Epzicom Prescribing Information
Package insert / product label
Generic name: abacavir sulfate and lamivudine
Dosage form: tablet, film coated
Drug class: Antiviral combinations
Medically reviewed by Drugs.com. Last updated on Jul 2, 2023.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
- Medication Guide
Highlights of Prescribing Information
EPZICOM (abacavir and lamivudine tablets), for oral use
Initial U.S. Approval: 2004
WARNING: HYPERSENSITIVITY REACTIONS and EXACERBATIONS OF HEPATITIS B
See full prescribing information for complete boxed warning
Hypersensitivity Reactions
- •
- Serious and sometimes fatal hypersensitivity reactions have occurred with abacavir-containing products. (5.1)
- •
- Hypersensitivity to abacavir is a multi-organ clinical syndrome. (5.1)
- •
- Patients who carry the HLA-B*5701 allele are at a higher risk of experiencing a hypersensitivity reaction to abacavir. (5.1)
- •
- EPZICOM is contraindicated in patients with a prior hypersensitivity reaction to abacavir and in HLA-B*5701-positive patients. (4)
- •
- Discontinue EPZICOM as soon as a hypersensitivity reaction is suspected. Regardless of HLA-B*5701 status, permanently discontinue EPZICOM if hypersensitivity cannot be ruled out, even when other diagnoses are possible. (5.1)
- •
- Following a hypersensitivity reaction to EPZICOM, NEVER restart EPZICOM or any other abacavir-containing product. (5.1)
Exacerbations of Hepatitis B
- •
- Severe acute exacerbations of hepatitis B have been reported in patients who are co-infected with hepatitis B virus (HBV) and human immunodeficiency virus (HIV-1) and have discontinued lamivudine, a component of EPZICOM. Monitor hepatic function closely in these patients and, if appropriate, initiate anti-hepatitis B treatment. (5.2)
Indications and Usage for Epzicom
EPZICOM, a combination of abacavir and lamivudine, both nucleoside analogue HIV-1 reverse transcriptase inhibitors, is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection. (1)
Epzicom Dosage and Administration
- •
- Before initiating EPZICOM, screen for the HLA‑B*5701 allele because EPZICOM contains abacavir. (2.1)
- •
- Adults: One tablet orally once daily. (2.2)
- •
- Pediatric patients weighing at least 25 kg: One tablet daily. (2.3)
- •
- Because EPZICOM is a fixed-dose tablet and cannot be dose adjusted, EPZICOM is not recommended in patients with creatinine clearance less than 30 mL per minute or patients with hepatic impairment. (2.4, 4)
Dosage Forms and Strengths
Tablets: 600 mg of abacavir and 300 mg of lamivudine. (3)
Contraindications
Warnings and Precautions
Adverse Reactions/Side Effects
The most commonly reported adverse reactions of at least moderate intensity (incidence greater than 5%) in an adult HIV-1 clinical trial were drug hypersensitivity, insomnia, depression/depressed mood, headache/migraine, fatigue/malaise, dizziness/vertigo, nausea, and diarrhea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact ViiV Healthcare at 1-877-844-8872 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- •
- Methadone: An increased methadone dose may be required in a small number of patients. (7.1)
- •
- Sorbitol: Coadministration of lamivudine and sorbitol may decrease lamivudine concentrations; when possible, avoid chronic coadministration. (7.2)
- •
- Riociguat: The riociguat dose may need to be reduced. (7.3)
Use In Specific Populations
- •
- Lactation: Women infected with HIV should be instructed not to breastfeed due to potential for HIV transmission. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 12/2022
Related/similar drugs
Biktarvy, Descovy, Truvada, tenofovir, emtricitabine, emtricitabine / tenofovir, zidovudine
Full Prescribing Information
WARNING: HYPERSENSITIVITY REACTIONS and EXACERBATIONS OF HEPATITIS B
Hypersensitivity Reactions
Serious and sometimes fatal hypersensitivity reactions, with multiple organ involvement, have occurred with abacavir, a component of EPZICOM (abacavir and lamivudine). Patients who carry the HLA‑B*5701 allele are at a higher risk of a hypersensitivity reaction to abacavir; although, hypersensitivity reactions have occurred in patients who do not carry the HLA‑B*5701 allele [see Warnings and Precautions (5.1)].
EPZICOM is contraindicated in patients with a prior hypersensitivity reaction to abacavir and in HLA‑B*5701-positive patients [see Contraindications (4), Warnings and Precautions (5.1)]. All patients should be screened for the HLA‑B*5701 allele prior to initiating therapy with EPZICOM or reinitiation of therapy with EPZICOM, unless patients have a previously documented HLA‑B*5701 allele assessment. Discontinue EPZICOM immediately if a hypersensitivity reaction is suspected, regardless of HLA-B*5701 status and even when other diagnoses are possible [see Contraindications (4), Warnings and Precautions (5.1)].
Following a hypersensitivity reaction to EPZICOM, NEVER restart EPZICOM or any other abacavir‑containing product because more severe symptoms, including death, can occur within hours. Similar severe reactions have also occurred rarely following the reintroduction of abacavir-containing products in patients who have no history of abacavir hypersensitivity [see Warnings and Precautions (5.1)].
Exacerbations of Hepatitis B
Severe acute exacerbations of hepatitis B have been reported in patients who are co‑infected with hepatitis B virus (HBV) and human immunodeficiency virus (HIV‑1) and have discontinued lamivudine, which is a component of EPZICOM. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue EPZICOM and are co-infected with HIV‑1 and HBV. If appropriate, initiation of anti‑hepatitis B therapy may be warranted [see Warnings and Precautions (5.2)].
1. Indications and Usage for Epzicom
EPZICOM, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV‑1) infection.
2. Epzicom Dosage and Administration
2.1 Screening for HLA-B*5701 Allele prior to Starting EPZICOM
Screen for the HLA‑B*5701 allele prior to initiating therapy with EPZICOM [see Boxed Warning, Warnings and Precautions (5.1)].
2.2 Recommended Dosage for Adult Patients
The recommended dosage of EPZICOM for adults is one tablet taken orally once daily, in combination with other antiretroviral agents, with or without food.
2.3 Recommended Dosage for Pediatric Patients
The recommended oral dose of EPZICOM for pediatric patients weighing at least 25 kg is one tablet daily in combination with other antiretroviral agents [see Clinical Studies (14.2)]. Before prescribing EPZICOM tablets, pediatric patients should be assessed for the ability to swallow tablets.
2.4 Not Recommended Due to Lack of Dosage Adjustment
Because EPZICOM is a fixed‑dose tablet and cannot be dose adjusted, EPZICOM is not recommended for:
- •
- patients with creatinine clearance less than 30 mL per minute [see Use in Specific Populations (8.6)].
- •
- patients with mild hepatic impairment. EPZICOM is contraindicated in patients with moderate or severe hepatic impairment [see Contraindications (4), Use in Specific Populations (8.7)].
Use of EPIVIR (lamivudine) oral solution or tablets and ZIAGEN (abacavir) oral solution may be considered.
3. Dosage Forms and Strengths
EPZICOM tablets contain 600 mg of abacavir as abacavir sulfate and 300 mg of lamivudine. The tablets are modified capsule-shaped, orange, film-coated, and debossed with “GS FC2” on one side with no markings on the reverse side.
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
Serious and sometimes fatal hypersensitivity reactions have occurred with abacavir, a component of EPZICOM. These hypersensitivity reactions have included multi-organ failure and anaphylaxis and typically occurred within the first 6 weeks of treatment with abacavir (median time to onset was 9 days); although abacavir hypersensitivity reactions have occurred any time during treatment [see Adverse Reactions (6.1)]. Patients who carry the HLA‑B*5701 allele are at a higher risk of abacavir hypersensitivity reactions; although, patients who do not carry the HLA‑B*5701 allele have developed hypersensitivity reactions. Hypersensitivity to abacavir was reported in approximately 206 (8%) of 2,670 patients in 9 clinical trials with abacavir-containing products where HLA‑B*5701 screening was not performed. The incidence of suspected abacavir hypersensitivity reactions in clinical trials was 1% when subjects carrying the HLA‑B*5701 allele were excluded. In any patient treated with abacavir, the clinical diagnosis of hypersensitivity reaction must remain the basis of clinical decision making.
