DAN DAN 5335 Pill - white round, 8mm
Pill with imprint DAN DAN 5335 is White, Round and has been identified as Trihexyphenidyl Hydrochloride 2 mg. It is supplied by Watson Laboratories, Inc.
Trihexyphenidyl is used in the treatment of Extrapyramidal Reaction; Cerebral Spasticity; Parkinson's Disease and belongs to the drug class anticholinergic antiparkinson agents. FDA has not classified the drug for risk during pregnancy. Trihexyphenidyl 2 mg is not a controlled substance under the Controlled Substances Act (CSA).
Images for DAN DAN 5335
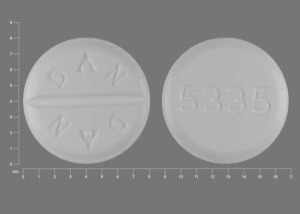



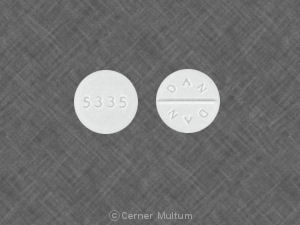
Trihexyphenidyl Hydrochloride
- Imprint
- DAN DAN 5335
- Strength
- 2 mg
- Color
- White
- Size
- 8.00 mm
- Shape
- Round
- Availability
- Prescription only
- Drug Class
- Anticholinergic antiparkinson agents
- Pregnancy Category
- N - Not classified
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Watson Laboratories, Inc.
- National Drug Code (NDC)
- 00591-5335
- Inactive Ingredients
-
silicon dioxide,
calcium phosphate dibasic anhydrous,
lactose monohydrate,
magnesium stearate,
microcrystalline cellulose,
sodium starch glycolate type A potato
Note: Inactive ingredients may vary.
More about trihexyphenidyl
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (24)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: anticholinergic antiparkinson agents
- Breastfeeding
- En español
Patient resources
Other brands
Professional resources
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
