (as described above). Emergence from anesthesia was similar between groups; median time to extubation was 5 to 6 minutes for both. ULTIVA provided effective analgesia in 58% of patients compared to 33% of patients who received morphine. Respiratory depression occurred in 12% of patients receiving ULTIVA compared to 4% of morphine patients. For patients who received ULTIVA, morphine sulfate (0.15 mg/kg) was administered in divided doses 5 and 10 minutes before discontinuing ULTIVA. Within 30 minutes after discontinuation of ULTIVA, the percentage of patients with effective analgesia decreased to 34%.
14.8 Monitored Anesthesia Care
ULTIVA has been studied in the monitored anesthesia care setting in 609 patients in eight clinical studies. Nearly all patients received supplemental oxygen in these studies. Two early dose-finding studies demonstrated that use of sedation as an endpoint for titration of ULTIVA led to a high incidence of muscle rigidity (69%) and respiratory depression. Subsequent trials titrated ULTIVA to specific clinical endpoints of patient comfort, analgesia, and adequate respiration (respiratory rate > 8 breaths/min) with a corresponding lower incidence of muscle rigidity (3%) and respiratory depression. With doses of midazolam > 2 mg (4 to 8 mg), the dose of ULTIVA could be decreased by 50%, but the incidence of respiratory depression rose to 32%.
The efficacy of a single dose of ULTIVA (1.0 mcg/kg over 30 seconds) was compared to alfentanil (7 mcg/kg over 30 seconds) in patients undergoing ophthalmic surgery. More patients receiving ULTIVA were pain free at the time of the nerve block (77% versus 44%, P = 0.02) and more experienced nausea (12% versus 4%) than those receiving alfentanil.
In a randomized, controlled study (n = 118), ULTIVA 0.5 mcg/kg over 30 to 60 seconds followed by a continuous infusion of 0.1 mcg/kg/min, was compared to a propofol bolus (500 mcg/kg) followed by a continuous infusion (50 mcg/kg/min) in patients who received a local or regional anesthetic nerve block 5 minutes later. The incidence of moderate or severe pain during placement of the block was similar between groups (2% with ULTIVA and 8% with propofol, P = 0.2) and more patients receiving ULTIVA experienced nausea (26% versus 2%, P < 0.001). The final mean infusion rate of ULTIVA was 0.08 mcg/kg/min.
In a randomized, double-blind study, ULTIVA with or without midazolam was evaluated in 159 patients undergoing superficial surgical procedures under local anesthesia. ULTIVA was administered without midazolam as a 1 mcg/kg dose over 30 seconds followed by a continuous infusion of 0.1 mcg/kg/min. In the group of patients that received midazolam, ULTIVA was administered as a 0.5 mcg/kg dose over 30 seconds followed by a continuous infusion of 0.05 mcg/kg/min and midazolam 2 mg was administered 5 minutes later. The occurrence of moderate or severe pain during the local anesthetic injection was similar between groups (16% and 20%). Other effects for ULTIVA alone and ULTIVA/midazolam were: respiratory depression with oxygen desaturation (SPO2 < 90%), 5% and 2%; nausea, 8% and 2%; and pruritus, 23% and 12%. Titration of ULTIVA resulted in prompt resolution of respiratory depression (median 3 minutes, range 0 to 6 minutes). The final mean infusion rate of ULTIVA was 0.12 mcg/kg/min (range 0.03 to 0.3) for the group receiving ULTIVA alone and 0.07 mcg/kg/min (range 0.02 to 0.2) for the group receiving ULTIVA/midazolam.
16. How is Ultiva supplied
ULTIVA (remifentanil hydrochloride) for Injection, for intravenous use, is supplied as follows:
| NDC Number | Container | Concentration | Quantity |
|---|---|---|---|
|
72078-034-01 |
3 mL Single-Dose Vial |
1 mg lyophilized powder |
Box of 10 |
|
72078-035-02 |
5 mL Single-Dose Vial |
2 mg lyophilized powder |
Box of 10 |
|
72078-036-05 |
10 mL Single-Dose Vial |
5 mg lyophilized powder |
Box of 10 |
ULTIVA should be stored at 2° to 25°C (36° to 77°F).
Discard unused portion.
17. Patient Counseling Information
Addiction, Abuse, and Misuse
Inform patients that the use of ULTIVA, even when taken as recommended, can result in addiction, abuse, and misuse, which can lead to overdose and death [see Warnings and Precautions (5.1)].
Life-Threatening Respiratory Depression
Inform patients of the risk of life-threatening respiratory depression, including information that the risk is greatest when starting ULTIVA or when the dosage is increased, and that it can occur even at recommended dosages.