Due to the potential for severe, serious, and possibly fatal hypersensitivity reactions with abacavir:
- •
- All patients should be screened for the HLA‑B*5701 allele prior to initiating therapy with EPZICOM or reinitiation of therapy with EPZICOM, unless patients have a previously documented HLA‑B*5701 allele assessment.
- •
- EPZICOM is contraindicated in patients with a prior hypersensitivity reaction to abacavir and in HLA‑B*5701-positive patients.
- •
- Before starting EPZICOM, review medical history for prior exposure to any abacavir-containing product. NEVER restart EPZICOM or any other abacavir‑containing product following a hypersensitivity reaction to abacavir, regardless of HLA‑B*5701 status.
- •
- To reduce the risk of a life‑threatening hypersensitivity reaction, regardless of HLA‑B*5701 status, discontinue EPZICOM immediately if a hypersensitivity reaction is suspected, even when other diagnoses are possible (e.g., acute onset respiratory diseases such as pneumonia, bronchitis, pharyngitis, or influenza; gastroenteritis; or reactions to other medications).
- •
- If a hypersensitivity reaction cannot be ruled out, do not restart EPZICOM or any other abacavir‑containing products because more severe symptoms, which may include life‑threatening hypotension and death, can occur within hours.
- •
- If a hypersensitivity reaction is ruled out, patients may restart EPZICOM. Rarely, patients who have stopped abacavir for reasons other than symptoms of hypersensitivity have also experienced life-threatening reactions within hours of reinitiating abacavir therapy. Therefore, reintroduction of EPZICOM or any other abacavir-containing product is recommended only if medical care can be readily accessed.
- •
- A Medication Guide and Warning Card that provide information about recognition of hypersensitivity reactions should be dispensed with each new prescription and refill.
5.2 Patients with Hepatitis B Virus Co-infection
Posttreatment Exacerbations of Hepatitis
Clinical and laboratory evidence of exacerbations of hepatitis have occurred after discontinuation of lamivudine. See full prescribing information for EPIVIR (lamivudine). Patients should be closely monitored with both clinical and laboratory follow‑up for at least several months after stopping treatment.
Emergence of Lamivudine‑Resistant Hepatitis B Virus
Safety and efficacy of lamivudine have not been established for treatment of chronic hepatitis B in subjects dually infected with HIV‑1 and hepatitis B virus (HBV). Emergence of hepatitis B virus variants associated with resistance to lamivudine has been reported in HIV‑1‑infected subjects who have received lamivudine‑containing antiretroviral regimens in the presence of concurrent infection with hepatitis B virus. See full prescribing information for EPIVIR (lamivudine).
5.3 Lactic Acidosis and Severe Hepatomegaly with Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogues, including abacavir and lamivudine (components of EPZICOM). A majority of these cases have been in women. Female sex and obesity may be risk factors for the development of lactic acidosis and severe hepatomegaly with steatosis in patients treated with antiretroviral nucleoside analogues. See full prescribing information for ZIAGEN (abacavir) and EPIVIR (lamivudine). Treatment with EPZICOM should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity, which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations.
5.4 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including EPZICOM. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves’ disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
5.5 Myocardial Infarction
Several prospective, observational, epidemiological studies have reported an association with the use of abacavir and the risk of myocardial infarction (MI). Meta-analyses of randomized, controlled clinical trials have observed no excess risk of MI in abacavir‑treated subjects as compared with control subjects. To date, there is no established biological mechanism to explain the potential increase in risk. In totality, the available data from the observational studies and from controlled clinical trials show inconsistency; therefore, evidence for a causal relationship between abacavir treatment and the risk of MI is inconclusive.
As a precaution, the underlying risk of coronary heart disease should be considered when prescribing antiretroviral therapies, including abacavir, and action taken to minimize all modifiable risk factors (e.g., hypertension, hyperlipidemia, diabetes mellitus, smoking).
6. Adverse Reactions/Side Effects
The following adverse reactions are discussed in other sections of the labeling:
- •
- Serious and sometimes fatal hypersensitivity reactions [see Boxed Warning, Warnings and Precautions (5.1)].
- •
- Exacerbations of hepatitis B [see Boxed Warning, Warnings and Precautions (5.2)].
- •
- Lactic acidosis and severe hepatomegaly with steatosis [see Warnings and Precautions (5.3)].
- •
- Immune reconstitution syndrome [see Warnings and Precautions (5.4)].
- •
- Myocardial infarction [see Warnings and Precautions (5.5)].
6.1 Clinical Trials Experience in Adult Subjects
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Serious and Fatal Abacavir-Associated Hypersensitivity Reactions
In clinical trials, serious and sometimes fatal hypersensitivity reactions have occurred with abacavir, a component of EPZICOM [see Boxed Warning, Warnings and Precautions (5.1)]. These reactions have been characterized by 2 or more of the following signs or symptoms: (1) fever; (2) rash; (3) gastrointestinal symptoms (including nausea, vomiting, diarrhea, or abdominal pain); (4) constitutional symptoms (including generalized malaise, fatigue, or achiness); (5) respiratory symptoms (including dyspnea, cough, or pharyngitis). Almost all abacavir hypersensitivity reactions include fever and/or rash as part of the syndrome.
Other signs and symptoms have included lethargy, headache, myalgia, edema, arthralgia, and paresthesia. Anaphylaxis, liver failure, renal failure, hypotension, adult respiratory distress syndrome, respiratory failure, myolysis, and death have occurred in association with these hypersensitivity reactions. Physical findings have included lymphadenopathy, mucous membrane lesions (conjunctivitis and mouth ulcerations), and maculopapular or urticarial rash (although some patients had other types of rashes and others did not have a rash). There were reports of erythema multiforme. Laboratory abnormalities included elevated liver chemistries, elevated creatine phosphokinase, elevated creatinine, and lymphopenia and abnormal chest x‑ray findings (predominantly infiltrates, which were localized).
Additional Adverse Reactions with Use of EPZICOM
Therapy‑Naive Adults: Treatment‑emergent clinical adverse reactions (rated by the investigator as moderate or severe) with greater than or equal to 5% frequency during therapy with ZIAGEN 600 mg once daily or ZIAGEN 300 mg twice daily, both in combination with lamivudine 300 mg once daily and efavirenz 600 mg once daily, are listed in Table 1.
| a Subjects receiving ZIAGEN 600 mg once daily, experienced a significantly higher incidence of severe drug hypersensitivity reactions and severe diarrhea compared with subjects who received ZIAGEN 300 mg twice daily. Five percent (5%) of subjects receiving ZIAGEN 600 mg once daily had severe drug hypersensitivity reactions compared with 2% of subjects receiving ZIAGEN 300 mg twice daily. Two percent (2%) of subjects receiving ZIAGEN 600 mg once daily had severe diarrhea while none of the subjects receiving ZIAGEN 300 mg twice daily had this event. b CNA30024 was a multi‑center, double-blind, controlled trial in which 649 HIV‑1‑infected, therapy‑naive adults were randomized and received either ZIAGEN (300 mg twice daily), EPIVIR (150 mg twice daily), and efavirenz (600 mg once daily); or zidovudine (300 mg twice daily), EPIVIR (150 mg twice daily), and efavirenz (600 mg once daily). CNA30024 used double‑blind ascertainment of suspected hypersensitivity reactions. During the blinded portion of the trial, suspected hypersensitivity to abacavir was reported by investigators in 9% of 324 subjects in the abacavir group and 3% of 325 subjects in the zidovudine group. |
||
|
Adverse Event |
ZIAGEN 600 mg q.d. plus EPIVIR plus Efavirenz (n = 384) |
ZIAGEN 300 mg b.i.d. plus EPIVIR plus Efavirenz (n = 386) |
|
Drug hypersensitivitya,b |
9% |
7% |
|
Insomnia |
7% |
9% |
|
Depression/Depressed mood |
7% |
7% |
|
Headache/Migraine |
7% |
6% |
|
Fatigue/Malaise |
6% |
8% |
|
Dizziness/Vertigo |
6% |
6% |
|
Nausea |
5% |
6% |
|
Diarrheaa |
5% |
6% |
|
Rash |
5% |
5% |
|
Pyrexia |
5% |
3% |
|
Abdominal pain/gastritis |
4% |
5% |
|
Abnormal dreams |
4% |
5% |
|
Anxiety |
3% |
5% |
Laboratory Abnormalities: Laboratory abnormalities observed in clinical trials of ZIAGEN were anemia, neutropenia, liver function test abnormalities, and elevations of creatine phosphokinase (CPK), blood glucose, and triglycerides. Additional laboratory abnormalities observed in clinical trials of EPIVIR were thrombocytopenia and elevated levels of bilirubin, amylase, and lipase.
The frequencies of treatment‑emergent laboratory abnormalities were comparable between treatment groups in CNA30021.
Other Adverse Events: In addition to adverse reactions listed above, other adverse events observed in the expanded access program for abacavir were pancreatitis and increased gamma-glutamyl transferase (GGT).
6.2 Clinical Trials Experience in Pediatric Subjects
The safety of once-daily compared with twice-daily dosing of abacavir and lamivudine, administered as either single products or as EPZICOM, was assessed in the ARROW trial (n = 336). Primary safety assessment in the ARROW (COL105677) trial was based on Grade 3 and Grade 4 adverse events. The frequency of Grade 3 and 4 adverse events was similar among subjects randomized to once-daily dosing compared with subjects randomized to twice-daily dosing. One event of Grade 4 hepatitis in the once-daily cohort was considered as uncertain causality by the investigator and all other Grade 3 or 4 adverse events were considered not related by the investigator. No additional safety issues were identified in pediatric subjects receiving abacavir and lamivudine once-daily compared with historical data in adults [see Adverse Reactions (6.1)].
6.3 Postmarketing Experience
The following adverse reactions have been identified during postmarketing use. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Abacavir
Cardiovascular: Myocardial infarction.
Skin: Suspected Stevens‑Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) have been reported in patients receiving abacavir primarily in combination with medications known to be associated with SJS and TEN, respectively. Because of the overlap of clinical signs and symptoms between hypersensitivity to abacavir and SJS and TEN, and the possibility of multiple drug sensitivities in some patients, abacavir should be discontinued and not restarted in such cases. There have also been reports of erythema multiforme with abacavir use [see Adverse Reactions (6.1)].
Abacavir and Lamivudine
Body as a Whole: Redistribution/accumulation of body fat.
Digestive: Stomatitis.
Endocrine and Metabolic: Hyperglycemia.
General: Weakness.
Hemic and Lymphatic: Aplastic anemia, anemia (including pure red cell aplasia and severe anemias progressing on therapy), lymphadenopathy, splenomegaly.
Hepatic: Lactic acidosis and hepatic steatosis [see Warnings and Precautions (5.3)], posttreatment exacerbations of hepatitis B [see Warnings and Precautions (5.2)].
Hypersensitivity: Sensitization reactions (including anaphylaxis), urticaria.
Musculoskeletal: Muscle weakness, CPK elevation, rhabdomyolysis.
Nervous: Paresthesia, peripheral neuropathy, seizures.
Respiratory: Abnormal breath sounds/wheezing.
Skin: Alopecia, erythema multiforme, Stevens-Johnson syndrome.
7. Drug Interactions
7.1 Methadone
In a trial of 11 HIV‑1‑infected subjects receiving methadone‑maintenance therapy with 600 mg of ZIAGEN twice daily (twice the currently recommended dose), oral methadone clearance increased [see Clinical Pharmacology (12.3)]. This alteration will not result in a methadone dose modification in the majority of patients; however, an increased methadone dose may be required in a small number of patients.
7.2 Sorbitol
Coadministration of single doses of lamivudine and sorbitol resulted in a sorbitol dose-dependent reduction in lamivudine exposures. When possible, avoid use of sorbitol-containing medicines with lamivudine-containing medicines [see Clinical Pharmacology (12.3)].
7.3 Riociguat
Coadministration with fixed-dose abacavir/dolutegravir/lamivudine resulted in increased riociguat exposure, which may increase the risk of riociguat adverse reactions [see Clinical Pharmacology (12.3)]. The riociguat dose may need to be reduced. See full prescribing information for ADEMPAS (riociguat).
8. Use In Specific Populations
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to EPZICOM during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Risk Summary
Available data from the APR show no difference in the overall risk of birth defects for abacavir or lamivudine compared with the background rate for birth defects of 2.7% in the Metropolitan Atlanta Congenital Defects Program (MACDP) reference population (see Data). The APR uses the MACDP as the U.S. reference population for birth defects in the general population. The MACDP evaluates women and infants from a limited geographic area and does not include outcomes for births that occurred at less than 20 weeks’ gestation. The rate of miscarriage is not reported in the APR. The estimated background rate of miscarriage in clinically recognized pregnancies in the U.S. general population is 15% to 20%. The background risk for major birth defects and miscarriage for the indicated population is unknown.
In animal reproduction studies, oral administration of abacavir to pregnant rats during organogenesis resulted in fetal malformations and other embryonic and fetal toxicities at exposures 35 times the human exposure (AUC) at the recommended clinical daily dose. However, no adverse developmental effects were observed following oral administration of abacavir to pregnant rabbits during organogenesis, at exposures approximately 9 times the human exposure (AUC) at the recommended clinical dose. Oral administration of lamivudine to pregnant rabbits during organogenesis resulted in embryolethality at systemic exposure (AUC) similar to the recommended clinical dose; however, no adverse development effects were observed with oral administration of lamivudine to pregnant rats during organogenesis at plasma concentrations (Cmax) 35 times the recommended clinical dose (see Data).
Data
Human Data: Abacavir: Based on prospective reports to the APR of exposures to abacavir during pregnancy resulting in live births (including over 1,300 exposed in the first trimester and over 1,300 exposed in the second/third trimester), there was no difference between the overall risk of birth defects for abacavir compared with the background birth defect rate of 2.7% in the U.S. reference population of the MACDP. The prevalence of defects in live births was 3.2% (95% CI: 2.3% to 4.3%) following first trimester exposure to abacavir-containing regimens and 2.9% (95% CI: 2.1% to 4.0%) following second/third trimester exposure to abacavir-containing regimens.
Abacavir has been shown to cross the placenta and concentrations in neonatal plasma at birth were essentially equal to those in maternal plasma at delivery [see Clinical Pharmacology (12.3)].
Lamivudine: Based on prospective reports to the APR of exposures to lamivudine during pregnancy resulting in live births (including over 5,300 exposed in the first trimester and over 7,400 exposed in the second/third trimester), there was no difference between the overall risk of birth defects for lamivudine compared with the background birth defect rate of 2.7% in the U.S. reference population of the MACDP. The prevalence of birth defects in live births was 3.1% (95% CI: 2.7% to 3.6%) following first trimester exposure to lamivudine-containing regimens and 2.9% (95% CI: 2.5%, 3.3%) following second/third trimester exposure to lamivudine-containing regimens.
Lamivudine pharmacokinetics were studied in pregnant women during 2 clinical trials conducted in South Africa. The trials assessed pharmacokinetics in 16 women at 36 weeks’ gestation using 150 mg lamivudine twice daily with zidovudine, 10 women at 38 weeks’ gestation using 150 mg lamivudine twice daily with zidovudine, and 10 women at 38 weeks’ gestation using lamivudine 300 mg twice daily without other antiretrovirals. These trials were not designed or powered to provide efficacy information. Lamivudine concentrations were generally similar in maternal, neonatal, and umbilical cord serum samples. In a subset of subjects, amniotic fluid specimens were collected following natural rupture of membranes and confirmed that lamivudine crosses the placenta in humans. Based on limited data at delivery, median (range) amniotic fluid concentrations of lamivudine were 3.9 (1.2 to 12.8)–fold greater compared with paired maternal serum concentration (n = 8).