Hyperalgesia and Allodynia
Advise patients to inform their healthcare provider if they experience symptoms of hyperalgesia, including worsening pain, increased sensitivity to pain, or new pain [see Warnings and Precautions (5.4), Adverse Reactions (6)].
Serotonin Syndrome
Inform patients that opioids could cause a rare but potentially life-threatening condition called serotonin syndrome resulting from concomitant administration of serotonergic drugs. Warn patients of the symptoms of serotonin syndrome and to seek medical attention right away if symptoms develop after discharge from the hospital. Instruct patients to inform their healthcare provider if they are taking, or plan to take serotonergic medications [see Warnings and Precautions (5.5), Drug Interactions (7)].
Constipation
Advise patients of the potential for severe constipation, including management instructions and when to seek medical attention [see Adverse Reactions (6), Clinical Pharmacology (12.2)].
© 2023 Viatris Inc.
ULTIVA is a registered trademark of Glaxo Group Limited, a GSK Company, licensed to the Viatris Companies.
Diprivan® is a registered trademark of Fresenius Kabi USA, LLC.
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Institutional
Galway, Ireland
MI:ULTIIJ:R9
PRINCIPAL DISPLAY PANEL – 1 mg
NDC 72078-034-01
Ultiva® CII
(remifentanil HCl)
for Injection
1 mg
Equivalent to
1 mg of remifentanil
For Intravenous Use Only
Rx only
10 x 1 mg Single-Dose Vials
Sterile.
Usual Dosage: See accompanying
prescribing information.
Store at 2° to 25°C (36° to 77°F).
To reconstitute solution, add 1 mL of diluent
per mg of remifentanil. Shake well to dissolve.
When reconstituted as directed, the solution
contains approximately 1 mg of remifentanil
activity per 1 mL.
Discard unused portion.
ULTIVA® should be diluted to a
recommended final concentration of 25,
50 or 250 mcg/mL prior to administration.
ULTIVA® should not be administered
without dilution. See package insert.
© 2022 Viatris Inc.
ULTIVA is a registered trademark
of Glaxo Group Limited, a GSK Company,
licensed to the Viatris Companies.
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Institutional
Galway, Ireland
MI:034:10C:R1
Mylan.com
PRINCIPAL DISPLAY PANEL – 2 mg
NDC 72078-035-02
Ultiva® CII
(remifentanil HCl)
for Injection
2 mg
Equivalent to
2 mg of remifentanil
For Intravenous Use Only
Rx only
10 x 2 mg Single-Dose Vials
Sterile.
Usual Dosage: See accompanying
prescribing information.
Store at 2° to 25°C (36° to 77°F).
To reconstitute solution, add 1 mL of diluent
per mg of remifentanil. Shake well to dissolve.
When reconstituted as directed, the solution
contains approximately 1 mg of remifentanil
activity per 1 mL.
Discard unused portion.
ULTIVA® should be diluted to a
recommended final concentration of 25,
50 or 250 mcg/mL prior to administration.
ULTIVA® should not be administered
without dilution. See package insert.
© 2022 Viatris Inc.
ULTIVA is a registered trademark
of Glaxo Group Limited, a GSK Company,
licensed to the Viatris Companies.
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Institutional
Galway, Ireland
MI:035:10C:R1
Mylan.com
PRINCIPAL DISPLAY PANEL – 5 mg
NDC 72078-036-05
Ultiva® CII
(remifentanil HCl)
for Injection
5 mg
Equivalent to
5 mg of remifentanil
For Intravenous Use Only
Rx only
10 x 5 mg Single-Dose Vials
Sterile.
Usual Dosage: See accompanying
prescribing information.
Store at 2° to 25°C (36° to 77°F).
To reconstitute solution, add 1 mL of diluent
per mg of remifentanil. Shake well to dissolve.
When reconstituted as directed, the solution
contains approximately 1 mg of remifentanil
activity per 1 mL.
Discard unused portion.
ULTIVA® should be diluted to a
recommended final concentration of 25,
50 or 250 mcg/mL prior to administration.
ULTIVA® should not be administered
without dilution. See package insert.
© 2022 Viatris Inc.
ULTIVA is a registered trademark
of Glaxo Group Limited, a GSK Company,
licensed to the Viatris Companies.
Manufactured for:
Mylan Institutional LLC
Morgantown, WV 26505 U.S.A.
Manufactured by:
Mylan Institutional
Galway, Ireland
MI:036:10C:R1
Mylan.com
| ULTIVA
remifentanil hydrochloride injection, powder, lyophilized, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ULTIVA
remifentanil hydrochloride injection, powder, lyophilized, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ULTIVA
remifentanil hydrochloride injection, powder, lyophilized, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Mylan Institutional LLC (790384502) |
More about Ultiva (remifentanil)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- Drug class: Opioids (narcotic analgesics)
- Breastfeeding
- En español