Animal Data: Abacavir: Abacavir was administered orally to pregnant rats (at 100, 300, and 1,000 mg per kg per day) and rabbits (at 125, 350, or 700 mg per kg per day) during organogenesis (on Gestation Days 6 through 17 and 6 through 20, respectively). Fetal malformations (increased incidences of fetal anasarca and skeletal malformations) or developmental toxicity (decreased fetal body weight and crown‑rump length) were observed in rats at doses up to 1,000 mg per kg per day, resulting in exposures approximately 35 times the human exposure (AUC) at the recommended daily dose. No developmental effects were observed in rats at 100 mg per kg per day, resulting in exposures (AUC) 3.5 times the human exposure at the recommended daily dose. In a fertility and early embryo-fetal development study conducted in rats (at 60, 160, or 500 mg per kg per day), embryonic and fetal toxicities (increased resorptions, decreased fetal body weights) or toxicities to the offspring (increased incidence of stillbirth and lower body weights) occurred at doses up to 500 mg per kg per day. No developmental effects were observed in rats at 60 mg per kg per day, resulting in exposures (AUC) approximately 4 times the human exposure at the recommended daily dose. Studies in pregnant rats showed that abacavir is transferred to the fetus through the placenta. In pregnant rabbits, no developmental toxicities and no increases in fetal malformations occurred at up to the highest dose evaluated, resulting in exposures (AUC) approximately 9 times the human exposure at the recommended dose.
Lamivudine: Lamivudine was administered orally to pregnant rats (at 90, 600, and 4,000 mg per kg per day) and rabbits (at 90, 300 and 1,000 mg per kg per day and at 15, 40, and 90 mg per kg per day) during organogenesis (on Gestation Days 7 through 16 [rat] and 8 through 20 [rabbit]). No evidence of fetal malformations due to lamivudine was observed in rats and rabbits at doses producing plasma concentrations (Cmax) approximately 35 times higher than human exposure at the recommended daily dose. Evidence of early embryolethality was seen in the rabbit at systemic exposures (AUC) similar to those observed in humans, but there was no indication of this effect in the rat at plasma concentrations (Cmax) 35 times higher than human exposure at the recommended daily dose. Studies in pregnant rats showed that lamivudine is transferred to the fetus through the placenta. In the fertility/pre- and postnatal development study in rats, lamivudine was administered orally at doses of 180, 900, and 4,000 mg per kg per day from prior to mating through postnatal Day 20). In the study, development of the offspring, including fertility and reproductive performance, were not affected by the maternal administration of lamivudine.
8.2 Lactation
Risk Summary
The Centers for Disease Control and Prevention recommends that HIV‑1‑infected mothers in the United States not breastfeed their infants to avoid risking postnatal transmission of HIV‑1 infection. Abacavir and lamivudine are present in human milk. There is no information on the effects of abacavir and lamivudine on the breastfed infant or the effects of the drug on milk production. Because of the potential for (1) HIV‑1 transmission (in HIV-negative infants), (2) developing viral resistance (in HIV-positive infants), and (3) adverse reactions in a breastfed infant similar to those seen in adults, instruct mothers not to breastfeed if they are receiving EPZICOM.
8.4 Pediatric Use
The dosing recommendations in this population are based on the safety and efficacy established in a controlled trial conducted using either the combination of EPIVIR and ZIAGEN or EPZICOM [see Dosage and Administration (2.3), Adverse Reactions (6.2), Clinical Studies (14.2)].
In pediatric patients weighing less than 25 kg, use of abacavir and lamivudine as single products is recommended to achieve appropriate dosing.
8.5 Geriatric Use
Clinical trials of abacavir and lamivudine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, caution should be exercised in the administration of EPZICOM in elderly patients reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Dosage and Administration (2.4), Use in Specific Populations (8.6, 8.7)].
8.6 Patients with Impaired Renal Function
EPZICOM is not recommended for patients with creatinine clearance less than 30 mL per min because EPZICOM is a fixed-dose combination and the dosage of the individual components cannot be adjusted. If a dose reduction of lamivudine, a component of EPZICOM, is required for patients with creatinine clearance less than 30 mL per min, then the individual components should be used [see Clinical Pharmacology (12.3)].
Patients with a creatinine clearance between 30 and 49 mL per min receiving EPZICOM may experience a 1.6- to 3.3-fold higher lamivudine exposure (AUC) than patients with a creatinine clearance ≥50 mL per min. There are no safety data from randomized, controlled trials comparing EPZICOM to the individual components in patients with a creatinine clearance between 30 and 49 mL per min who received dose-adjusted lamivudine. In the original lamivudine registrational trials in combination with zidovudine, higher lamivudine exposures were associated with higher rates of hematologic toxicities (neutropenia and anemia), although discontinuations due to neutropenia or anemia each occurred in <1% of subjects. Patients with a sustained creatinine clearance between 30 and 49 mL per min who receive EPZICOM should be monitored for hematologic toxicities. If new or worsening neutropenia or anemia develop, dose adjustment of lamivudine, per lamivudine prescribing information, is recommended. If lamivudine dose adjustment is indicated, EPZICOM should be discontinued and the individual components should be used to construct the treatment regimen.
8.7 Patients with Impaired Hepatic Function
EPZICOM is a fixed-dose combination and the dosage of the individual components cannot be adjusted. If a dose reduction of abacavir, a component of EPZICOM, is required for patients with mild hepatic impairment (Child-Pugh Class A), then the individual components should be used [see Clinical Pharmacology (12.3)].
The safety, efficacy, and pharmacokinetic properties of abacavir have not been established in patients with moderate (Child-Pugh Class B) or severe (Child-Pugh Class C) hepatic impairment; therefore, EPZICOM is contraindicated in these patients [see Contraindications (4)].
10. Overdosage
There is no known specific treatment for overdose with EPZICOM. If overdose occurs, the patient should be monitored, and standard supportive treatment applied as required.
Abacavir: It is not known whether abacavir can be removed by peritoneal dialysis or hemodialysis.
Lamivudine: Because a negligible amount of lamivudine was removed via (4-hour) hemodialysis, continuous ambulatory peritoneal dialysis, and automated peritoneal dialysis, it is not known if continuous hemodialysis would provide clinical benefit in a lamivudine overdose event.
11. Epzicom Description
EPZICOM
EPZICOM tablets contain the following 2 synthetic nucleoside analogues: abacavir (ZIAGEN, also a component of TRIZIVIR) and lamivudine (also known as EPIVIR or 3TC) with inhibitory activity against HIV‑1.
EPZICOM tablets are for oral administration. Each orange, film‑coated tablet contains the active ingredients 600 mg of abacavir as abacavir sulfate and 300 mg of lamivudine, and the inactive ingredients magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The tablets are coated with a film (OPADRY orange YS-1-13065-A) that is made of FD&C Yellow No. 6, hypromellose, polyethylene glycol 400, polysorbate 80, and titanium dioxide.
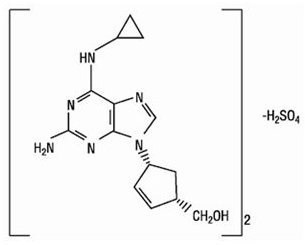
Abacavir Sulfate
The chemical name of abacavir sulfate is (1S,cis)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol sulfate (salt) (2:1). Abacavir sulfate is the enantiomer with 1S, 4R absolute configuration on the cyclopentene ring. It has a molecular formula of (C14H18N6O)2•H2SO4 and a molecular weight of 670.76 g per mol. It has the following structural formula:
Abacavir sulfate is a white to off‑white solid and is soluble in water.
In vivo, abacavir sulfate dissociates to its free base, abacavir. Dosages are expressed in terms of abacavir.
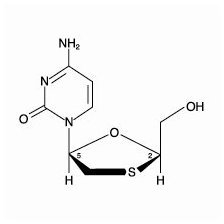
Lamivudine
The chemical name of lamivudine is (2R,cis)-4-amino-1-(2-hydroxymethyl-1,3-oxathiolan-5-yl)-(1H)-pyrimidin-2-one. Lamivudine is the (‑)enantiomer of a dideoxy analogue of cytidine. Lamivudine has also been referred to as (‑)2′,3′-dideoxy, 3′-thiacytidine. It has a molecular formula of C8H11N3O3S and a molecular weight of 229.3 g per mol. It has the following structural formula:
Lamivudine is a white to off‑white crystalline solid and is soluble in water.
12. Epzicom - Clinical Pharmacology
12.1 Mechanism of Action
EPZICOM is an antiretroviral agent with activity against HIV-1 [see Microbiology (12.4)].
12.3 Pharmacokinetics
Pharmacokinetics in Adults
In a single-dose, 3-way crossover bioavailability trial of 1 EPZICOM tablet versus 2 ZIAGEN tablets (2 x 300 mg) and 2 EPIVIR tablets (2 x 150 mg) administered simultaneously in healthy subjects (n = 25), there was no difference in the extent of absorption, as measured by the area under the plasma concentration-time curve (AUC) and maximal peak concentration (Cmax), of each component.
Abacavir: Following oral administration, abacavir is rapidly absorbed and extensively distributed. After oral administration of a single dose of 600 mg of abacavir in 20 subjects, Cmax was 4.26 ± 1.19 mcg per mL (mean ± SD) and AUC∞ was 11.95 ± 2.51 mcg•hour per mL. Binding of abacavir to human plasma proteins is approximately 50% and was independent of concentration. Total blood and plasma drug-related radioactivity concentrations are identical, demonstrating that abacavir readily distributes into erythrocytes. The primary routes of elimination of abacavir are metabolism by alcohol dehydrogenase to form the 5′-carboxylic acid and glucuronyl transferase to form the 5′-glucuronide.
Lamivudine: Following oral administration, lamivudine is rapidly absorbed and extensively distributed. After multiple-dose oral administration of lamivudine 300 mg once daily for 7 days to 60 healthy subjects, steady-state Cmax (Cmax,ss) was 2.04 ± 0.54 mcg per mL (mean ± SD) and the 24-hour steady-state AUC (AUC24,ss) was 8.87 ± 1.83 mcg•hour per mL. Binding to plasma protein is low. Approximately 70% of an intravenous dose of lamivudine is recovered as unchanged drug in the urine. Metabolism of lamivudine is a minor route of elimination. In humans, the only known metabolite is the trans-sulfoxide metabolite (approximately 5% of an oral dose after 12 hours).
In humans, abacavir and lamivudine are not significantly metabolized by cytochrome P450 (CYP) enzymes.
The pharmacokinetic properties of abacavir and lamivudine in fasting subjects are summarized in Table 2.
| a Data presented as mean ± standard deviation except where noted. b Approximate range. |
||||
|
Parameter |
Abacavir |
Lamivudine |
||
|
Oral bioavailability (%) |
86 ± 25 |
n = 6 |
86 ± 16 |
n = 12 |
|
Apparent volume of distribution (L/kg) |
0.86 ± 0.15 |
n = 6 |
1.3 ± 0.4 |
n = 20 |
|
Systemic clearance (L/h/kg) |
0.80 ± 0.24 |
n = 6 |
0.33 ± 0.06 |
n = 20 |
|
Renal clearance (L/h/kg) |
0.007 ± 0.008 |
n = 6 |
0.22 ± 0.06 |
n = 20 |
|
Elimination half-life (h) |
1.45 ± 0.32 |
n = 20 |
13 to 19b |
|
Effect of Food on Absorption of EPZICOM
EPZICOM may be administered with or without food. Administration with a high-fat meal in a single-dose bioavailability trial resulted in no change in AUClast, AUC∞, and Cmax for lamivudine. Food did not alter the extent of systemic exposure to abacavir (AUC∞), but the rate of absorption (Cmax) was decreased approximately 24% compared with fasted conditions (n = 25). These results are similar to those from previous trials of the effect of food on abacavir and lamivudine tablets administered separately.
Specific Populations
Patients with Renal Impairment: The pharmacokinetics for the individual lamivudine component of EPZICOM has been evaluated in patients with renal impairment (see the U.S. prescribing information for the individual lamivudine component).
Patients with Hepatic Impairment: The pharmacokinetics for the individual components of EPZICOM have been evaluated in patients with varying degrees of hepatic impairment (see the U.S. prescribing information for the individual abacavir and lamivudine components).
Pregnant Women: Abacavir: Abacavir pharmacokinetics were studied in 25 pregnant women during the last trimester of pregnancy receiving abacavir 300 mg twice daily. Abacavir exposure (AUC) during pregnancy was similar to those in postpartum and in HIV-infected non-pregnant historical controls. Consistent with passive diffusion of abacavir across the placenta, abacavir concentrations in neonatal plasma cord samples at birth were essentially equal to those in maternal plasma at delivery.
Lamivudine: Lamivudine pharmacokinetics were studied in 36 pregnant women during 2 clinical trials conducted in South Africa. Lamivudine pharmacokinetics in pregnant women were similar to those seen in non-pregnant adults and in postpartum women. Lamivudine concentrations were generally similar in maternal, neonatal, and umbilical cord serum samples.
Pediatric Patients: Abacavir and Lamivudine: The pharmacokinetic data for abacavir and lamivudine following administration of EPZICOM in pediatric subjects weighing 25 kg and above are limited. The dosing recommendations in this population are based on the safety and efficacy established in a controlled trial conducted using either the combination of EPIVIR and ZIAGEN or EPZICOM. Refer to the EPIVIR and ZIAGEN USPI for pharmacokinetic information on the individual products in pediatric patients [see Dosage and Administration (2.3), Adverse Reactions (6.2), Clinical Studies (14.2)].
Geriatric Patients: The pharmacokinetics of abacavir and lamivudine have not been studied in subjects over 65 years of age.
Male and Female Patients: There are no significant or clinically relevant gender differences in the pharmacokinetics of the individual components (abacavir or lamivudine) based on the available information that was analyzed for each of the individual components.
Racial Groups: There are no significant or clinically relevant racial differences in pharmacokinetics of the individual components (abacavir or lamivudine) based on the available information that was analyzed for each of the individual components.
Drug Interaction Studies
The drug interactions described are based on trials conducted with abacavir or lamivudine as single entities; no drug interaction trials have been conducted with EPZICOM.
Effect of Abacavir and Lamivudine on the Pharmacokinetics of Other Agents: In vitro studies have shown that abacavir has potential to inhibit CYP1A1 and limited potential to inhibit metabolism mediated by CYP3A4. Lamivudine does not inhibit or induce CYP3A4. Abacavir and lamivudine do not inhibit or induce other CYP enzymes (such as CYP2C9 or CYP2D6). Based on in vitro study results, abacavir and lamivudine at therapeutic drug exposures are not expected to affect the pharmacokinetics of drugs that are substrates of the following transporters: organic anion transporter polypeptide (OATP)1B1/3, breast cancer resistance protein (BCRP) or P-glycoprotein (P-gp), organic cation transporter (OCT)1, OCT2, OCT3 (lamivudine only), or multidrug and toxic extrusion protein (MATE)1 and MATE2-K.
Riociguat: Coadministration of a single dose of riociguat (0.5 mg) to HIV-1–infected subjects receiving fixed-dose abacavir/dolutegravir/lamivudine is reported to increase riociguat AUC(∞) compared with riociguat AUC(∞) reported in healthy subjects due to CYP1A1 inhibition by abacavir. The exact magnitude of increase in riociguat exposure has not been fully characterized based on findings from two studies [see Drug Interactions (7.3)].
Effect of Other Agents on the Pharmacokinetics of Abacavir or Lamivudine: Abacavir and lamivudine are not significantly metabolized by CYP enzymes; therefore, CYP enzyme inhibitors or inducers are not expected to affect their concentrations. In vitro, abacavir is not a substrate of OATP1B1, OATP1B3, OCT1, OCT2, OAT1, MATE1, MATE2-K, multidrug resistance-associated protein 2 (MRP2) or MRP4; therefore, drugs that modulate these transporters are not expected to affect abacavir plasma concentrations. Abacavir is a substrate of BCRP and P-gp in vitro; however, considering its absolute bioavailability (83%), modulators of these transporters are unlikely to result in a clinically relevant impact on abacavir concentrations.
Lamivudine is a substrate of MATE1, MATE2-K, and OCT2 in vitro. Trimethoprim (an inhibitor of these drug transporters) has been shown to increase lamivudine plasma concentrations. This interaction is not considered clinically significant as no dose adjustment of lamivudine is needed.
Lamivudine is a substrate of P-gp and BCRP; however, considering its absolute bioavailability (87%), it is unlikely that these transporters play a significant role in the absorption of lamivudine. Therefore, coadministration of drugs that are inhibitors of these efflux transporters is unlikely to affect the disposition and elimination of lamivudine.
Abacavir: Lamivudine and/or Zidovudine: Fifteen HIV‑1‑infected subjects were enrolled in a crossover‑designed drug interaction trial evaluating single doses of abacavir (600 mg), lamivudine (150 mg), and zidovudine (300 mg) alone or in combination. Analysis showed no clinically relevant changes in the pharmacokinetics of abacavir with the addition of lamivudine or zidovudine or the combination of lamivudine and zidovudine. Lamivudine exposure (AUC decreased 15%) and zidovudine exposure (AUC increased 10%) did not show clinically relevant changes with concurrent abacavir.
Lamivudine: Zidovudine: No clinically significant alterations in lamivudine or zidovudine pharmacokinetics were observed in 12 asymptomatic HIV‑1-infected adult subjects given a single dose of zidovudine (200 mg) in combination with multiple doses of lamivudine (300 mg every 12 h).
Other Interactions
Ethanol: Abacavir has no effect on the pharmacokinetic properties of ethanol. Ethanol decreases the elimination of abacavir causing an increase in overall exposure.
Interferon Alfa: There was no significant pharmacokinetic interaction between lamivudine and interferon alfa in a trial of 19 healthy male subjects.
Methadone: In a trial of 11 HIV‑1‑infected subjects receiving methadone‑maintenance therapy (40 mg and 90 mg daily), with 600 mg of ZIAGEN twice daily (twice the currently recommended dose), oral methadone clearance increased 22% (90% CI: 6% to 42%) [see Drug Interactions (7)]. The addition of methadone has no clinically significant effect on the pharmacokinetic properties of abacavir.
Ribavirin: In vitro data indicate ribavirin reduces phosphorylation of lamivudine, stavudine, and zidovudine. However, no pharmacokinetic (e.g., plasma concentrations or intracellular triphosphorylated active metabolite concentrations) or pharmacodynamic (e.g., loss of HIV‑1/hepatitis C virus [HCV] virologic suppression) interaction was observed when ribavirin and lamivudine (n = 18), stavudine (n = 10), or zidovudine (n = 6) were coadministered as part of a multi‑drug regimen to HIV‑1/HCV co‑infected subjects.
Sorbitol (Excipient): Lamivudine and sorbitol solutions were coadministered to 16 healthy adult subjects in an open-label, randomized-sequence, 4-period, crossover trial. Each subject received a single 300-mg dose of lamivudine oral solution alone or coadministered with a single dose of 3.2 grams, 10.2 grams, or 13.4 grams of sorbitol in solution. Coadministration of lamivudine with sorbitol resulted in dose-dependent decreases of 20%, 39%, and 44% in the AUC(0-24); 14%, 32%, and 36% in the AUC(∞); and 28%, 52%, and 55% in the Cmax; of lamivudine, respectively.
The effects of other coadministered drugs on abacavir or lamivudine are provided in Table 3.
| ↑ = Increase; ↔ = No significant change; AUC = Area under the concentration versus time curve; CI = Confidence interval. a The drug-drug interaction was only evaluated in males. |
||||||||||
|
Coadministered Drug and Dose |
Drug and Dose |
n |
Concentrations of Abacavir or Lamivudine |
Concentration of Coadministered Drug |
||||||
|
AUC |
Variability |
|||||||||
|
Ethanol 0.7 g/kg |
Abacavir Single 600 mg |
24 |
↑41% |
90% CI: 35% to 48% |
↔a |
|||||
|
Nelfinavir 750 mg every 8 h x 7 to 10 days |
Lamivudine Single 150 mg |
11 |
↑10% |
95% CI: 1% to 20% |
↔ |
|||||
|
Trimethoprim 160 mg/ Sulfamethoxazole 800 mg daily x 5 days |
Lamivudine Single 300 mg |
14 |
↑43% |
90% CI: 32% to 55% |
↔ |
|||||
12.4 Microbiology
Mechanism of Action
Abacavir: Abacavir is a carbocyclic synthetic nucleoside analogue. Abacavir is converted by cellular enzymes to the active metabolite, carbovir triphosphate (CBV-TP), an analogue of deoxyguanosine-5′-triphosphate (dGTP). CBV-TP inhibits the activity of HIV-1 reverse transcriptase (RT) both by competing with the natural substrate dGTP and by its incorporation into viral DNA.
Lamivudine: Lamivudine is a synthetic nucleoside analogue. Intracellularly lamivudine is phosphorylated to its active 5′-triphosphate metabolite, lamivudine triphosphate (3TC-TP). The principal mode of action of 3TC-TP is inhibition of RT via DNA chain termination after incorporation of the nucleotide analogue.
Antiviral Activity
Abacavir: The antiviral activity of abacavir against HIV-1 was assessed in a number of cell lines including primary monocytes/macrophages and peripheral blood mononuclear cells (PBMCs). EC50 values ranged from 3,700 to 5,800 nM (1 nM = 0.28 ng per mL) and 70 to 1,000 nM against HIV-1IIIB and HIV-1BaL, respectively, and the mean EC50 value was 260 ± 180 nM against 8 clinical isolates. The median EC50 values of abacavir were 344 nM (range: 14.8 to 676 nM), 16.9 nM (range: 5.9 to 27.9 nM), 8.1 nM (range: 1.5 to 16.7 nM), 356 nM (range: 35.7 to 396 nM), 105 nM (range: 28.1 to 168 nM), 47.6 nM (range: 5.2 to 200 nM), 51.4 nM (range: 7.1 to 177 nM), and 282 nM (range: 22.4 to 598 nM) against HIV-1 clades A-G and group O viruses (n = 3 except n = 2 for clade B), respectively. The EC50 values against HIV-2 isolates (n = 4) ranged from 24 to 490 nM.
Lamivudine: The antiviral activity of lamivudine against HIV-1 was assessed in a number of cell lines including monocytes and PBMCs using standard susceptibility assays. EC50 values were in the range of 3 to 15,000 nM (1 nM = 0.23 ng per mL). The median EC50 values of lamivudine were 60 nM (range: 20 to 70 nM), 35 nM (range: 30 to 40 nM), 30 nM (range: 20 to 90 nM), 20 nM (range: 3 to 40 nM), 30 nM (range: 1 to 60 nM), 30 nM (range: 20 to 70 nM), 30 nM (range: 3 to 70 nM), and 30 nM (range: 20 to 90 nM) against HIV-1 clades A-G and group O viruses (n = 3 except n = 2 for clade B), respectively. The EC50 values against HIV-2 isolates (n = 4) ranged from 3 to 120 nM in PBMCs. Ribavirin (50,000 nM) used in the treatment of chronic HCV infection decreased the anti-HIV-1 activity of lamivudine by 3.5-fold in MT-4 cells.
The combination of abacavir and lamivudine has demonstrated antiviral activity in cell culture against non‑subtype B isolates and HIV‑2 isolates with equivalent antiviral activity as for subtype B isolates. Neither abacavir, nor lamivudine, were antagonistic to all tested anti-HIV agents. See full prescribing information for ZIAGEN (abacavir) and EPIVIR (lamivudine). Ribavirin, used in the treatment of HCV infection, decreased the anti-HIV-1 potency of abacavir/lamivudine reproducibly by 2- to 6-fold in cell culture.
Resistance
HIV‑1 isolates with reduced susceptibility to the combination of abacavir and lamivudine have been selected in cell culture with amino acid substitutions K65R, L74V, Y115F, and M184V/I emerging in HIV‑1 RT. M184V or I substitutions resulted in high-level resistance to lamivudine and an approximately 2-fold decrease in susceptibility to abacavir. Substitutions K65R, L74M, or Y115F with M184V or I conferred a 7- to 8-fold reduction in abacavir susceptibility, and combinations of three substitutions were required to confer more than an 8-fold reduction in susceptibility.
Cross-Resistance
Cross‑resistance has been observed among nucleoside reverse transcriptase inhibitors (NRTIs). The combination of abacavir/lamivudine has demonstrated decreased susceptibility to viruses with a K65R substitution with or without an M184V/I substitution, viruses with L74V plus the M184V/I substitution, and viruses with thymidine analog mutation substitutions (TAMs: M41L, D67N, K70R, L210W, T215Y/F, K219E/R/H/Q/N) plus M184V. An increasing number of TAMs is associated with a progressive reduction in abacavir susceptibility.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
Abacavir: Abacavir was administered orally at 3 dosage levels to separate groups of mice and rats in 2-year carcinogenicity studies. Results showed an increase in the incidence of malignant and non-malignant tumors. Malignant tumors occurred in the preputial gland of males and the clitoral gland of females of both species, and in the liver of female rats. In addition, non-malignant tumors also occurred in the liver and thyroid gland of female rats. These observations were made at systemic exposures in the range of 6 to 32 times the human exposure at the recommended dose of 600 mg.
Lamivudine: Long‑term carcinogenicity studies with lamivudine in mice and rats showed no evidence of carcinogenic potential at exposures up to 10 times (mice) and 58 times (rats) the human exposures at the recommended dose of 300 mg.
Mutagenicity
Abacavir: Abacavir induced chromosomal aberrations both in the presence and absence of metabolic activation in an in vitro cytogenetic study in human lymphocytes. Abacavir was mutagenic in the absence of metabolic activation, although it was not mutagenic in the presence of metabolic activation in an L5178Y mouse lymphoma assay. Abacavir was clastogenic in males and not clastogenic in females in an in vivo mouse bone marrow micronucleus assay. Abacavir was not mutagenic in bacterial mutagenicity assays in the presence and absence of metabolic activation.
Lamivudine: Lamivudine was mutagenic in an L5178Y mouse lymphoma assay and clastogenic in a cytogenetic assay using cultured human lymphocytes. Lamivudine was not mutagenic in a microbial mutagenicity assay, in an in vitro cell transformation assay, in a rat micronucleus test, in a rat bone marrow cytogenetic assay, and in an assay for unscheduled DNA synthesis in rat liver.
Impairment of Fertility
Abacavir: Abacavir did not affect male or female fertility in rats at a dose associated with exposures (AUC) approximately 3.3 times (male) or 4.1 times (female) those in humans at the clinically recommended dose.
Lamivudine: Lamivudine did not affect male or female fertility in rats at doses up to 4,000 mg per kg per day, associated with concentrations approximately 42 times (male) or 63 times (female) higher than the concentrations (Cmax) in humans at the dose of 300 mg.
13.2 Animal Toxicology and/or Pharmacology
Myocardial degeneration was found in mice and rats following administration of abacavir for 2 years. The systemic exposures were equivalent to 7 to 24 times the expected systemic exposure in humans at a dose of 600 mg. The clinical relevance of this finding has not been determined.
14. Clinical Studies
14.1 Adults
One EPZICOM tablet given once daily is an alternative regimen to EPIVIR tablets 300 mg once daily plus ZIAGEN tablets 2 x 300 mg once daily as a component of antiretroviral therapy.
The following trial was conducted with the individual components of EPZICOM.
Therapy-Naive Adults
CNA30021 was an international, multicenter, double-blind, controlled trial in which 770 HIV-1-infected, therapy-naive adults were randomized and received either ZIAGEN 600 mg once daily or ZIAGEN 300 mg twice daily, both in combination with EPIVIR 300 mg once daily and efavirenz 600 mg once daily. The double-blind treatment duration was at least 48 weeks. Trial participants had a mean age of 37 years; were male (81%), white (54%), black (27%), and American Hispanic (15%). The median baseline CD4+ cell count was 262 cells per mm3 (range: 21 to 918 cells per mm3) and the median baseline plasma HIV-1 RNA was 4.89 log10 copies per mL (range: 2.60 to 6.99 log10 copies per mL).
The outcomes of randomized treatment are provided in Table 4.
| a Subjects achieved and maintained confirmed HIV-1 RNA less than 50 copies per mL (less than 400 copies per mL) through Week 48 (Roche AMPLICOR Ultrasensitive HIV-1 MONITOR standard test version 1.0). b Includes viral rebound, failure to achieve confirmed less than 50 copies per mL (less than 400 copies per mL) by Week 48, and insufficient viral load response. c Includes consent withdrawn, lost to follow-up, protocol violations, clinical progression, and other. |
||
|
Outcome |
ZIAGEN 600 mg q.d. plus EPIVIR plus Efavirenz (n = 384) |
ZIAGEN 300 mg b.i.d. plus EPIVIR plus Efavirenz (n = 386) |
|
Respondera |
64% (71%) |
65% (72%) |
|
Virologic failureb |
11% (5%) |
11% (5%) |
|
Discontinued due to adverse reactions |
13% |
11% |
|
Discontinued due to other reasonsc |
11% |
13% |
After 48 weeks of therapy, the median CD4+ cell count increases from baseline were 188 cells per mm3 in the group receiving ZIAGEN 600 mg once daily and 200 cells per mm3 in the group receiving ZIAGEN 300 mg twice daily. Through Week 48, 6 subjects (2%) in the group receiving ZIAGEN 600 mg once daily (4 CDC classification C events and 2 deaths) and 10 subjects (3%) in the group receiving ZIAGEN 300 mg twice daily (7 CDC classification C events and 3 deaths) experienced clinical disease progression. None of the deaths were attributed to trial medications.
14.2 Pediatric Subjects
ARROW (COL105677) was a 5-year, randomized, multicenter trial which evaluated multiple aspects of clinical management of HIV-1 infection in pediatric subjects. HIV-1–infected, treatment-naive subjects aged 3 months to 17 years were enrolled and treated with a first-line regimen containing abacavir and lamivudine, dosed twice daily according to World Health Organization recommendations. After a minimum of 36 weeks of treatment, subjects were given the option to participate in Randomization 3 of the ARROW trial, comparing the safety and efficacy of once-daily dosing with twice-daily dosing of abacavir and lamivudine, in combination with a third antiretroviral drug, for an additional 96 weeks. Virologic suppression was not a requirement for participation at baseline for Randomization 3. At baseline for Randomization 3 (following a minimum of 36 weeks of twice-daily treatment), 75% of subjects in the twice-daily cohort were virologically suppressed, compared with 71% of subjects in the once-daily cohort.
Of the 1,206 original ARROW subjects, 669 participated in Randomization 3. Subjects randomized to receive once-daily dosing (n = 336) and who weighed at least 25 kg received abacavir 600 mg and lamivudine 300 mg, as either the single entities or as EPZICOM.
The proportions of subjects with HIV-1 RNA less than 80 copies per mL through 96 weeks are shown in Table 5. The differences between virologic responses in the two treatment arms were comparable across baseline characteristics for gender and age.
| a Analyses were based on the last observed viral load data within the Week 96 window. b Risk difference (95% CI) of response rate is -2.4% (-9% to 5%) at Week 96. c Includes subjects who discontinued due to lack or loss of efficacy or for reasons other than an adverse event or death, and had a viral load value of greater than or equal to 80 copies per mL, or subjects who had a switch in background regimen that was not permitted by the protocol. d Other includes reasons such as withdrew consent, loss to follow-up, etc. and the last available HIV-1 RNA less than 80 copies per mL (or missing). |
||
|
Outcome |
Abacavir plus Lamivudine
|
Abacavir plus Lamivudine
|
|
HIV-1 RNA <80 copies/mLb |
70% |
67% |
|
HIV-1 RNA ≥80 copies/mLc |
28% |
31% |
|
No virologic data | ||
|
Discontinued due to adverse event or death |
1% |
<1% |
|
Discontinued study for other reasonsd |
0% |
<1% |
|
Missing data during window but on study |
1% |
1% |
16. How is Epzicom supplied
EPZICOM is available as tablets. Each tablet contains 600 mg of abacavir as abacavir sulfate and 300 mg of lamivudine. The tablets are orange, film-coated, modified capsule-shaped, and debossed with GS FC2 on one side with no markings on the reverse side. They are packaged as follows:
Bottles of 30 tablets (NDC 49702-206-13).
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) (see USP Controlled Room Temperature).
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Hypersensitivity Reactions
Inform patients:
- •
- that a Medication Guide and Warning Card summarizing the symptoms of the abacavir hypersensitivity reaction and other product information will be dispensed by the pharmacist with each new prescription and refill of EPZICOM and instruct the patient to read the Medication Guide and Warning Card every time to obtain any new information that may be present about EPZICOM. The complete text of the Medication Guide is reprinted at the end of this document.
- •
- to carry the Warning Card with them.
- •
- how to identify a hypersensitivity reaction [see Warnings and Precautions (5.1), Medication Guide].
- •
- that if they develop symptoms consistent with a hypersensitivity reaction they should call their healthcare provider right away to determine if they should stop taking EPZICOM.
- •
- that a hypersensitivity reaction can worsen and lead to hospitalization or death if EPZICOM is not immediately discontinued.
- •
- to not restart EPZICOM or any other abacavir-containing product following a hypersensitivity reaction because more severe symptoms can occur within hours and may include life-threatening hypotension and death.
- •
- that if they have a hypersensitivity reaction, they should dispose of any unused EPZICOM to avoid restarting abacavir.
- •
- that a hypersensitivity reaction is usually reversible if it is detected promptly and EPZICOM is stopped right away.
- •
- that if they have interrupted EPZICOM for reasons other than symptoms of hypersensitivity (for example, those who have an interruption in drug supply), a serious or fatal hypersensitivity reaction may occur with reintroduction of abacavir.
- •
- to not restart EPZICOM or any other abacavir‑containing product without medical consultation and only if medical care can be readily accessed by the patient or others.
Patients with Hepatitis B or C Co-infection
Advise patients co-infected with HIV‑1 and HBV that worsening of liver disease has occurred in some cases when treatment with lamivudine was discontinued. Advise patients to discuss any changes in regimen with their physician [see Warnings and Precautions (5.2)].
Lactic Acidosis/Hepatomegaly with Steatosis
Advise patients that lactic acidosis and severe hepatomegaly with steatosis have been reported with use of nucleoside analogues and other antiretrovirals. Advise patients to stop taking EPZICOM if they develop clinical symptoms suggestive of lactic acidosis or pronounced hepatotoxicity [see Warnings and Precautions (5.3)].
Immune Reconstitution Syndrome
Advise patients to inform their healthcare provider immediately of any signs and symptoms of infection as inflammation from previous infection may occur soon after combination antiretroviral therapy, including when EPZICOM is started [see Warnings and Precautions (5.4)].
Pregnancy Registry
Advise patients that there is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to EPZICOM during pregnancy [see Use in Specific Populations (8.1)].
Lactation
Instruct women with HIV-1 infection not to breastfeed because HIV-1 can be passed to the baby in the breast milk [see Use in Specific Populations (8.2)].
Missed Dose
Instruct patients that if they miss a dose of EPZICOM, to take it as soon as they remember. Advise patients not to double their next dose or take more than the prescribed dose [see Dosage and Administration (2)].
Availability of Medication Guide
Instruct patients to read the Medication Guide before starting EPZICOM and to re-read it each time the prescription is renewed. Instruct patients to inform their physician or pharmacist if they develop any unusual symptom, or if any known symptom persists or worsens.
EPIVIR, EPZICOM, TRIZIVIR, and ZIAGEN are trademarks owned by or licensed to the ViiV Healthcare group of companies.
The other brands listed are trademarks owned by or licensed to their respective owners and are not owned by or licensed to the ViiV Healthcare group of companies. The makers of these brands are not affiliated with and do not endorse the ViiV Healthcare group of companies or its products.
Manufactured for:
ViiV Healthcare
Durham, NC 27701
©2022 ViiV Healthcare group of companies or its licensor.
EPZ:18PI
|
MEDICATION GUIDE EPZICOM (ep' zih com) (abacavir and lamivudine tablets) |
|||
|
What is the most important information I should know about EPZICOM? EPZICOM can cause serious side effects, including:
|
|||
|
Symptom(s) | |||
|
Group 1 |
Fever | ||
|
Group 2 |
Rash | ||
|
Group 3 |
Nausea, vomiting, diarrhea, abdominal (stomach area) pain | ||
|
Group 4 |
Generally ill feeling, extreme tiredness, or achiness | ||
|
Group 5 |
Shortness of breath, cough, sore throat | ||
If your healthcare provider tells you that you can take EPZICOM again, start taking it when you are around medical help or people who can call a healthcare provider if you need one.
|
|||
|
What is EPZICOM? EPZICOM is a prescription medicine used with other HIV-1 medicines to treat HIV-1 infection. HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS). EPZICOM contains the prescription medicines abacavir and lamivudine. EPZICOM should not be used in children weighing less than 55 pounds (25 kg). |
|||
|
Do not take EPZICOM if you:
|
|||
|
Before you take EPZICOM tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines interact with EPZICOM. Keep a list of your medicines to show your healthcare provider and pharmacist when you get a new medicine.
|
|||
|
How should I take EPZICOM?
|
|||
|
What are the possible side effects of EPZICOM?
|
|||
|
|
||
|
|||
|
|
||
|
You may be more likely to get lactic acidosis or serious liver problems if you are female or very overweight (obese).
The most common side effects of EPZICOM include: |
|||
|
|
||
|
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of EPZICOM. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1‑800‑FDA‑1088. |
|||
|
How should I store EPZICOM?
Keep EPZICOM and all medicines out of the reach of children. |
|||
|
General information for safe and effective use of EPZICOM. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use EPZICOM for a condition for which it was not prescribed. Do not give EPZICOM to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for the information about EPZICOM that is written for health professionals. |
|||
|
What are the ingredients in EPZICOM? Active ingredients: abacavir and lamivudine Inactive ingredients: magnesium stearate, microcrystalline cellulose, sodium starch glycolate. Tablet film coating contains: OPADRY orange YS-1-13065-A made of FD&C Yellow No. 6, hypromellose, polyethylene glycol 400, polysorbate 80, and titanium dioxide.
EPZICOM, TRIUMEQ, TRIZIVIR, and ZIAGEN are trademarks owned by or licensed to the ViiV Healthcare group of companies. The other brands listed are trademarks owned by or licensed to their respective owners and are not owned by or licensed to the ViiV Healthcare group of companies. The makers of these brands are not affiliated with and do not endorse the ViiV Healthcare group of companies or its products. ©2022 ViiV Healthcare group of companies or its licensor. EPZ:16MG For more information call 1-877-844-8872. |
|||
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised: 12/2022 |
||
PRINCIPAL DISPLAY PANEL
NDC 49702-206-13
EPZICOM
(abacavir and lamivudine)
600 mg 300 mg
TABLETS
Notice to Authorized Dispenser:
Each time EPZICOM is dispensed, give the patient a Medication Guide and Warning Card.
Rx only
30 Tablets
Each tablet contains 600 mg of abacavir as abacavir sulfate and 300 mg of lamivudine.
Store at 25oC (77oF); excursions permitted to 15o to 30oC (59o to 86oF) (see USP Controlled Room Temperature).
See prescribing information for dosage information.
Keep out of reach of children.
Do not accept if membrane seal under cap is missing or broken.
Manufactured for:
ViiV Healthcare
Durham, NC 27701
Made in UK
Trademarks owned or licensed by ViiV Healthcare.
- ©2023 ViiV Healthcare or licensor.
- 62000000086425 Rev. 3/23
| EPZICOM
abacavir sulfate and lamivudine tablet, film coated |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - ViiV Healthcare Company (027295585) |
More about Epzicom (abacavir / lamivudine)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (6)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: antiviral combinations
- En español